20 Managing Complications in Breast Augmentation
Summary
Breast augmentation is a highly successful operation. However, it carries with it a significant risk of complications. Many of these are related to operative technique. Meticulous attention to detail, pocket development, implant positioning, and bacterial contamination reduction techniques can reduce these issues. This chapter seeks to explore the commonest complications and how to manage them.
Key Teaching Points
Hematomas and infection increase capsular contracture rates.
The use of a 14-point protocol to reduce bacterial contamination reduces both infection and capsular contracture.
Symmetry should be checked in the erect position.
Acellular dermal matrices help to secure capsulorrhaphy position.
Subfascial or dual-plane augmentation can reduce double bubble deformity.
Wide-spaced breasts are best corrected with subfascial conversion.
20.1 Introduction
Breast augmentation is a commonly performed procedure with a high degree of success. Careful planning, attention to detail, and meticulous technique ensure low complication rates. Despite all these factors, complications occur, and when they do, they can be distressing as these are cosmetic self-pay patients who often have little financial leeway for dealing with the expense and emotional trauma of a poor outcome.
Complications can be divided into three major categories: acute (7–10 days), intermediate (2–6 weeks), and long term.
Complications
Acute:
Hematoma.
Asymmetry.
Wound dehiscence.
Infection.
Intermediate:
Infection.
Implant malposition.
Double bubble deformity.
Widely spaced breasts.
Long term:
Capsular contracture.
Implant malposition.
Inframammary crease asymmetry.
Synmastia.
Rippling.
Ptosis of the gland off the implant.
20.2 Acute Complications
20.2.1 Hematoma
Hematoma after breast augmentation is a rare complication occurring in less than 1% of patients in most series. It usually occurs within the first 24 hours after surgery and most commonly within the first 6 to 12 hours postoperatively, although late hematomas can be found several weeks after surgery. Causes may include poor attention to hemostasis, elevated blood pressure secondary to poorly controlled pain postoperatively as well as sudden rises in blood pressure caused by factors such as coughing and straining.
Patients usually present with a sudden increase in breast tenderness and swelling with associated bruising. Swelling occurs rapidly and can cause major discomfort including difficulty taking a deep breath when intramammary pressure is significantly elevated. Blood may begin to leak from an incision and wound dehiscence can occur in severe cases. This may cause implant exposure.
Management of Hematoma
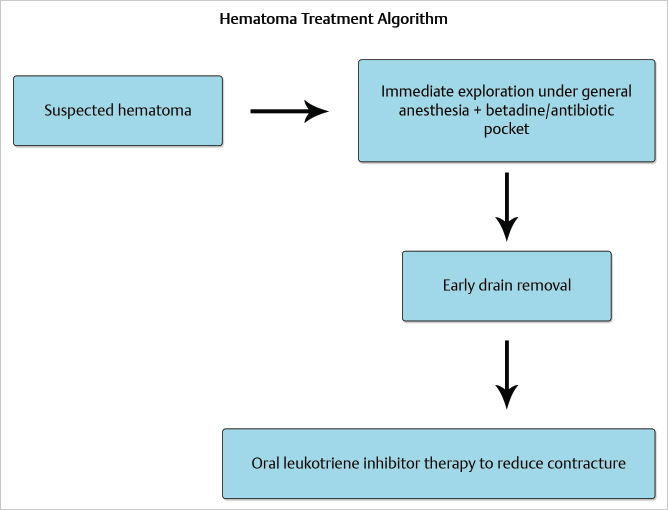
Immediate exploration under general anesthesia is indicated for breast hematomas. Attempts at suction evacuation of a hematoma in a physician’s office should be discouraged at all costs. Attempts at such intervention may lead to contamination of the breast pocket in less than ideal surgical conditions with increasing risk for infection and later capsular contracture. The patient should be returned to the operating room immediately and under general anesthesia, the wound should be opened, the implant removed, and the hematoma evacuated. The pocket should be washed copiously with a 50:50 solution of Betadine and triple antibiotic solution after inspecting for hemostasis. As with most hematomas, it is often the case that there are no distinct sites of hemorrhage to be found. If a discreet bleeding vessel is noted, it should be carefully cauterized or if particularly large such as an internal mammary perforator, it can be oversewn with a figure-of-eight suture for additional security.
Technical Pearl
Once hemostasis has been secured, the implant pocket should be thoroughly washed to remove any traces of clot from the operative field. Blood is intensely inflammatory in the extravascular space, and periprosthetic hemorrhage has been strongly implicated in the development of capsular contracture.
Prior to closure, consideration may be given as to whether to insert a drain. If hemostasis is secure and generalized oozing has not been a problem, a case can be made for not inserting a drain as they are almost invariably contaminated to their tips within 24 hours of insertion. If a drain is inserted, it should be removed early to reduce risks of infection. Postoperatively, the patient should be monitored closely for the development of capsular contracture. The use of breast massage could be encouraged although I personally am not a great believer in its ability to reduce contracture. If there is any suggestion of capsular contracture developing, the use of oral leukotriene inhibitors could be attempted (▶Fig. 20.1).
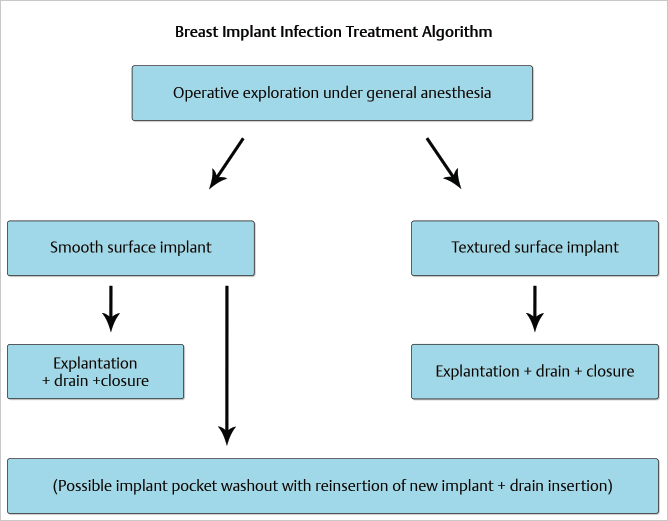
20.2.2 Infection
Acute periprosthetic infection following breast augmentation is a rare but disastrous complication. Most series report the complication in approximately 1% of cases or less, with the use of drains increasing the risk. Superficial cellulitis occurs in approximately 1 to 1.2% of cases while deep periprosthetic infection is reported in 0.5-1% of patients. The symptoms include a feeling of malaise and fever, associated with redness and tenderness of the breast. There may be associated wound healing complications of the incision, or drainage from the wound. Patients presenting acutely with this problem may not have had time to mount a significant leukocytosis.
A patient presenting with mild cellulitis around the incision but with no other symptoms suggestive of overt implant infection may be treated with oral antibiotics with close observation. If there is any suggestion that the implant is frankly infected, the patient should be taken back to the operating room immediately for exploration and lavage.
Technical Pearl
At the time of exploration, the safest course of action is to simply remove the implant, wash out the pocket, and insert a drain followed by wound closure. The patient is allowed to recover fully, and re-augmentation can be considered 6 to 12 weeks later following complete wound healing.
If smooth surface implants are used, a case could be made for carefully washing the wound with Betadine solution and triple antibiotic solution, inserting a new implant and drain insertion. Sforza reported the results in 17 patients treated with one-stage implant salvage. All the patients achieved implant salvage with no increased risk of capsular contracture.
By far the safest course of action should always be implant removal and secondary re-augmentation at a later date once the wound has completely healed.
Following treatment, patient should be observed carefully for contracture, as infection significantly increases the risk of this occurring. Again, the use of leukotriene inhibitors could be considered (▶Fig. 20.2).
20.2.3 Wound Dehiscence
Earlier wound dehiscence is typically a function of either inadequate attention to detail regarding closure, or excessive tension of the implant within the overlying skin and breast tissue. Closure should always be performed with at least two and preferably three layers. The first layer should anchor the breast tissue and Scarpa’s fascia to the chest wall followed by a second subcutaneous layer of absorbable sutures and then running subcuticular layout. An overlying layer of cyanoacrylate superglue completes the seal on the wound together with Steri-Strip application.
Care should be taken not to insert excessively large implants, especially in women who are very small-breasted to start off with. Women with small breast and tight chest wall skin envelopes tend to be at greater risk for wound healing problems if large devices are placed at the index operation. Careful preoperative planning as well as three-dimensional digital imaging can go a long way toward preventing this complication entirely.
20.2.4 Implant Malposition
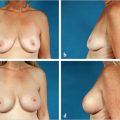
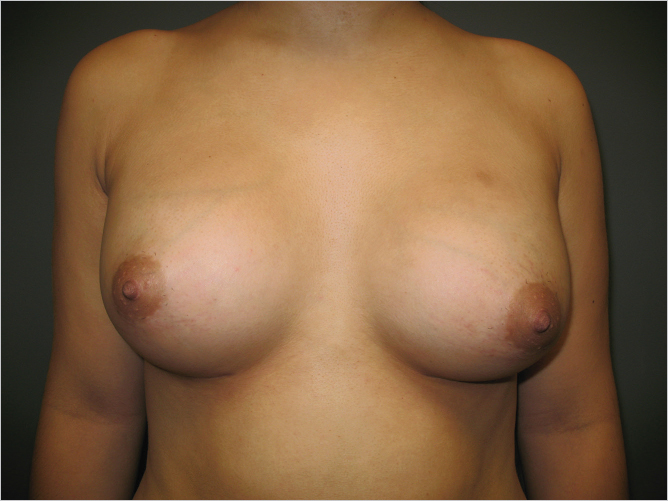
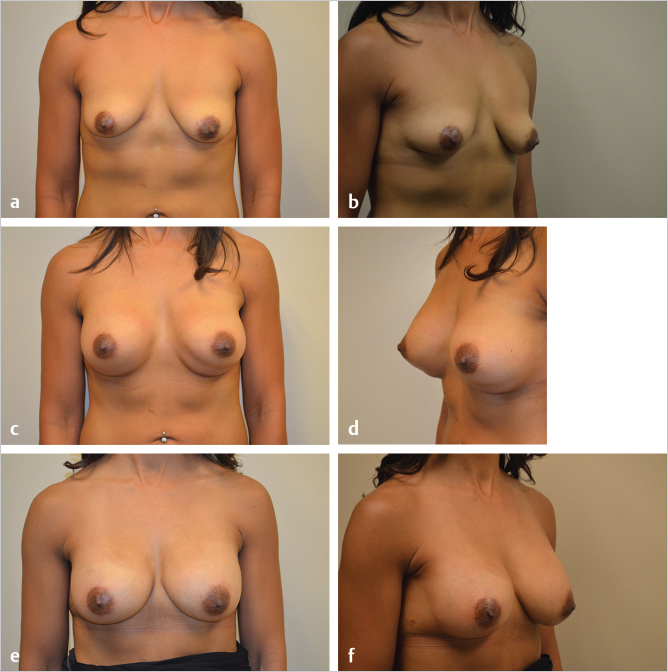
It goes without saying that careful symmetrical implant placement is a critical component of unsuccessful breast augmentation surgery. While implant malposition is the most common cause of asymmetry after augmentation, it should be remembered that prior developmental asymmetry can create postoperative asymmetry as shown in ▶Fig. 20.3, where asymmetric nipple position is still evident despite correct implant position. This patient declined periareolar mastopexy to correct the nipple asymmetry as it did not bother her.
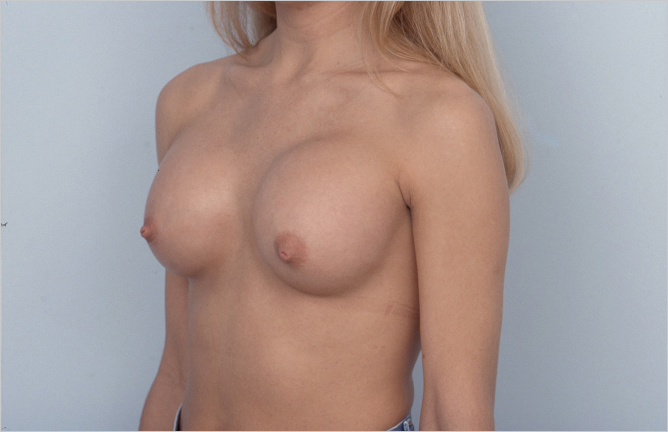
Essential to correct implant placement is the development of precise, symmetrical pocket dissection. The footprint of the implant pockets should be exactly where the surgeon desires the implants to lie at the end of the procedure. Excessive pocket development in any direction can result in implant displacement in an unwanted direction. A common error with inexperienced surgeons is lack of lower pole pocket dissection resulting in excessively high placement of the implant. This is particularly easy to do during endoscopic transaxillary augmentation as illustrated by the case below (▶Fig. 20.4).
Great care should be taken at the inframammary fold (IMF) to ensure that this is not overdissected, allowing descent of the implants along the chest wall beneath the breast gland.
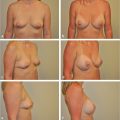
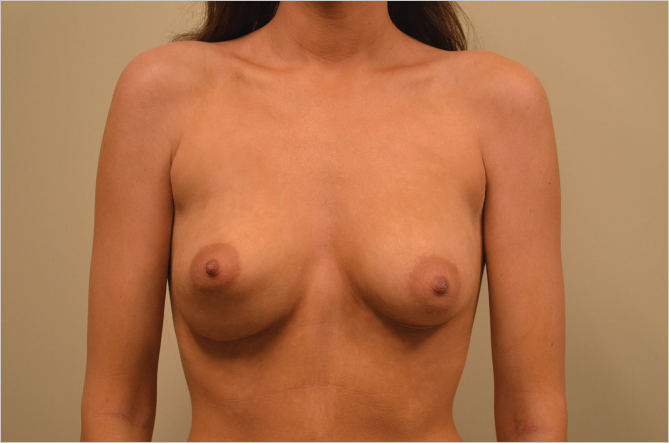
Overdissection of the lower pole can result in either lowering of the IMF compared with the contralateral crease or the generation of a double bubble effect if the patient has a very short IMF to nipple distance, both of which are seen in the patient below (▶Fig. 20.5).
Similarly, overdissection of the medial aspect of the breast can result in synmastia with direct contact between the implants resulting in distortion or obliteration of the cleavage as seen in this patient with severely misshapen breasts after prior augmentation. She also had an underlying pectus excavatum deformity (▶Fig. 20.6).
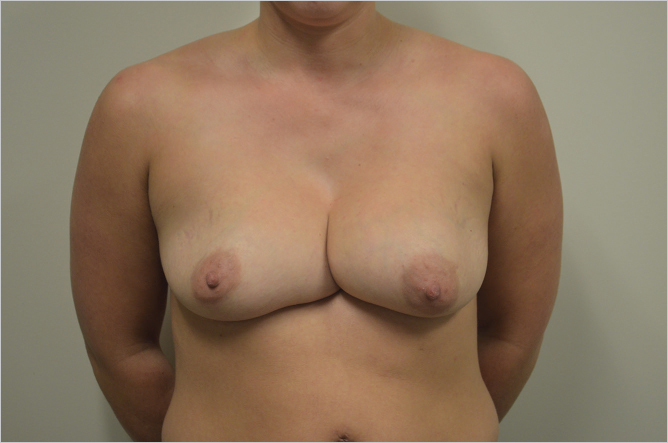
This patient had subpectoral augmentation through an inframammary approach, and overdissection laterally caused a double bubble type deformity at the lateral breast crease causing unsightly bulging and loss of lateral breast definition (▶Fig. 20.7).
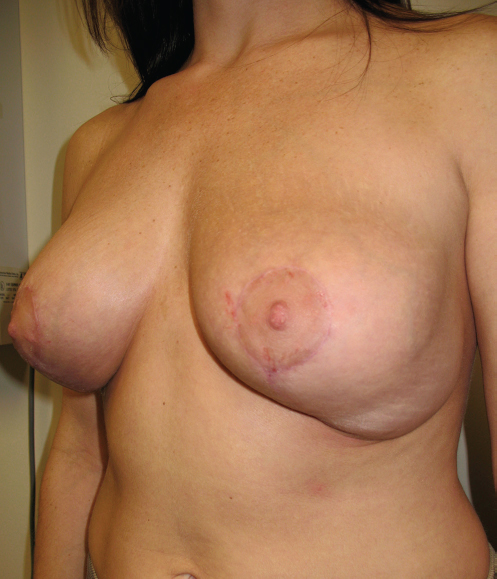
Technical Pearl
The anatomical landmarks of the breast should be respected at all times. Any attempt at changing either the medial or lateral breast border or inframammary fold should be undertaken with caution and with careful planning in a very deliberate fashion.
Correction of implant malposition will usually require either capsulorrhaphy or capsulotomy depending on the direction in which the implant needs to be moved. Excessively high implant position will require inferior capsulotomy in order to lower the device to the appropriate level.
Overdissection medially, laterally, or inferiorly, will require some form of capsulorrhaphy. For limited amounts of capsular tightening, the so-called “popcorn” technique may be all that is required. This involves touching the electrocautery tip to the existing capsule and applying current to allow the capsular tissue to tighten and shrink at the point of contact. Simple suture plication can be difficult to maintain, particularly when large implants are exerting significant force against the repair over time. All too often, these sutures pull away from the chest wall component where tissues are often too thin and weak to retain suture fixation. A far better approach is to raise capsular flaps which can either be imbricated or transposed up onto the chest wall at a higher level to pull the breast tissue into the desired position for implant pocket control. Whenever performing capsular flaps, I always suture them with soft, braided, nonabsorbable sutures such as Ethibond (Ethicon Inc).
By far the most powerful and effective repair for retaining a capsulorrhaphy is the use of some form of acellular dermal matrix (ADM). My personal preference is for Strattice, which is a porcine ADM that is characterized by very little tendency to stretch over the long term. Artia is the latest version of Strattice, (Lifecell Corporation, New Jersey) in that it handles more like human skin with easier suturing characteristics while retaining the strength of Strattice. When sutured to the chest wall, ADM provides a very robust barrier to implant migration and has the added advantage of reducing capsular contracture. Given that many patients presenting secondarily with late implant malposition are suffering from capsular contracture, the incorporation of ADM into the repair provides a very powerful tool for dealing with both implant malposition and the tendency for recurrent contracture.
Excessively High Implant Placement
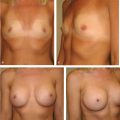
As mentioned earlier, excessively high implant positioning is usually due to over-enthusiastic superior dissection with inadequate pocket development in the lower pole. The net result is more implant fill in the superior portion of the pocket with less projection in the inferior quadrant. If the pectoralis major muscle has not been adequately released, the implant cannot sit at the correct level in the natural inframammary crease, resulting in abnormally high implant position (▶Fig. 20.8).
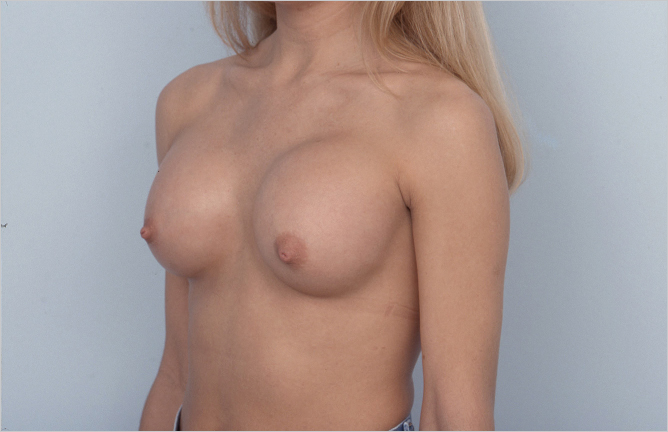
Treatment of this problem is usually operative. Attempts can be made to use elasticated compression banding of the upper chest, but this is rarely successful in any but the most subtle of cases. Surgical intervention requires reopening of the incision with inferior capsulotomy to lower the implant to the correct level in relation to the inframammary crease. If the crease is very tight and well defined, it may require radial scoring in order to achieve an adequate release to create enough room for the implant to sit comfortably in its new position. In the case illustrated above, the transaxillary incision used by the initial surgeon was reopened, and using an endoscopic approach, inferomedial capsulotomy was performed without disturbing the implants themselves. This resulted in adequate lowering of the implants to the point where the nipples lay closer to the equator of the implant instead of at the junction of the middle and lower thirds, as was the case with her preoperative situation (▶Fig. 20.9).
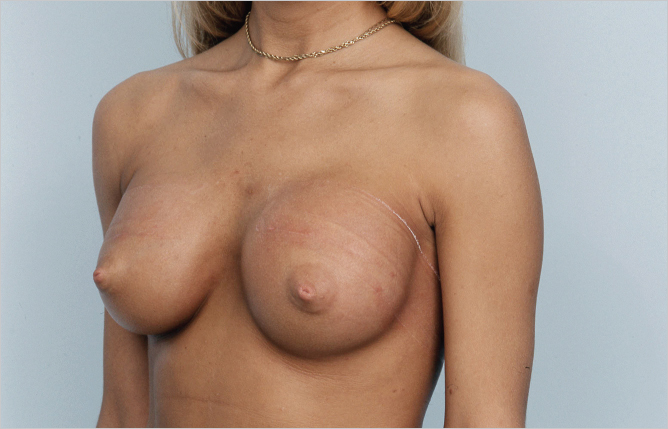
20.2.5 Overdissection of the Inframammary Crease
Overenthusiastic dissection of the inframammary crease is probably the commonest cause of implant malposition in augmentation mammoplasty. Two factors commonly give rise to this problem:
Overdissection at the inframammary crease while releasing pectoralis major in submuscular dual-plane augmentation.
Failure to respect and re-create the IMF during closure after augmentation.
Release of the pectoralis major during dual plane augmentation requires good visualization of the inferolateral border of the muscle. Great care should be taken to ensure that any muscle division is done at the inframammary crease and not below it. In patients with a short IMF to nipple distance, it is all too easy to perform excessive dissection while attempting to lower the crease and perform radial scoring to expand it in order to prevent double bubble deformity. The implant is then able to slide down the chest wall beneath Scarpa’s fascia, creating gross asymmetry at the fold.
When closing the inframammary incision after breast augmentation, it is important to tack Scarpa’s fascia to the chest wall, particularly in patients who have had fold adjustment performed, in order to prevent appearance of the double bubble deformity. This is especially true of very small-breasted women who have a tightly defined IMF with short IMF to nipple distance (▶Fig. 20.10, ▶Fig. 20.11).
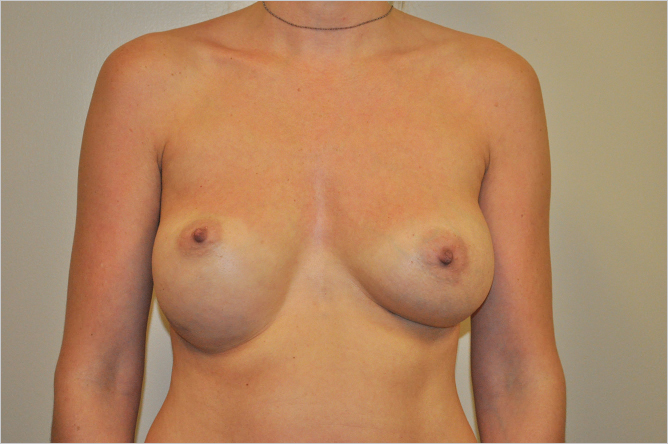
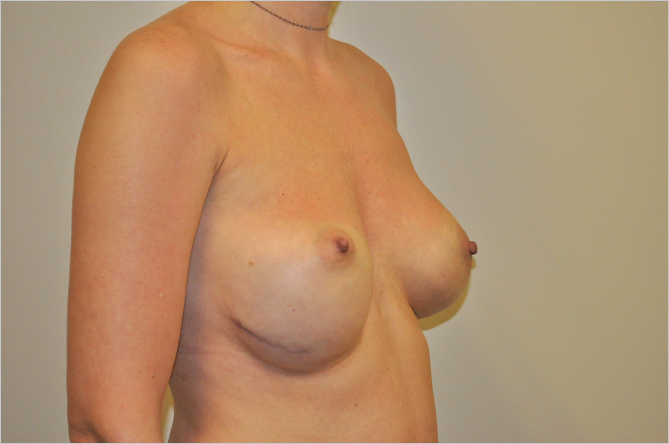
Options for Correction of the Asymmetric Inframammary Fold
Popcorn capsulorrhaphy.
Suture capsulorrhaphy.
Capsular flap capsulorrhaphy.
Acellular dermal matrix-reinforced capsulorrhaphy.
Popcorn Capsulorrhaphy
The term “popcorn capsulorrhaphy” refers to the technique of touching the implant capsule with electrocautery causing scattered small burns in the capsular tissue resulting in shrinkage and fibrosis of the capsule. The shrinkage effectively creates a capsulorrhaphy effect. It is not a tremendously powerful technique for major defects but can be useful as an adjunct to any of the other forms of capsulorrhaphy or in isolation in treating cases of mild capsular laxity requiring tightening. It can be performed very precisely, and the effect is immediately seen, allowing the surgeon to perform additional capsular work when necessary in order to achieve the desired effect. This technique can be used in any portion of the capsule to achieve selected tightening to create implant displacement as necessary (▶Fig. 20.12).
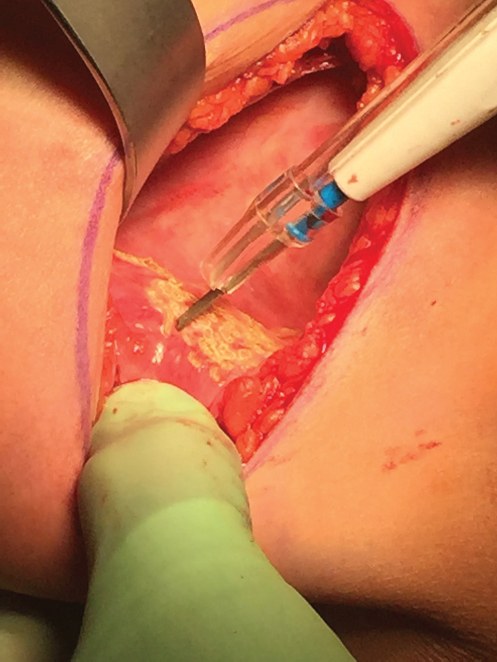
Suture Capsulorrhaphy
Suture capsulorrhaphy involves creating two parallel raw surfaces using electrocautery and then suturing the two surfaces together with either absorbable or nonabsorbable sutures. Typically, one line is inscribed within the capsule on the inferior breast mound while the other is incised parallel along the chest wall at the level to which the breast crease needs to be elevated. The two raw surfaces are then sutured together with interrupted or running the sutures to fix the crease at its new level. When performing this technique, which I now do rarely, I use interrupted Ethibond sutures as they are softer, nonabsorbable, and hold knots well. Absorbable sutures such as Vicryl or PDS lose 50% of their tensile strength at 4 weeks which may be too early to hold against the constant pressure of the implant’s weight on the inferior suture line. This can lead to premature failure of the repair and recurrence of the deformity. In my experience, suture capsulorrhaphy has not been highly successful as the intact sutures tend to pull out from the somewhat rigid, thin capsule of the posterior implant pocket wall. It is not unusual to re-explore a breast where someone has performed a suture capsulorrhaphy in the past, only to find a neat row of sutures lying in the anterior capsule with nothing adhering to the posterior capsule (▶Fig. 20.13).
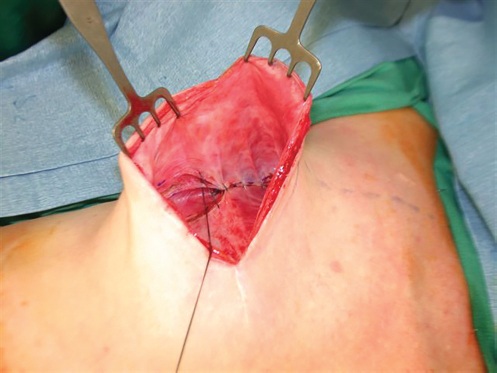
Capsular Flap Technique
The use of capsular flaps has been a major advance in the success of capsulorrhaphy. The tissue tends to be more robust and can be sutured in multiple sites as a “vest over pants” type of repair to provide increased strength when compared with a single suture line within a capsular incision. The flap is incised adjacent to the chest wall and elevated up onto the breast mound as needed to provide a sewing edge. It is then advanced onto the chest wall to the appropriate level for fixation to the posterior capsule. The posterior capsular tissue on the chest wall can be incised with electrocautery to provide a raw surface for adhesion. The capsular flap is then sutured down at multiple points with nonabsorbable sutures to provide fixation, while the flap becomes adherent in the postoperative period. This technique has provided far better correction and longevity than simple suture capsulorrhaphy, which has more of a reputation for suture line failure and recurrence (▶Fig. 20.14).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree