INTRODUCTION
Foam Sclerotherapy in the Treatment of Leg Veins
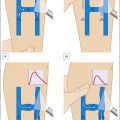
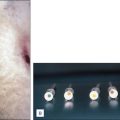
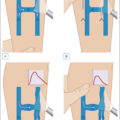
Venous disease is treated using various interventional methods, depending on the size and location of the veins. Among these techniques, foam sclerotherapy stands out as a highly effective, minimally invasive treatment, especially for nontruncal veins like tributaries of the saphenous veins. It is commonly employed for varicose veins, reticular veins, and incompetent perforating veins, particularly those that are small, tortuous, or difficult to treat areas with other modalities such as endovenous laser therapy or radiofrequency ablation ( Fig. 11.1 ). It has also gained favor over ambulatory phlebectomy in terms of improved cosmesis for nonbulging varicosities ( Fig. 11.2 ).
Algorithm of treatment options for venous disease of the legs.
Multiple linear scars from a previous ambulatory phlebectomy tracking down the posterior lateral leg.
(Courtesy of Sirunya Silapunt, MD, DABVLM, RPhS; Department of Dermatology, University of Texas Medical School at Houston, Houston, TX.)
History
The development of modern sclerotherapy was shaped by two key factors: the ability to access veins through the invention of the syringe and the need to address the complications associated with early sclerosants. In the 17th century, Sir Christopher Wren and Robert Boyle pioneered intravenous access, using a goose quill as a syringe to inject alcohol into dog veins. In 1665, Johann Sigismund Elsholtz used a plant alkaloid as the first sclerosant to treat a venous ulcer in a Duke’s bodyguard. However, early sclerosants, such as ferric chloride and iodine-tannin, caused severe complications, including skin necrosis and pulmonary embolism. These adverse effects led to a temporary ban on sclerotherapy in Lyon in 1894. In the 20th century, further developments improved safety. Joseph Hodgson’s 1815 theory of thrombosis in varicose veins led to the use of sclerotherapy. Alexander Wood and Charles Gabriel Pravaz’s invention of the modern syringe enabled more precise injections.
The first foam sclerotherapy technique was introduced in 1939 by McAusland, who mixed sodium morrhuate with air to create a foam that could be injected more efficiently than traditional liquid sclerosants. In 1944, Orbach further refined this concept, creating a frothy mixture of air and sodium tetradecyl sulfate (STS) for treating varicose veins. The procedure gained widespread popularity in 2000, following Lorenzo Tessari’s development of a method to prepare foam using two disposable syringes and a three-way tap. This simplified technique, which remains the standard today, involves rapidly mixing the sclerosant with air to produce a stable foam that can be injected more effectively and safely into veins.
The introduction of ultrasound guidance in the 1980s further revolutionized foam sclerotherapy. Schadeck’s 1985 description of ultrasound-guided sclerotherapy significantly enhanced the precision of foam injection, allowing real-time vein visualization. This development, alongside Cabrera’s 1995 introduction of microfoam, has improved both the safety and effectiveness of the procedure by ensuring more controlled delivery and confirming complete vein closure. Today, ultrasound-guided foam sclerotherapy (UGFS) is recognized as a safe and effective treatment for varicose veins and other forms of chronic venous disease. It is endorsed by leading clinical practice guidelines, including those from the Society for Vascular Surgery and the American Venous Forum and the American Vein and Lymphatic Society. While UGFS offers many advantages, careful patient selection, proper technique, and ongoing monitoring are essential to minimize risks and achieve optimal outcomes.
This chapter will explore ultrasound-guided foam sclerotherapy in detail, focusing on its technique, clinical applications, and important considerations for the treatment of superficial venous insufficiency.
SCLEROSANTS
Mechanism of Action
Foam sclerotherapy involves mixing a sclerosing solution with one or more gases to create foam. The three main categories of sclerosants are detergents, osmotics, and irritants/chemicals. Only detergents may be utilized in foam sclerotherapy as they act as surfactants to lower the surface tension between liquid and gas, enabling the formation of a stable foam. Detergents disrupt the cell membrane through protein denaturation. The result is destruction of endothelial cells, exposure of subendothelial collagen fibers and ultimately occlusion through fibrosis of the vessel lumen.
Types of Detergents
Detergents that may be used to create foam include sodium tetradecyl sulfate (STS), polidocanol (POL), sodium morrhuate, and ethanolamine oleate. STS and POL are the two most commonly utilized sclerosing agents in foam sclerotherapy.
Though equally effective, STS has been demonstrated to be approximately twice as destructive as POL and maintains greater stability at lower concentrations, so it is therefore used at one-half the concentration of POL. Given this difference, the concentrations of the two sclerosants may not be used interchangeably.
The concentration and volume of foam necessary increase as the target vessel diameter increases ( Table 11.1 ). The concentration of sclerosant used in foam sclerotherapy for any particular vessel is approximately 10% to 50% lower than what would be used in the same vessel with a liquid sclerosant. This is due to the increased potency of the foam formulation. A maximum of 10 mL of foam has been suggested per treatment session, according to the European consensus guidelines.
Table 11.1
Maximum recommended Foamed Sclerosant Concentration
| Polidocanol (%) | Sodium Tetradecyl Sulfate (%) | |
|---|---|---|
| Telangiectasias | 0.5 | 0.25 |
| Reticular varicose veins | 0.5 | 0.5 |
| Nonsaphenous tributary veins | 2 | 1 |
| Saphenous veins <4 mm | 1 | 1 |
| Saphenous veins ≥4 mm | 3 | 3 |
| Incompetent perforating veins | 3 | 3 |
| Recurrent varicose veins | 3 | 3 |
| Venous malformation | 3 | 3 |
Varithena (Polidocanol Foam 1%) is the only available commercial microfoam shown to have greater stability than physician-compounded foams in in vitro studies. It contains a 7:1 ratio of gas to liquid, with the gas component composed of oxygen, carbon dioxide, and a small amount of nitrogen. The use of preformulated foam ensures more stable characteristics, such as consistent sclerosant concentrations and small bubbles. While other foam sclerosants return to their liquid state within a matter of 30 to 60 seconds, Varithena retains its foam state for 75 seconds, allowing for more efficient displacement of blood and longer contact time with the vessel wall, ultimately leading to more reliable patient outcomes.
OVERVIEW OF FOAM SCLEROTHERAPY
Indications
Indications for foam sclerotherapy are for both cosmetic and incompetent veins, including those that lead to venous ulcers. Based on size of the vein, they can be divided into direct visualization versus ultrasound-guided placement of foam.
Direct visualization foam sclerotherapy is indicated for the treatment of reticular veins and varicose veins that can be easily seen or palpated. While foam is generally too potent for telangiectasias, it can be useful in treating resistant cases of telangiectatic matting at very low potencies. Liquid sclerotherapy is the preferred treatment for telangiectasias.
Conversely, UGFS is more effective in the treatment of larger veins such as nonsaphenous tributary varicosities, perforating veins, and varicose veins that are difficult to visualize. Ultrasound guidance will also improve efficacy of treatment of smaller vessels in patients who are obese. While other forms of thermal and nonthermal ablation may be more efficacious, saphenous trunks and accessory veins without junctional incompetence are also indicated to be treated with UGFS. Instances in which treatment with foam sclerotherapy is favorable to endovenous thermal ablation include tortuous veins, veins with a diameter too small for endovenous ablation fibers, or those in close proximity to structures that cannot withstand thermal energy such as nerve bundles in the distal GSV and distal SSV. Patients whose comorbidities preclude more invasive treatments may also be more appropriate candidates for UGFS.
Additionally, foam sclerotherapy is effective in the treatment of venous malformations, hemorrhoids, and pelvic varicosities, which requires advanced training in sclerotherapy.
Contraindications
Though UGFS is a safe and generally well-tolerated procedure, there are contraindications for the phlebologist to be aware of ( Table 11.2 ). Absolute contraindications to sclerotherapy include known allergy to the sclerosant, acute deep vein thrombosis and/or pulmonary embolism, local infection at the site of sclerotherapy or severe systemic infection, and patient immobility. Foam sclerotherapy in particular carries a contraindication in patients with known symptomatic right-to-left shunts, such as patent foramen ovale (PFO), which may lead to paradoxical emboli. In patients in whom there is a high clinical suspicion of a right-to-left shunt, it is advisable to perform an ECHO with bubble study for further evaluation.
Table 11.2
Contraindications to Sclerotherapy
| Absolute | Relative |
|---|---|
| Known sclerosant allergy | Pregnancy |
| Acute deep vein thrombosis or pulmonary embolism | Breastfeeding |
| Local infection at sclerotherapy site | Severe peripheral arterial disease |
| Severe systemic infection | Poor general health |
| Immobility | Strong allergic predisposition |
| Known symptomatic right-to-left shunt | High thromboembolic risk |
| Acute superficial venous thrombosis | |
| Neurological disturbances following previous foam sclerotherapy treatment | |
| Fear of needles |
Relative contraindications to all sclerotherapy include pregnancy, breastfeeding, severe peripheral arterial disease, poor general health, strong predisposition to allergies, patients with high thromboembolic risk, and acute superficial venous thrombosis. Foam sclerotherapy in particular carries a relative contraindication in patients with a history of neurological disturbances following previous treatments with foam sclerotherapy.
Comparison to Liquid Sclerotherapy
While liquid and foam sclerotherapy may use the same sclerosant, the use of foam results in longer contact between the sclerosant and vessel wall. This is achieved as the foam creates a “vapor lock” that blocks blood flow and allows prolonged contact between sclerosant and endothelium. The sclerosing effect is therefore maximized by increasing the contact surface area between the sclerosant and vessel wall. This also prevents circulating plasma proteins from reaching and deactivating the drug, resulting in a stable concentration of the sclerosant. This allows for the use of lower concentrations and volumes of a given sclerosant in foam sclerotherapy as compared to liquid sclerotherapy. Ultimately, these properties allow the phlebologist to maintain better control of a stable concentration of sclerosant and duration of its contact with the vessel wall.
Foam sclerotherapy is more user-friendly than liquid, given that the gas is echogenic and can be visualized with ultrasound. The foam will often have a bubbled appearance. This visualization allows for more precise and controlled administration, minimizing the risk of inadvertent intraarterial or extravenous injection. Foam also allows for a higher volume to be created from a given quantity of liquid sclerosant. This property, in addition to decreased dilution with blood, enables the treatment of longer vein segments when using foam.
Studies have demonstrated foam sclerotherapy to be more effective than liquid sclerotherapy in the treatment of varicose veins, with no increase in side effects or complications. The proposed mechanism by which foam is more efficacious than liquid is the prolonged contact that sclerosant maintains with the vessel wall.
TREATMENT TECHNIQUE
Although the technique of ultrasound-guided foam sclerotherapy is nuanced and dependent on the phlebologist’s technical skills, the basic concept is straightforward (
Box 11.1
). Foam is generated in a syringe using a mixture of sclerosant and gas (or use of a prefoamed sclerosant), which is then attached to a needle that is placed into an incompetent vein under ultrasound visualization. The foam is injected into the vein and tracked proximally (Supplemental
![]() Video 11.2
). This process is repeated until all targeted vessels have been treated or until the maximum sclerosant per session is reached. Sessions may be repeated on the same vein every 6–12 weeks, until the vein is successfully sclerosed. The vein is absorbed by the body over time. Treatment success is
dependent on both the phlebologist and patient-compliance with postoperative care.
Video 11.2
). This process is repeated until all targeted vessels have been treated or until the maximum sclerosant per session is reached. Sessions may be repeated on the same vein every 6–12 weeks, until the vein is successfully sclerosed. The vein is absorbed by the body over time. Treatment success is
dependent on both the phlebologist and patient-compliance with postoperative care.
Box 11.1
Treatment Steps for Ultrasound-Guided Foam Sclerotherapy (UGFS)
-
Obtain history and physical examination
-
Perform ultrasound mapping of the legs
-
Acquire written informed consent
-
Set up equipment and position patient
-
Prepare clean surgical field
-
Mark injection sites and perforators
-
Generate foam sclerosant
-
Inject foam sclerosant under real-time ultrasound guidance
-
Compress perforators during and postinjection while patient activates calf muscles
-
Apply dressings to injection sites
-
Don compression stockings
-
Instruct patient to ambulate frequently
-
Assess results with a postoperative ultrasound
Video 11.1 Ultrasound imaging of needle placement into vein with subsequent injection of foam sclerosant, tracked proximally. Courtesy of Sirunya Silapunt, MD, DABVLM, RPhS, and Solmaz Abbasi; Department of Dermatology, University of Texas Medical School at Houston, Houston, TX .
Preparation
UGFS is performed by a phlebologist trained in sclerotherapy. The treatment team may also include an assistant and a vascular sonographer trained in superficial venous disease (Registered Phlebology Sonographer, RPhS).
Patient Selection
Potential candidates for UGFS are screened using a detailed history and clinical examination, as well as a duplex ultrasound to identify venous dilation and reflux in incompetent veins. These steps have been discussed in detail in previous chapters. Indications and contraindications for UGFS are described earlier.
Larger veins that are appropriate for endovenous ablation or vein stripping should be treated prior to pursuing UGFS to decrease the risk of revascularizing the treated veins. Conversely, veins that are more superficial and/or cosmetic should be treated afterwards. Although UGFS has been reported to successfully treat incompetent perforators, this has remained a controversial treatment method given the high risk of complications.
A written informed consent should be obtained from the patient after they are informed of the risks, as discussed later in this chapter, as well as the expected benefits of the procedure. Ultimately, the goal is to redirect blood flow to competent veins by inducing sclerosis of the incompetent veins. While success rates for UGFS vary from 50% to 90%, recurrence at 5 years can also vary widely depending on the treatment approach and vessel size. These results may take many treatment sessions to achieve, and each treatment session’s effect may take up to 3 months to ascertain. Reticular veins and telangiectasias may improve with redirection of blood flow from proximal incompetent veins, although this is not guaranteed given they may not be interconnected.
Equipment
Given that foam sclerosants rapidly liquefy, foam sclerotherapy must be performed swiftly and requires that equipment and supplies are prepared prior to beginning the procedure ( Box 11.2 , Fig. 11.3 ).
Box 11.2
Equipment for Ultrasound-Guided Foam Sclerotherapy (UGFS)
-
Chlorhexidine or 70% isopropyl alcohol
-
Gloves
-
Ultrasound, B-mode and Doppler mode
-
Nitroglycerin paste
-
Syringes, 3-mL or larger
-
Needles, 27- or 30-gauge and ¾″ to 1.5″ length
-
Stopcock, two- or three-way
-
Sclerosant of choice
-
Gauze dressings or cotton balls
-
Compressive bandages
-
Compression stockings, preferably 20–30 mm Hg or higher
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree