Surgical Treatment of Congenital Midline Nasal Mass
Patrick A. Gerety
Scott P. Bartlett
DEFINITION
Congenital midline nasal masses are a rare occurrence but require a thorough workup for an intracranial component, which then necessitates a transcranial resection.
These masses typically represent one of three diagnoses: dermoid cyst, encephalocele, or glioma.
A nasal dermoid sinus cyst (NDSC) is lined by stratified squamous epithelium and contains dermal adnexal structures such as hair follicles and sebaceous glands. The cystic contents are sebum. If the mass has a punctum with drainage to the outside world, it is a dermoid sinus; otherwise, it is a dermoid cyst.
A nasal encephalocele is an extracranial protrusion of a cerebrospinal fluid (CSF)-containing dural mass. Twentyfive percent of all encephaloceles are in the nasofrontal region (sincipital). If brain matter is within the mass, this is referred to as meningoencephalocele.
A nasal glioma represents extracranial brain matter with no patent CSF connection.
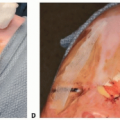
Intracranial extension—extension of midline nasal masses from extracranial nasal location to the anterior cranial fossa
Transcranial approach—a combined extracranial and intracranial resection with reconstruction
ANATOMY
Formation of a midline nasal mass results from disruption of early embryologic processes. The exact theory of embryology responsible for the formation of transcranial nasal masses remains debated.1 Several operating theories lend rationale to the occurrence and anatomy of these masses. Some of these theories include the prenasal space theory, the nonseparation theory, and the ectodermal inclusion theory.
Transcranial nasal congenital masses are believed to exist for two important reasons (FIG 1):
Failure of normal regression of a transcranial embryologic structure (dural diverticulum)
Embryologic contact of surface and neural ectoderm
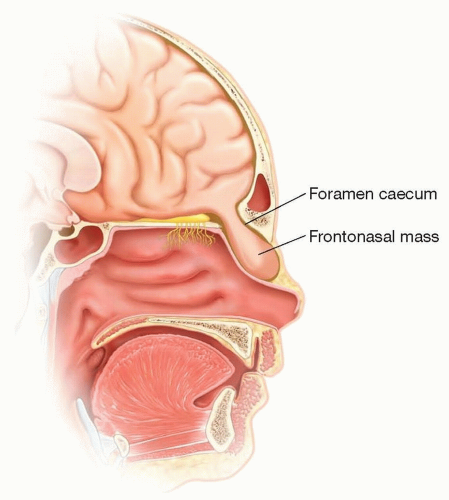
Per one theory, a dural diverticulum from the anterior cranial fossa normally exits the cranium via the nasofrontal junction (the fonticulus (naso) frontalis) and ends between the nasal bones and nasal soft tissues.
This diverticulum normally involutes and can be identified as the foramen cecum. This foramen has no contents typically but may contain a fibrous stalk.
If this process is disrupted, a persistent transcranial connection may be present at birth.
Importantly, the neural ectoderm of this diverticulum makes contact with the surface ectoderm of nasal skin. Normally, this connection also regresses. If it does not, it may pull skin elements beneath the nasal skin resulting in an NDSC.
The transcranial connection may occur at a number of anterior cranial fossa midline structures: cribriform plate, crista galli, foramen cecum, nasofrontal junction, or any combination of these.
Anterior cranial encephaloceles can also occur via the nasofrontal, nasoethmoidal, or naso-orbital bony junctions.
PATHOGENESIS
The etiologic basis of this occurrence is likely multifactorial with a genetic component. Family history and multiple siblings have been identified with these masses. Midline nasal masses are not related to any known syndromes or other known congenital anomalies.
Conceptually and for disease categorization, these masses particularly the encephalocele are related to the embryologic failure of anterior neuropore (normally embryologic day 24) during early embryologic development.
A cranial defect must be present for either of the three main masses to be seen nasally and transmitted transcranially.
Dermoid cysts likely result from trapping of skin structures subcutaneously as the dural diverticulum involutes normally.
Encephalocele represents a neural tube defect. Terminology varies depending upon what the extracranial mass contains.
Meningocele—meninges only
Meningoencephalocele—meninges and brain matter
Encephalocele and glioma represent neuroectodermal malformations.
Dermoid cysts and sinuses represent a somatic (surface) ectodermal malformation.
Intranasal gliomas and encephaloceles likely have a herniation point in the anterior cranial fossa that is more posterior (ie, posterior to the cribriform plate) as compared to masses that are seen on the nasal dorsum.
NATURAL HISTORY
Congenital midline nasal masses are rare with an approximate incidence of 1 in 30 000 live births.
These masses, particularly encephaloceles, occur in higher rates in Asia (Western incidence, 1 in 35 000; Asian incidence, 1 in 5000).
NDSC—midline nasal cysts represent 3% to 12% of head and neck dermoid cysts.2
Intracranial extension of midline nasal dermoids has been found to be common among these masses on the order of 25% to 45% of cases.
Encephalocele—approximately 80% are occipital and 20% frontonasal (sincipital).3
Glioma
Approximately 15% have a transcranial connection; thus, nasal resection without intracranial exclusion can lead to CSF leak and meningitis.
3:1 male predominance4
Sixty percent are extranasal, 30% are intranasal, and 10% are mixed.
There is a reported association with other nonspecific congenital abnormalities (often quoted as approximately 40%). However, several series have not shown the same association. A transcranial midline nasal mass may occur in isolation.
Risk of meningitis—with good soft tissue coverage and no sinus punctum, patients are likely at low risk of developing a meningeal infection even in the presence of intracranial CSF connection.
PATIENT HISTORY AND PHYSICAL FINDINGS
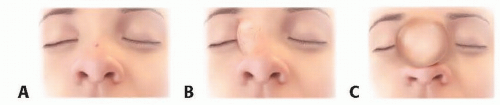
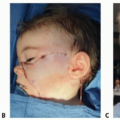
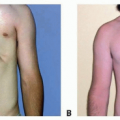
Visible and palpable midline nasal mass (FIG 2)
May occur from the columella to the radix but most commonly along the nasal dorsum
Wide nasal bridge
Hypertelorism—this may be true hypertelorism with total orbital widening or more commonly in this case interorbital hypertelorism with widening of only the medial orbital wall.
May be present in 70% of encephalocele patients
Normal interorbital distance increases from approximately 18 mm in infancy to 25 to 30 mm in adulthood.
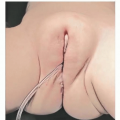
NDSC are firm, round masses.
NDSC may present with a punctum. The punctum may contain hair, which is a pathognomonic sign of the diagnosis.
If a punctum is present, patients are more likely to present with infection.
Gliomas are firm, round masses.
Surface telangiectasia is present and this is pathognomonic.
May be extranasal, intranasal, or both
Does not transilluminate or change in size
Intranasal glioma may cause nasal airway obstruction or nasolacrimal dysfunction. Airway obstruction is particularly problematic in neonates/infants who are obligate nasal breathers.
Encephalocele may occur through one of three sutures (junctions): nasofrontal, nasoethmoidal, and naso-orbital.
Often appear blue in color
Transillumination is pathognomonic because of CSF content.
Size change with crying may occur (Valsalva).
Size change may also be elicited by compression of the internal jugular veins (Furstenberg sign).
Patients may present with CSF leak or frank meningitis.
The cranial exit point has been found to be fairly variable and may occur at the nasofrontal suture but also through other cranial bone junction points.
This can be difficult to differentiate on physical examination.
IMAGING
Because of the occurrence of transcranial extension, there should be a low threshold for obtaining cross-sectional imaging of congenital midline nasal masses.
In general, both a maxillofacial CT scan and brain MRI are obtained.
CT scan provides a better examination of bony distortion and bony relationships.
MRI is thought to be more sensitive and specific for transcranial soft tissue extension and for brain structural examination.5
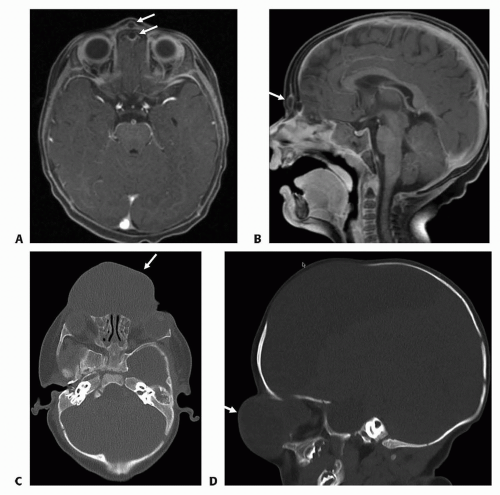
Typical imaging findings (FIG 3):
Interorbital distance—widened
Frontonasal cranial defect
Nasal bone—displaced and angulated
Bifid crista galli
Tissue diagnosis via biopsy is contraindicated in these masses before a noninvasive, cross-sectional imaging workup has been performed.
DIFFERENTIAL DIAGNOSIS
The following are other diagnoses outside of NDSC, encephalocele, and glioma, which may occur on the nasal dorsum congenitally.6
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree