Vaginal and Vulvar Reconstruction
Elizabeth B. Yerkes
Julia Corcoran
DEFINITION
Neovaginal creation or augmentation of the vaginal canal may be requested in several congenital conditions and in acquired vaginal stenosis. This chapter will address vaginal and vulvar reconstruction in emerging and young adults. It does not include vaginoplasty or labioplasty techniques that may be applied in reconstruction of urogenital sinus in cases of ambiguous genitalia, intersex states, and differences of sex development.
Vaginal agenesis or congenital vaginal atresia (Mayer-Rokitansky-Küster-Hauser syndrome)
Vaginal agenesis or insufficiency in the setting of complete androgen insensitivity and intersex/differences of sex development
Classic bladder exstrophy or cloacal exstrophy
Vaginal stenosis after early childhood surgery (congenital adrenal hyperplasia, intersex/differences of sex development, or persistent cloaca)
Secondary changes after pelvic malignancy or graft versus host disease (GVHD)
Vulvar reconstruction in the form of monsplasty and labioplasty may be requested to align the laterally displaced or offset mons tissue and pubic hair and to reconfigure and stabilize the labia minora after pubertal development in congenital conditions such as exstrophy-epispadias and its covered variants.
Reduction labioplasty may be requested to address physically bothersome bulk of the labia minora.
ANATOMY
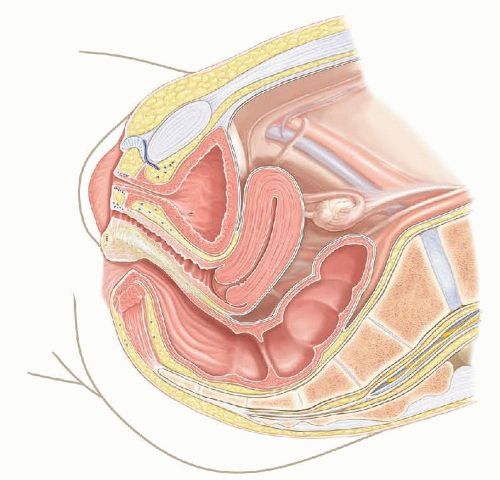
The paired Müllerian (paramesonephric) bodies fuse in the midline and contact the urogenital sinus. The sinovaginal bulb is induced and the vaginal plate is formed.
Müllerian development occurs within first 10 weeks of gestation.
The labia minora are formed from the urogenital folds and the labia majora and mons from the labioscrotal folds (FIG 1).
PATHOGENESIS
Absent or incomplete development of the Müllerian bodies results in absence or atresia of the female genital tract.
Ovaries have a different embryologic development and are spared.
Failure of the fusion of the Müllerian bodies results in duplication of the vagina or unilateral obstruction or atresia.
Anomalous descent of the urorectal septum in dividing the cloaca, as in anorectal malformations, may interfere with fusion of the Müllerian bodies.
Renal development and a portion of skeletal development occur at a similar time to Müllerian development, so anomalies may coexist.
Pelvic diastasis associated with abdominal wall defects such as bladder exstrophy and cloacal exstrophy also results in nonfusion of the labial and mons tissues. Bifid clitoris also occurs in these conditions.
Thickening of the labia minora with puberty results in extensive variation in size and shape. Sufficient hypertrophy may occur that some clothing and physical activity are physically bothersome.
Hygiene and tissue health may be a concern in young women with limited mobility or ability for self-care.
NATURAL HISTORY
Vaginal agenesis or atresia does not resolve over time, but maturation of the vulvar tissues and proximal vaginal segment, if present, may facilitate the reconstruction.
If a uterus and proximal vagina are present, judiciously allowing the vagina to distend with menstrual products may bring it closer to perineum or expand to create more tissue for pull-through reconstruction.
Local tissue flaps or nongenital grafts can be used to bridge the distance to the perineum.
Surgical benefits of this natural resource must be balanced against pain and the potential for retrograde menstrual flow and peritoneal irritation and deposits that could compromise future fertility.
PATIENT HISTORY AND PHYSICAL FINDINGS
Patient age and autonomy as well as family values and motivations will impact discussions about goals and surgical options.
General discussions may have already involved the parent due to the professional relationship from infancy.
Once ready to discuss interventions, private discussions with the patient are essential but the parent must also be included for surgical discussions in minors.
Patient readiness and goals for long-term results must be ascertained.
Readiness to undertake daily vaginal dilations for indefinite period.
Goal may be simply unobstructed menstrual flow or use of tampon.
Understanding of need for progressive dilations, and potential additional procedures, thereafter for functional canal
Readiness or anticipated timeline for intimate sexual contact.
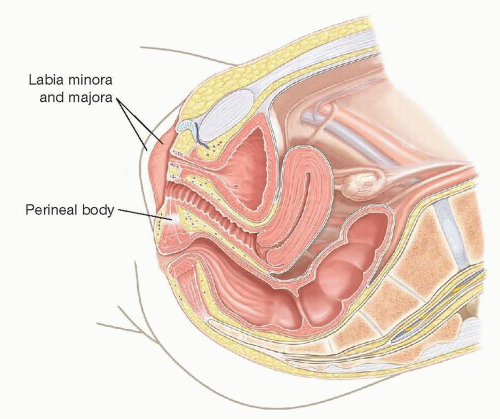
FIG 1 • Sagittal view of pelvic anatomy depicting relationship of urethra, vagina, and rectum in typical development.
Maturity level to be able to discuss with partner
Desire for heterosexual penetrative intercourse vs nonpenetrative sexual activities
Anticipated life events (graduation, prom, marriage) that could be complicated by surgical recovery or complications
Conflicts between family values/guarantor values and patient’s desire for surgery
Anatomical and patient factors that may inform management
Quality of distal UG sinus/vaginal pouch and vulvar skin
Presence of functional uterus and proximal vaginal segment
Degree of estrogenization and maturation of tissues
Location of hair bearing tissues
Prior pelvic or genital surgery
Prior abdominal, urinary, or rectal surgical procedures
Prior chemotherapy or regional radiation therapy in setting of pelvic malignancy
Inflammatory bowel disease
Dermatologic conditions
Continence status for urine and stool
Patient factors that may impact perioperative care and success
Resilience
Willingness to ask for and accept support in the perioperative period
Compliance
Lack of privacy to complete required care (college dorm)
Timing relative to next expected menstrual period
History of hypercoagulable state or bleeding diathesis
Increased surgical risk in terms of bleeding after deep dissection or from graft donor site
Increased risk of perioperative thromboembolic complications after pelvic surgery and prolonged bed rest
Gastrointestinal or dermatologic conditions
Continence status for urine and stool
IMAGING
Imaging is not specifically required for surgical planning, although pelvic ultrasound or MRI is often obtained in the course of establishing certain diagnoses and individual anatomy.
NONOPERATIVE MANAGEMENT
Daily serial dilation program can achieve a functional vaginal canal in properly selected and motivated candidates.
Preferred initial therapy in vaginal agenesis, CAIS, and other intersex/differences of sex development with a vaginal pit or pouch to guide positioning of dilator.
May be used to progressively dilate vaginal stenosis.
Tissue health and surgical scarring may limit success.
Assess maturity and commitment to goals of program
Program is voluntary and timing should be dictated by patient.
Ongoing office support is important to verify technique and to enhance success.
Daily or twice daily dilation for 10 to 20 minutes
Goal is to achieve and maintain vaginal depth while increasing caliber.
Dilations may cease when consistently sexually active but may otherwise need maintenance dilations.
SURGICAL MANAGEMENT
Vaginoplasty
The individual anatomic situation will dictate whether neovagina construction or augmentation of the caliber of the vaginal canal is required.
Skin or mucosal grafts, nongenital pedicled flaps (bowel), and local tissue flaps may be incorporated in the vaginal reconstruction.
Choice of donor tissue may be based upon surgeon experience, patient expectations and preferences, donor tissue availability, harvest site morbidity and scarring, and donor tissue properties.
Monsplasty and labioplasty
Inferomedial rotation of mons tissue in bladder exstrophy or cloacal exstrophy
Addresses soft tissue asymmetry
Allows for concomitant midline abdominal scar revision, with or without umbilicoplasty if desired, and removal of non-hair-bearing midline tissue
Melds with labia majora to create greater privacy for the clitoris, labia minora, and vaginal vestibule
Supports concomitant revision of asymmetric or mobile labia minora tissue and/or vaginoplasty
Mobilization of tissue flaps to allow cosmetic and functional coverage of glans clitoris if overexposed after prior feminizing genitoplasty
Reduction labioplasty
Excision or reconfiguration of physically bothersome hypertrophic labia minora
Preoperative Planning
Review of prior operative notes in individuals with prior pelvic reconstructive procedures.
A thorough pelvic examination with or without cystoscopy and vaginoscopy is often helpful in surgical planning and patient preparation and counseling.
Offer anesthesia or sedation due to the invasive and physically or emotionally uncomfortable nature of the examination.
Schedule one or more detailed consultations with patient to review all surgical options in the context of individual anatomy and goals.
Surgical scheduling with consideration of menstrual cycle
Avoid menses on day of surgery and for first postoperative week when vaginal stent or mold is used.
Continuous menstrual suppression may be considered perioperatively if cycles predictably unpredictable.
Mechanical bowel preparation in patients with anorectal anomaly and in those who will have bowel vaginoplasty. Enema advised in other vaginal cases.
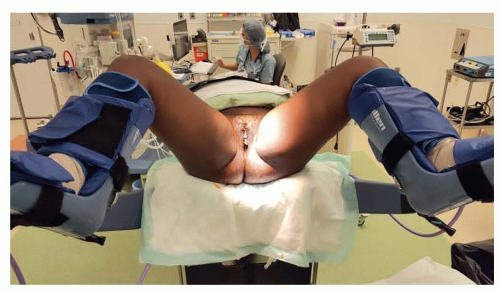
Positioning
Sequential compression devices in place prior to positioning
Position in dorsal lithotomy with all pressure points carefully padded (FIG 2).
Adequate padding on bed and/or protective barrier on sacral bony prominence, if present
Gentle support of atypical lumbosacral anatomy, if present
Attention to limited range of motion at hip or lower extremity with some diagnoses
Standard to low dorsal lithotomy is preferred.
Minimizes stress on the spine and nerves in those with pre-existing pathology.
Acute neurologic change may occur with more exaggerated or prolonged lithotomy.
Allows participation of cosurgeon from step stool at patient’s hip.
TECHNIQUES
▪ Augmentation of Caliber of Vaginal Canal: Vaginoplasty and Introitoplasty
This technique is not neovagina or vaginal replacement but rather augmentation of the existing vaginal canal caliber or circumference (TECH FIG 1).
Introital, distal, or mid vaginal stenosis
Globally narrow but patent vaginal canal
Angulation of vaginal canal at introitus
Anteriorly positioned and narrow introitus relative to axis of vaginal canal after having undergone exstrophy repair techniques without deep mobilization of the urogenital complex
Laterally positioned and angulated introitus in vaginal duplication of cloacal exstrophy
Remarkably increases vaginal caliber but cannot increase vaginal length due to presence of uterus.
Relevant to exstrophy where cervix may sit more distally even when well supported.
Enlargement of the Vaginal Canal Caliber
Radial incisions made at 3:00 and 9:00 with electrocautery (TECH FIG 2).
Lateral location of incisions least likely to injure urethra, bladder base, or rectum.
Incisions sequentially deepened to expand vaginal caliber without compromising support.
Desired caliber for penetrative intercourse: two fingers, spread to 2.5 to 3 fingerbreadths.
For tight stenosis or atresia or with anterior introitus/angulated canal, the authors prefer oral (buccal) mucosa free graft.
For isolated introital stenosis, a local perineal skin flap can be utilized, alone or in combination with free graft, if it does not deform or compromise surrounding anatomy.
Resurfacing With Native Tissues: Buccal Mucosa
Grafts are harvested from one or both cheeks, depending upon amount of substrate required.
Graft is defatted and meshed by hand with strokes of scalpel. Alternatively, it may be intermittently temporarily folded and incised with fine scissors (TECH FIG 3A).
Triangular defects will be created bilaterally by the radial incisions.
The rectangular meshed buccal graft is cut obliquely into two triangles.
4-0 Vicryl suture is preplaced in apex of graft and advanced to apex of the triangular incision.
The meshed graft is expanded to fill the defect and quilted into the dissected bed with resorbable suture (TECH FIG 3B,C). With the introitus and labia at rest, vagina appears normal, not gaping.
Insert vaginal mold (TECH FIG 3D).
An inflatable vaginal mold with central drainage port is not currently commercially available, but the concept elements are ideally applied to employed substitute.
Graft stability without undue pressure on urethra; sterile; impermeable; central drainage port for mucus and blood
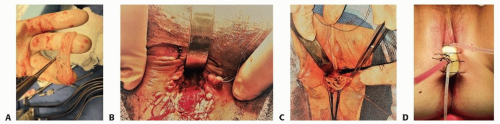 TECH FIG 3 • A. Harvested buccal tissue has been defatted and hand meshed to cover wider defect. For use in bilateral defects, it is divided obliquely into two triangular grafts to advance into defect. B,C. Two uses of buccal for augmentation procedures. B. Triangular grafts quilted in place bilaterally. Note that native vaginal mucosa remains along posterior wall. C. Young adult with deferred repair of urogenital sinus. The vagina was mobilized toward the perineum and spatulated. Buccal mucosa is being inset to bridge to gap and to improve introital caliber. D. Vaginal mold carefully inserted. Labia sutured across to prevent expulsion.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|