17 Transaxillary Approach to Endoscopic Breast Augmentation
Summary
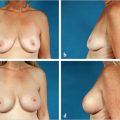
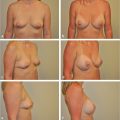
This chapter reviews the current status of transaxillary breast augmentation using primarily an endoscopic approach. The procedure is described in detail together with its limitations and outcomes. It is illustrated with appropriate clinical examples.
Key Teaching Points
Endoscopic approach to this procedure has revolutionized its safety and predictability.
The precision of transaxillary breast augmentation is improved with the use of the endoscopic approach.
Gel-filled implants are more stressed during insertion using the transaxillary approach.
Care should be taken to dissect inframammary folds precisely to avoid asymmetry.
The endpoint of inferomedial pectoral division is the visualization of the prepectoral fascia.
17.1 Introduction
Transaxillary breast augmentation was first performed by Peterson in 1976. The procedure was essentially a blind one and its main drawback was that it was difficult to adequately release the inferomedial origin of pectoralis major to adequately lower the implant pocket. In addition, if bleeding occurred, it was difficult to stop without converting to an open approach through an inframammary incision. The procedure was almost exclusively performed into the subpectoral plane using saline-filled implants. Transaxillary endoscopic breast augmentation was developed at the Emory clinic in 1993, again utilizing saline-filled implants. It experienced a resurgence of interest over the ensuing decade, which has waned somewhat with the increasing use and acceptance of silicone gel–filled implants in the United States. It has been a substantial improvement over the original nonendoscopic approach.
17.2 Endoscopic Subpectoral Transaxillary Augmentation
Endoscopic transaxillary augmentation provides the same benefits as the standard open transaxillary augmentation, including short, inconspicuous incisions away from the central breast area, ready access for submuscular implant placement, possibly improved preservation of breast sensibility, and avoidance of parenchymal breast dissection, which may reduce the incidence of postoperative infection, scarring, and fibrosis.
Endoscopic techniques can help reduce the scar from 4 to 5 cm to an average of 2.5 to 3 cm. Furthermore, the muscle and fascia to be divided are at right angles to the endoscopic view, making the axillary approach particularly well suited to endoscopic techniques. Control of the inframammary crease is greatly enhanced by accurate division of the pectoralis major muscle origins and prepectoral fascia under direct vision. Because the muscle is divided with electrocautery, bleeding is avoided or minimized, and when bleeding does occur, hemostasis is readily achieved with coagulation of the vessel under magnified endoscopic visualization. Although difficult to quantitate objectively, postoperative pain may be reduced by eliminating most of the blunt dissection. Control of the subpectoral cavity is maintained by careful endoscopic dissection of the pocket. The ability to clearly visualize the subpectoral pocket under magnification permits cavity revision, implant exchange, or capsulotomy through the same incision if necessary. The procedure is well suited to the use of saline-filled implants; gel-filled implants require significant compression to insert through these very small incisions and are difficult to remove once placed. It is the author’s preference to only use this technique with saline-filled devices.
Technical Pearl
However, the advent of the Keller funnel has increased the ease with which implants can be inserted through a transaxillary incision, making the procedure safer for gel implant usage. In addition, several authors have explored the use of the transaxillary approach to perform subfascial breast augmentation with reported success.
Benefits of the Transaxillary Endoscopic Approach
Magnified view.
Bright illumination during dissection.
Clear visualization of the release of the pectoralis origin inferomedially.
Easier access to hemostasis within the pocket.
Less reliance on blunt dissection.
Access to either the subpectoral or subfascial plane of dissection.
Better lowering of the inframammary fold when necessary.
17.2.1 Operating Room Setup and Patient Positioning
When an endoretractor is used, the most comfortable position for the surgeon is one that provides a direct view of the endoscope and the monitor in a straight line. For transaxillary endoscopic procedures, this alignment is most effectively accomplished with the surgeon positioned above the patient’s extended arm and shoulder and the monitor positioned at the foot of the operating table. Although this positioning does not provide ideal alignment, the monitor will not have to be repositioned as the surgeon moves to the opposite side of the table. The light source, video cable, cautery cord, and suction tubing are brought up from the foot of the operating table and also will not require repositioning. To provide adequate space for the surgeon, the operating table is moved approximately 2 feet away from the anesthesia cart.
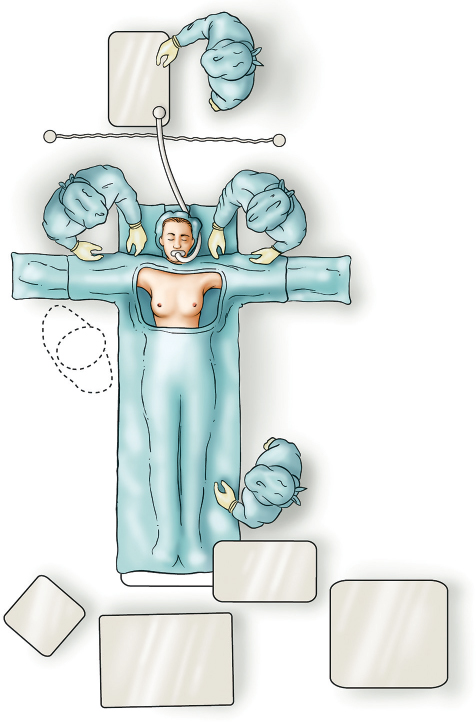
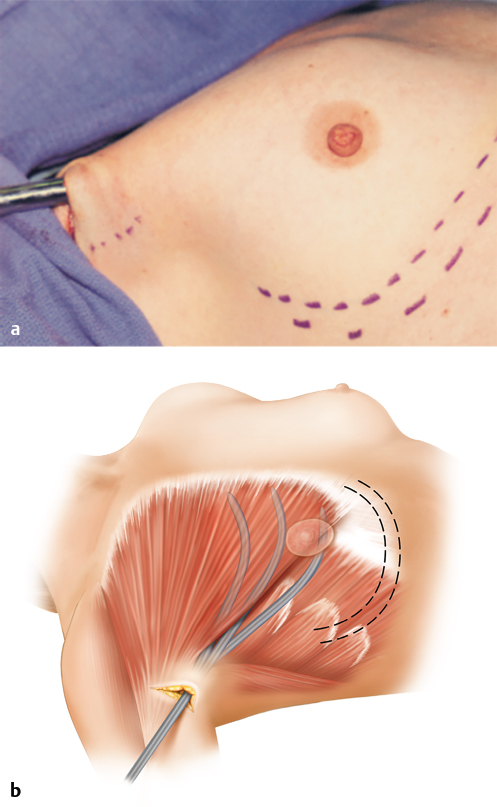
The patient is placed on the operating table in a supine position and secured with her arms abducted. After anesthesia is induced, the patient’s arms are carefully positioned and secured to the arm boards at 90 degrees of abduction with soft roll padding. The patient must be positioned so as to allow the table to be adjusted to a full upright (sitting) position for optimal pocket and implant evaluation and adjustment at the end of the operation. Standard padding precautions at pressure points are observed. The incision site is infiltrated with 1% lidocaine with 1:100,000 epinephrine, and the entire anterior chest and axilla from the clavicles to the costal margin are then prepped and draped. The drapes are adjusted so that the surgeon can stand above either arm without being contaminated by the anesthesia equipment (▶Fig. 17.1).
Surgical Plan
Axillary incision approach.
Subpectoral dissection of the implant pocket.
Endoscopic release of medial and inferior pectoralis muscle fibers.
Development of symmetrical subpectoral pockets.
Placement of round, smooth-surface saline or gel implants.
17.2.2 Markings
A prominent transverse axillary crease is marked for the access incision.
Because of the reach afforded by the endoscope and endoscopic instrumentation, this line can be placed at essentially any point within the axilla; however, I generally place it as high in the axilla as will allow a comfortable angle of the scope to enter the subpectoral pocket, providing good visualization of the entire medial and inferior portions of the breast. The endoscopic axillary incision can be as short as 2 cm (measured at 2.5 cm on stretch), but the procedure is easier to perform if the incision is 2.5 to 3 cm. However, the primary benefit of endoscopic transaxillary augmentation is control of the dissection, which should not be sacrificed for a reduction of incision length (▶Fig. 17.2).
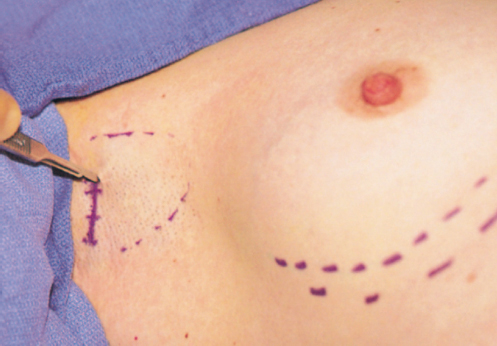
The incision and initial subpectoral pocket dissection are facilitated if the surgeon stands above the patient’s extended arm. The axillary skin is placed under tension with the nondominant hand, and the incision is made as marked through the skin only. The anterior edge of the incision is then elevated with sharp double hooks. Spreading scissors dissection creates a tunnel directly medial to the thick anterior axillary fold marking the pectoral muscle.
Technical Pearl
Elevating the anterior skin margin and maintaining this dissection in a superficial plane minimize the risk of injury to the intercostobrachial nerve.
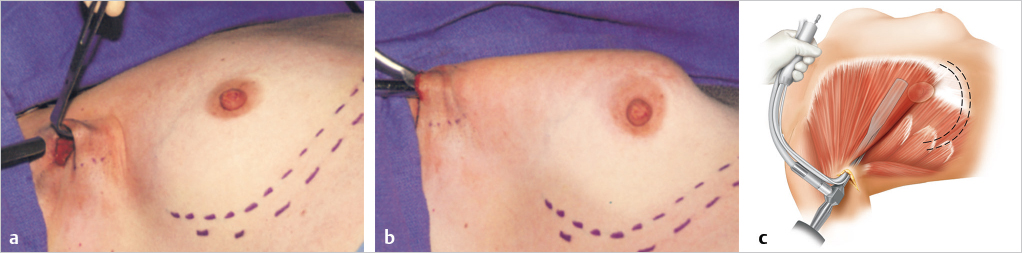
The subcutaneous tissue underlying the incision tends to restrict instrument insertion and mobility unless it is opened widely. This is accomplished by sharp division of the tissue immediately underneath the ends of the incision and blunt creation of the subcutaneous tunnel to the lateral pectoral border with the scissors (▶Fig. 17.3).
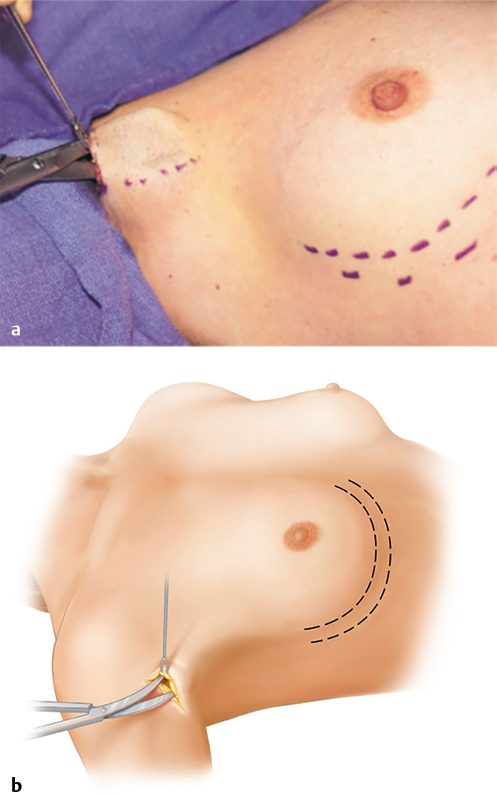
17.2.3 Development of the Subpectoral Space
Blunt scissors are placed with the tips closed against the lateral pectoral border and slid posteriorly underneath the muscle in a medial direction toward the sternum. Resistance to advancement of the scissors indicates the lateral pectoral fascia has been encountered. This resistance is overcome by firm pressure, and the scissors tips are guided into the subpectoral space. Spreading the tips to the side will enlarge the opening in the lateral pectoral fascia (▶Fig. 17.4).
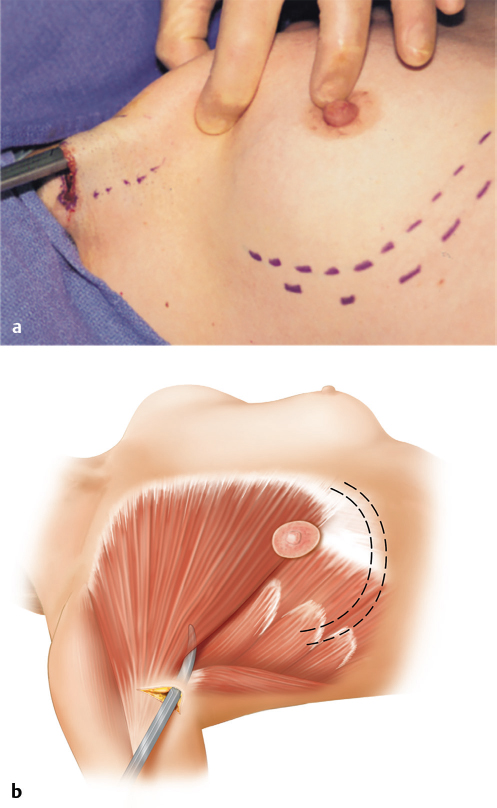
After the incision is extended down to the subpectoral space, the index finger is used to begin the dissection of the subpectoral space; the finger is gently swept between the upper portions of the pectoralis major muscle and the underlying ribs (▶Fig. 17.5).
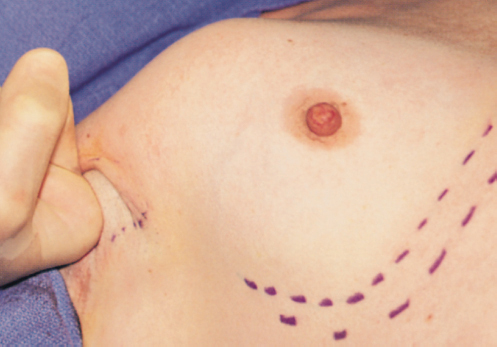
A 36-F blunt breast dissector is placed through this opening into the subpectoral space. To facilitate placement of instruments, the blunt breast dissector is used to sweep the superolateral pocket laterally. The skin incision then lies at the apex of the optical cavity. A gentle sweeping motion within this subpectoral space divides any loose areolar tissue separating the posterior surface of the pectoralis major from the chest wall inferiorly and pectoralis minor fibers superiorly.
Technical Pearl
A vigorous sweeping motion of the entire space must be avoided, because this creates bleeding that obscures the endoscopic view.
Before the endoscopic approach was developed, I used blunt division of the pectoralis major muscle from the chest wall with the Agris-Dingman breast dissectors. However, pectoral muscle fibers, when viewed endoscopically, have been shown to be intact after blunt dissection. The modest bleeding that ensues made visualization for division of these fibers more difficult. In addition, it appears that vigorous dissection may lead to increased postoperative bruising. The use of endoscopy has eliminated these problems (▶Fig. 17.6).
17.2.4 Insertion of the Endoretractor
While the skin incision and initial pocket dissection are being completed by the surgeon, the scrub nurse removes the 20-cm long, 10-mm diameter, 30-degree endoscope from the warm fluid bath. Alternatively, an antifogging solution may be applied to the endoscope at this time. The endocamera is connected to the endoscope, and if a fixating pin is present on the camera coupler, it is tightened to prevent camera rotation. The light cord is attached, and the camera is white-balanced using a clean laparotomy sponge as a reference. The video image is inspected simultaneously, and any initial problems with fogging or blurring are addressed before the scope is inserted into the endoretractor.
The surgeon then moves to a position above the patient’s extended arm. The endoretractor is inserted through the incision and into the subpectoral space. The endoscope is not placed within the retractor sheath until the retractor is fully in place within the subpectoral space to avoid smearing the lens with blood. Once the sheath is in place, the endoscope is advanced into the retractor and focused. As the endoscope is inserted farther into the sheath, the image is followed on the video screen. For the same reason, the endoretractor is lifted off the chest wall before the end of the endoscope emerges from the end of the tubing. The light cord is inserted into its retaining slot on the retractor handle. While the lift is maintained, the endoretractor is advanced or withdrawn and pivoted from a medial to a lateral position to adjust the endoscopic view and permit inspection of the subpectoral space. The focus ring on the camera is adjusted as necessary to obtain a clear view. The endoscope can then be advanced fully into the retractor sheath without fear of blood contacting the lens and distorting visualization (▶Fig. 17.7).
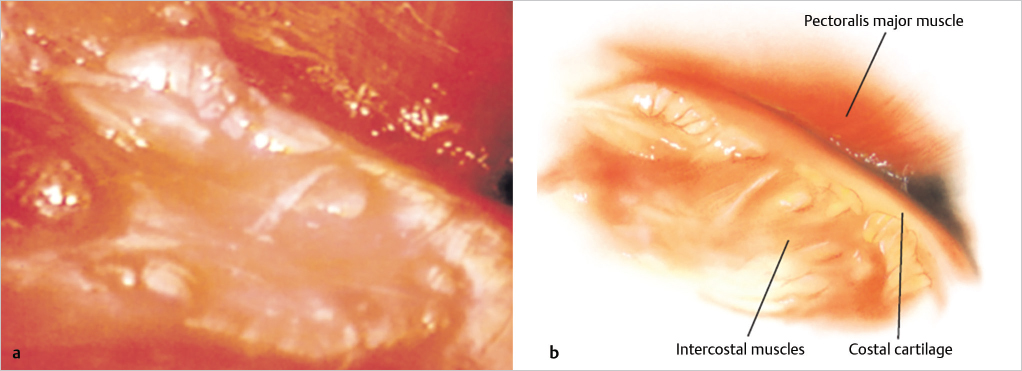
The costal cartilage and intercostal muscle are visible on the inferior aspect of the monitor. The fibers of the pectoralis major muscle can be seen clearly. In the medial and superomedial views, the fibers are seen to course anteriorly and cephalad; inferiorly, the fibers course more directly toward the endoscope. Pressing a finger along the breast allows the surgeon to correlate the internal structures and the external markings (▶Fig. 17.8).
Technical Pearl
Orientation is critical at this point, because a lack of orientation may result in the surgeon penetrating the pleural space when dissecting in the wrong direction.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree