Vascular Lesions
Benjamin C. Garden
Jerome M. Garden
DEFINITION
Vascular lesions can be classified into two main categories: proliferative and malformations.1
Vascular proliferative lesions are characterized by a proliferation of blood vessels.
Infantile hemangiomas are the most common form of vascular proliferative lesions.
Other vascular proliferative lesions include kaposiform hemangioendotheliomas, tufted angiomas, and pyogenic granulomas.
Vascular malformations are characterized by vessels with abnormal structure and normal endothelial cell turnover. Types of malformations include capillary, venous, lymphatic, arterial, and arteriovenous malformations (AVMs).
Capillary malformations include nevus simplex (salmon patch) and nevus flammeus (port-wine stain, PWS).
Venous malformations (VM) are a fairly common slowflow type of vascular malformation present at birth. Various names used to describe these lesions include venous angioma and cavernous angioma.
Lymphatic malformations (LM) are composed of interconnected lymphatic channels.2
Older terminologies used include cavernous lymphangioma, lymphangioma circumscriptum, and cystic hygroma.
LM can be further differentiated into macrocystic and microcystic depending on the size of the involved channels.
AVMs are rare vascular malformations consisting of both arterial and venous components with AV shunting.2
ANATOMY
Vascular lesions may occur on any cutaneous surface but are most commonly found on the head and neck.
Hemangiomas have been noted to have a nonrandom distribution, with the majority of lesions occurring on the central face at sites of development fusion.3
Capillary malformations are often, but not always, unilateral.
PATHOGENESIS
Hemangiomas are thought to occur due to a first-trimester developmental error regarding angiogenesis or a result of embolized placental cells. There is a suggested autosomal dominance inheritance.4
Although their exact cause is unknown, PWSs evolve due to a progressive ectasia of the superficial vascular plexus. This progression is likely due to an abnormal neural regulation of blood flow. PWSs have significantly less nerve density and a higher vessel to nerve ratio.4
NATURAL HISTORY
Infantile hemangiomas become evident in the first weeks of life and exhibit a proliferative phase, with continued growth, until around 8 to 12 months. This is followed by a plateau phase, followed by a period of spontaneous regression as the lesion involutes.
Initial involution is usually noted with a color change from the original bright red to dull red or gray.
It is estimated that completed involution occurs at a rate of 10% per year, such that 30% have involuted by 3 years of age, 50% by 5 years of age, and greater than 90% by 9 to 10 years of age.3
These lesions occasionally have residual changes, including telangiectasias, atrophy, scarring, or fibrofatty material.
Ulcerations occur in 5% to 13% of infantile hemangiomas.
Most frequent locations include the lip, perineum, and intertriginous areas.
Ulcerations are painful, increase the risk of infection, and can cause scarring and textural changes.3
Vascular malformations tend to be present at birth and persist for a lifetime.
PWS may darken progressively over many years, and occasional lesions develop secondary vascular blebs on their surface. They may also become hypertrophic later in life.
VMs are present at birth but may not become obvious until later in life. Occasionally, a patient first notices the lesion after trauma to the overlying area. In those cases, the trauma may cause the underlying vessels to temporally enlarge allowing the patient to notice it for the first time.
PATIENT HISTORY AND PHYSICAL FINDINGS
The preoperative evaluation for patients with vascular lesions includes a detailed history and exam to determine the type of lesions and to assess the risks of the patient having any associated complications or syndromes.
Infantile hemangiomas may occur as superficial, deep, or mixed lesions.
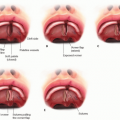
Superficial lesions tend to appear as bright red nodules or plaques (FIG 1A).
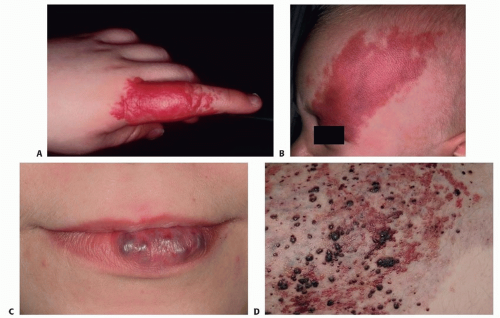
FIG 1 • A. Superficial infantile hemangioma. B. Port-wine stain. C. Venous malformation. D. Mixed microcystic lymphatic and venous malformation.
Deep hemangiomas are subcutaneous nodules, and usually present with an overlying blue hue, venous network, or telangiectatic vessels.
Hemangiomas in certain locations may suggest a higher probability of complications or associated findings.3
With periorbital hemangiomas, an ophthalmologic examination is generally recommended in many cases.
Larger lesions of the beard area require evaluation for potential airway obstructions.
Hemangiomas on the tip of the nose, the lip, and the parotid areas can have very slow involution periods.
PWSs are normally easy to recognize with their well-defined red macular stains (FIG 1B). They do not proliferate, but will grow proportional to the child’s growth. Patients with PWS should be assessed for any associated syndromes that would necessitate additional referrals.
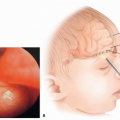
The most common syndrome is Sturge-Weber syndrome (SWS), which most commonly occurs with a PWS occurring in the distribution of the ophthalmic branch of the trigeminal nerve (V1).
PWS can be associated with many other syndromes including Klippel-Trenaunay, Parkes-Weber, Proteus, Cobb, Rubinstein-Taybi, and Beckwith-Wiedemann syndrome.
VMs usually present as blue to purple nodules in the skin (FIG 1C). They may have prominent surrounding veins and calcified phleboliths within the lesion. These lesions may expand with dependent drainage of venous blood; this may be elicited by placing the patient in the Trendelenburg position or by performing the Valsalva maneuver.2
Most VM are asymptomatic, although they may occasionally become painful in association with their gradual enlargement and pressure on surrounding structures.
They may be found in association with syndromes, including Maffucci and blue rubber bleb nevus syndrome.
Macrocystic LMs, sometimes referred to as cystic hygromas, present as large, translucent masses under normal-appearing skin. Microcystic LM usually present as erythematous to flesh-colored papules, which may be somewhat translucent, and have traditionally been compared to the appearance of frog spawn (FIG 1D).
AVMs may be clinically pulsatile and warm and have a thrill. They present as a vascular stain at birth, with increasing erythema and size with age. They can spontaneously bleed.
IMAGING
One should strongly consider doing a liver MRI in those patients having five or more hemangiomas due to the increased potential of having visceral lesions.3
Large segmental facial hemangiomas should prompt imaging to assess possible PHACES syndrome:
Posterior fossa malformations
Hemangiomas
Arterial anomalies
Coarctation of the aorta
Eye abnormalities
Sternal or supraumbilical raphe
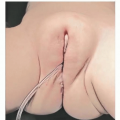
Perineal hemangiomas should prompt evaluation for PELVIS syndrome:
Perineal hemangiomas
External genital malformations
Lipomyelomeningocele
Vesicorenal abnormalities
Imperforate anus
Skin tags
In patients with a PWS, imaging may be necessary if there is concern for a related syndrome, such as SWS. MRI is the preferred screening modality of SWS. Patients with a vascular malformation located in the midline lumbar location may have an underlying AVM, as seen in Cobb syndrome, which would be imaged by an MRI.
VM can be confirmed or the depth of involvement can be ascertained with MRI with and without contrast.
NONOPERATIVE MANAGEMENT
Generally, appropriate treatment for infantile hemangiomas is nonintervention.5 However, in those lesions where there is high visibility, social concern or functional impairment, as well as complications such as ulceration, infection or bleeding, therapy may be warranted.4,6
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree