Introduction
The object of this chapter is to enable the reader to recognize a variety of clinically observable features that underlie the occurrence of successful or unsuccessful results following sclerotherapy. For the sake of brevity, a number of the strategies and concepts that have evolved and proved useful in the treatment of thousands of patients over a 32-year period will be presented in bullet list, table, or question and answer formats. For many dermatologists, telangiectasias and small reticular veins unassociated with significant venous disease will be present in most of the patients who request treatment. Accordingly, the preponderance of information presented here will deal with small vessel sclerotherapy.
•
Variability
Sclerotherapy, which employs chemical cauterants to scarify and obliterate vascular tissue, is very much an art and not a science. Modalities that deliberately destroy tissue can produce a wide range of responses that are not always easy to predict or precisely control. An enormous number of variables make it difficult to create uniform treatment protocols including: (1) the inability to standardize venous tissue or treatment techniques; (2) unpredictable variation in sensitivity to sclerosants not only from patient-to-patient, but between different vessels of the same size on the same patient; and (3) the influence of multiple, clinically distinguishable but largely uncharacterized venous subtypes, anatomical location, and circulatory connections between visible veins and the deeper venous circulation. Unexplained innate variability and lack of understanding about the multiple factors that affect it, has provoked a great deal of controversy regarding the ‘ideal’ way to treat unwanted veins. Personal experience, confirmed by review of tens of thousands of before and after photographs, suggests that:
- •
Of all the identifiable characteristics that affect treatment outcomes, vessel size is often the best (but not the only) prognostic factor of treatment outcomes and the occurrence of common complications ( Fig. 6.1 ).
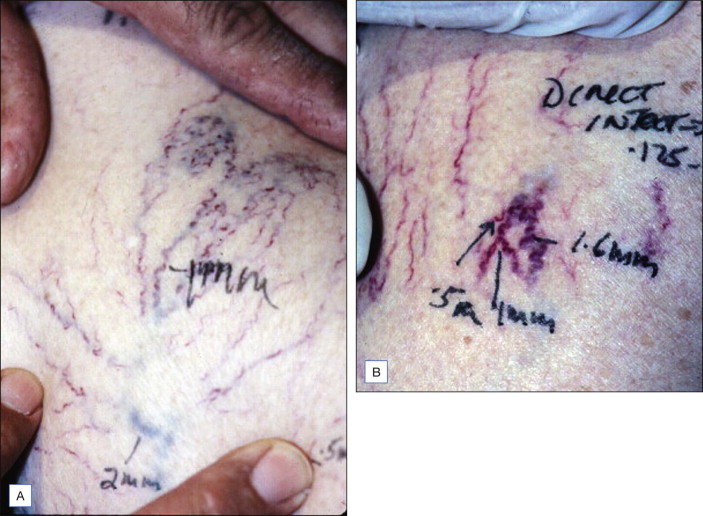
Figure 6.1
Measurements of vessel size reveal typical variability associated with specific vessel sizes
- •
There is no cure for varicose or spider veins, and good or bad results can occur using any treatment protocol.
- •
What happens in the short term often changes in the long run.
- •
No single treatment ‘recipe’ will consistently produce optimum results.
- •
Treatment of telangiectasias 0.5 mm in diameter and smaller will often provide the greatest range of outcomes and patterns of response, while vessels larger than this size usually respond to treatment much more predictably.
- •
Good results can be obtained by treating telangiectasia directly without treating reticular veins.
- •
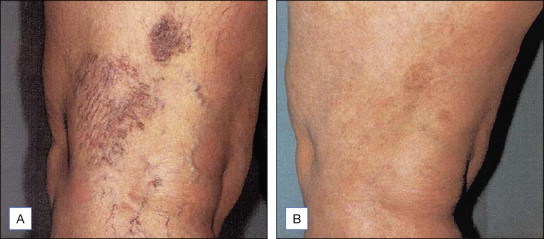
Reticular veins are sometimes more fragile than associated telangiectasia and can be destroyed with pigmentation/matting, sometimes leaving telangiectasia unaffected ( Fig. 6.2 ).
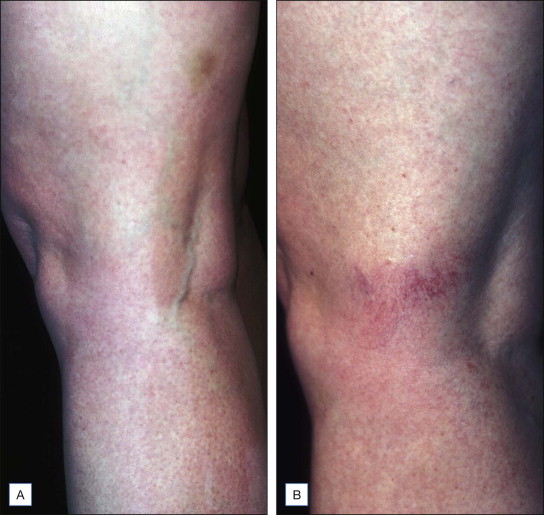
Figure 6.2
( A ) This reticular vein measuring 1.3 mm in diameter was cosmetically unattractive to this patient. ( B ) A post-treatment photograph reveals neovascularization (matting) which was even more unattractive to the patient. Although almost nothing has been published regarding complications following injection of reticular veins, the author has found matting to be a relatively common occurrence in this area
- •
Adequately treated telangiectasia rarely recur, photographic analysis reveals that ‘most recurrences are in fact new vessels’.
•
Divide and conquer
From the standpoint of treatment strategies and expected outcomes it is useful to divide sclerotherapy into three broad categories. (1) Large-vessel sclerotherapy, which treats refluxing axial varicosities (saphenofemoral and saphenopopliteal junctions), nonsaphenous truncal varicosities, perforators and large (>3 mm in diameter) reticular veins. (2) Small vessel sclerotherapy which deals with telangiectasia and reticular veins <3 mm in size. (3) Sclerotherapy which deals with non-lower extremity veins i.e. veins located in other anatomical sites, AV malformations, etcetera. Although sclerotherapy has generally been considered to be the treatment of choice for small lower extremity vessels, it has long been regarded as a poor second to surgical intervention for the treatment of junctional reflux and large varicose veins. The use of foamed sclerosants under duplex guidance and imaging techniques which are facilitated by the use of foams may produce results which compare favorably with surgery.
Advances in Large Vessel Sclerotherapy
•
Foamed / detergent sclerosants
First described over 70 years ago, detergent foams are generally considered to be three to four times more potent than equivalent concentrations and volumes of liquid sclerosants. Increased potency is related to several factors, including an increase in the effective surface area of foams compared with liquid, and the displacement of blood from the treated vein, which produces prolonged undiluted intimal contact.
Foams also produce increased vasospasm and sclerosis of veins at a greater distance from the injection site than liquid preparations, and may be used in much smaller volumes and concentrations (with presumably less risk of tissue necrosis or allergies). Foams are ‘dramatically more visible’ than liquids on duplex imaging, facilitating needle placement and the ability to monitor sclerosant flow in real time, they have also proved useful in a wide range of other applications. Wollmann (2004) has exhaustively detailed both the history and enormous number of variables (e.g., bubble size, uniformity, temperature, sclerosant/gas – room air versus CO 2 , type of sclerosant, etc.) that impact the clinical effects of foam preparations. Although foam sclerotherapy has the potential to revolutionize the treatment of large refluxing veins, it is at this time very much in the early phase of integration into common use.
For large vessels associated with significant reflux, some combination of foam sclerotherapy, surgical intervention, and endovenous laser or radiofrequency devices may be employed synergistically by individuals experienced in their use. Good results using any modality often depend not so much upon the specific treatment employed, but on the expertise of the practitioner.
Foam Disadvantages
Foam takes time to prepare, deteriorates quickly at room temperature and is difficult to aspirate. Foam preparations are generally not suitable for the treatment of telangiectasia and small reticular veins and are associated in that setting with an increased tissue necrosis, pigmentation and neovascularization (matting); phlebologists who believe that refluxing reticular veins are the ‘cause’ of telangiectasia sometimes combine dilute foamed sclerosants to treat ‘feeding’ reticular veins while using liquid sclerosants to treat telangiectasia. No commercially produced standardized foam preparations are available; extemporaneous preparations of foam are hard to duplicate making it difficult to establish uniform treatment protocols which compare the efficacy of different types of foam in specific applications. In addition, the US FDA has not scrutinized, let alone approved, any type of foam for any purpose. When treating patients with asymptomatic patent foramen ovale (that occurs in around 25% of the population) foam may lodge in the cerebral circulation resulting in temporary ischemia associated with migrainoid visual disturbances, amaurosis, and strokes. The substitution of CO 2 for room air (which provides better control over bubble size and endothelial adhesion) may decrease the risk of embolization. Complications associated with extremely potent sclerosants should also be expected to increase following foam sclerotherapy. These include an increased risk of uncontrolled thrombosis and destruction of vascular tissue outside targeted veins.
Duplex Problems
Although real-time duplex imaging is an invaluable aid to placing needles in the right position, it is by no means perfect. Duplex scans, after all, are a two-dimensional view of a three-dimensional process. In at least one case, a lawsuit has been filed (unpublished personal communication) when severe tissue necrosis necessitating in the amputation of a lower limb occurred despite the use of duplex guidance during the administration of foam. The concentration of foam in this case may have been much greater than recommended by most authorities.
Personal Experience with Foam
Over the last several years the author has treated approximately 600 patients presenting with a wide range of vessel types using one part polidocanol (POL) to three or four parts room air prepared using the double-syringe method. The author has found foam preparations to be particularly effective for refluxing vessels 4–5 mm in diameter but unnecessarily cumbersome and no more effective than liquid sclerosants for smaller vessels, particularly telangiectasia. Viscous foams are difficult to inject through a number 30 needle. The use of foam also necessitates certain technique changes aimed at keeping the foam in contact only with the treated areas. This includes post treatment elevation of the treated extremities, and a several minute delay following treatment before applying compression. These techniques may minimize displacement of foam into areas where unintended thrombosis can occur. Spot compression using tape dressings and padding must also be applied after a delay to avoid displacement of residual foam. When I first started using foam a tenfold increase in the occurrence of superficial thrombophlebitis was observed following the use of foam compared with liquids. Ordinarily, a patient only requires one thrombectomy to evacuate thrombi in treated vessels. However, following the use of foam preparations, recurrent thrombi and thrombophlebitis occurring up to one to two months after treatment necessitated multiple thrombectomies. The use of lower volumes and sclerosant concentration and the application of class II compression hosiery has essentially remedied these problems.
•
Small-vessel versus large-vessel sclerotherapy
Several important differences separate the treatment of small vessels from symptomatic large refluxing varicose veins. Varicose veins are treated as a medical necessity, and appropriately billed for insurance compensation. Lower extremity small-vessel sclerotherapy and sclerotherapy for vessels located in other areas is often, but not always, an elective cosmetic procedure (although lower extremity telangiectasia and small reticular veins are occasionally symptomatic). Patients need to understand that insurance companies should not be expected to pay for cosmetic procedures. They should also be counseled regarding the need for multiple treatments, and the possibility of minor and sometimes major complications. Individual risk factors for common complications should be explained. Patients who seek treatment for cosmetic problems sometimes express unrealistic expectations which must be addressed by very careful oral and written instructions, photographs, and consent forms.
Advances and Alternatives for the Treatment of Small Vessels
Advances in small-vessel sclerotherapy are evolutionary, not revolutionary. Although some authorities have proposed specific guidelines, there are very few absolutes.
Controversies regarding small-vessel sclerotherapy involve the importance of venous reflux and venous hypertension, the need (or lack thereof) to treat large vessels before small, and the importance of compression when treating telangiectasia. Current orthodoxy designates refluxing reticular veins as a prime etiologic factor for the development of telangiectasia; although Green (1998) and others have presented credible arguments which challenge this concept. Transdermal lasers which need to traverse the skin, work very well for facial telangiectasia. For the treatment of lower extremity veins, although heavily promoted, lasers are expensive, painful, time consuming, and decidedly inferior alternatives to sclerotherapy as noted ( Figs 6.3 , 6.4 ). Their use should be reserved for patients who are allergic to sclerosants, are needle phobic, or have veins too small for inexperienced phlebologists to cannulate. Lasers have also been advocated for the treatment of telangiectasia that have proved unresponsive to previous sclerotherapy (matting) or to treat patients ‘prone’ to matting. Unfortunately lasers are often unsuccessful in all these settings. In contrast, endovenous radiofrequency and laser devices are becoming more popular for the treatment of refluxing axial varicosities and certain types of reticular veins. Some of this popularity may be due to favorable insurance reimbursement policies which promote the use of these modalities.
Classifying Veins, New Paradigms
In 1988, the author formulated a classification system which included approximate vessel size, anatomical features, color, and relationship to the saphenous system. This classification in an expanded more sophisticated form has resurfaced in later publications. Although this earlier scheme has proved useful for categorizing and optimizing treatment of specific types of veins it neither factors in the existence of a large number of other variables which affect treatment outcomes, nor acknowledges the relationship of vessel size to intrinsic patterns of long and short-term responses to sclerotherapy. In addition, it fails to emphasize the clinical importance of a specific class of extremely resistant telangiectasia (usually 0.2 mm and smaller), which develop after previous sclerotherapy treatments. This phenomenon may represent persistent long-lasting vascular remodeling initiated by the trauma of sclerotherapy in patients with poor control over vessel growth. Vessels of this type are a source of great frustration for patients.
•
Basic patterns of response
Although a large number of interactive variables affect treatment outcomes, three fundamental patterns of response can be predicted largely on the basis of vessel size. Since almost all patients present themselves with veins of various calibers a mixture of these patterns is commonplace.
- 1
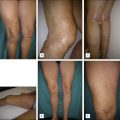
Gradual destruction (telangiectatic pattern) in which small telangiectasia fade and fragment, a process not usually not associated with pigment or palpable thrombi occurring following multiple treatments over several months (small vessel pattern; Fig. 6.5 ). Extremely dilute sclerosants can sometimes produce these effects more slowly with fewer complications (and more treatment failures).
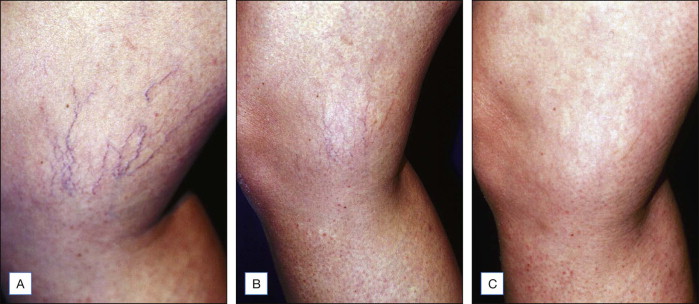
Figure 6.5
( A ) Pretreatment appearance of vessels involving the inner knee, varying in size from 0.1 mm to approximately 0.5 mm in diameter. ( B ) This photograph taken 3 months after two treatments reveals complete disappearance of the large vessels with substantial fading in the smaller ones, a typical pattern. ( C ) This photograph taken 10 months after the second treatment reveals almost complete resolution. The inner knees are an area subject to matting and should be treated cautiously with low concentration of sclerosants
- 2
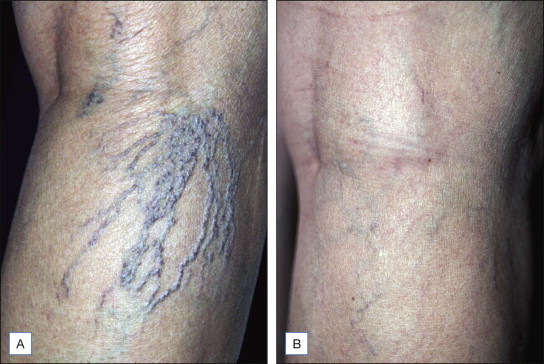
Rapid destruction (large vessel pattern) are typically associated with pigment and palpable thrombi, this pattern can occur within hours after treatment ( Fig. 6.6 ). In contrast to the treatment of small telangiectasia which are relatively insensitive to changes in sclerosant concentrations, outcomes obtained following treatment of large telangiectasia, reticular veins, and varicose veins, are sensitive to changes in sclerosant type, concentration and the use of compression.
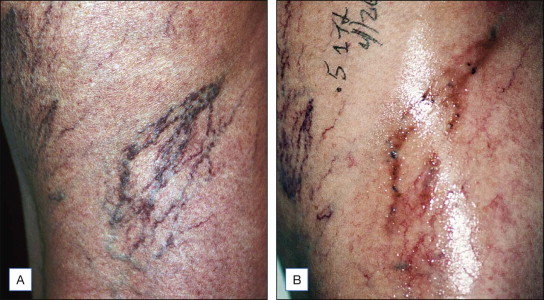
Figure 6.6
( A ) These pretreatment photographs reveal fragile, thin-walled, elevated tortuous vessels 0.6–1.0 mm in diameter. ( B ) This photo demonstrates pigmentation, which can occur following low sclerosant concentrations when treating vessels of this type
- 3
Resistance (two types). Although veins of any size can fail to respond to treatment there is a clear difference between dilutional resistance that occurs following treatment of larger vessels and microtelangiectatic resistance that occurs in very small vessels. Dilutional resistance can often be overcome using more concentrated sclerosants, foams, or more vigorous compression. In contrast, resistance occurring in very small telangiectasia (0.1–0.2 mm in diameter) is usually unaffected or worsened by increasing sclerosant concentrations. This type of resistance is poorly understood. It has been attributed to venous hypertension, failure to treat reticular veins, or to employ compression. The authors experience suggests that this type of resistance is more complex and may be unassociated with any of the ‘predisposing’ conditions. A more detailed description of this type of resistance will be presented later in this chapter.
•
Vessel size: The new golden rules
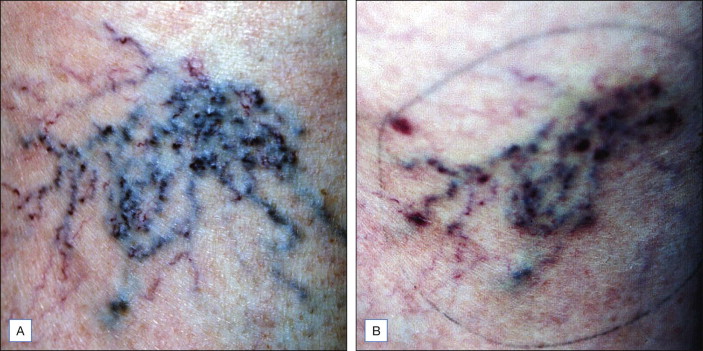
Vessel size is by far the most meaningful prognosticator for treatment outcomes although many other variables affect the process. This is not to say that veins will always respond stereotypically on the basis of vessel size alone. The concentration of sclerosant necessary to destroy veins varies a great deal from person-to-person, while vessels of the same size will sometimes display similar patterns of response despite wide variations in sclerosant concentrations ( Figs 6.7 , 6.8 ). Given this degree of variability it is prudent to consider typical patterns rather than make precise predictions. When treating telangiectasia 0.5 mm and smaller, miniscule (0.1 mm) changes in vessel size can produce fundamental differences in both treatment response patterns and the occurrence of common complications. Although sclerosant concentrations and other factors can alter treatment responses, careful measurement of vessel size will often accurately predict:
- •
Whether vessels will respond gradually to repeated treatments (usually without associated pigment or thrombi).
- •
Whether vessels respond rapidly (often associated with thrombi and pigmentation).
- •
Be resistant to treatment.
- •
The number of treatments necessary.
- •
How long vessels have been present.
- •
Complications (pigment, thrombi, resistance).
- •
Likelihood of underlying reflux/venous hypertension.
- •
Optimal sclerosant concentrations.
- •
Sensitivity to changes in sclerosant concentration.
- •
The need for compression.
•
A word about the literature and vessel size
Definitions
By convention, tortuous, dilated veins 4 mm and larger in diameter are defined as varicose veins. The term reticular vein designates veins between 1–3 mm in size. Telangiectasia are defined as veins between 0.1–1 mm in diameter, although the terms capillaries, telangiectasia, and venulectasia are used interchangeably.
Telangiectasia
Phlebology literature at large views all vessels between 0.1–1 mm in diameter as a homogenous entity. One author notes that the only difference between varicose and telangiectatic leg veins is size. In truth, telangiectasia constitutes a extremely heterogeneous group of vessels both from the standpoint of etiology and response to treatment. They do not always respond to treatment like tiny varicose veins. There are at least seven categories of lower extremity telangiectasia between 0.1 mm and 0.9 mm in diameter. Each exhibits a more or less typical pattern of response and sensitivity to sclerosants as well as distinct differences involving a host of other variables.
When practitioners attribute optimal results (particularly the number of treatments to eradicate telangiectasia and/or occurrence of pigmentation or thrombosis) to some technique strategy, their observations have limited utility unless the specific size of the telangiectasia is duly noted in 0.1 mm increments. Telangiectasia (≤0.3 mm in diameter) routinely require several treatments for eradication. The use of higher concentrations will often not produce more rapid results, pigmentation, or palpable thrombi. Concentrated sclerosants will, however, routinely produce more neovascularization (matting). Conversely, telangiectasia ≥0.4 mm in diameter will often respond more quickly with more pigmentation and thrombi when higher concentrations of sclerosants are employed.
•
Categorizing vessels by size / effect of previous treatments upon microtelangiectasia
There are two types of very tiny telangiectasia which although they look alike respond quite differently to treatment.
Responsive Microtelangiectasia
- •
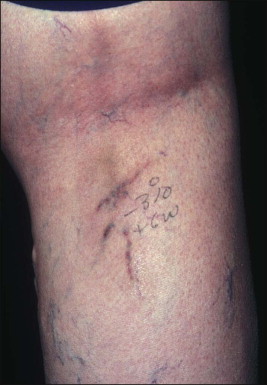
0.1–0.2 mm in diameter: Previously untreated (virgin) veins in this size range can usually be effectively treated. They often require at least three treatments to induce a slow process of fading, which may involve the body’s own genetic machinery to destroy partially damaged cells in the vessel walls (apoptosis). The use of higher concentrations often will not produce faster results and may lead to more matting. This type of vessel often occurs in the absence of reflux. Compression and the injection of reticular vein does not appear to be of any value when treating this type of vessel. Treatment is usually carried out at 4–6-week intervals. Resistance is extremely uncommon in previously untreated vessels measuring <0.2 mm in diameter. Pigmentation is extremely rare for all vessels in this size range ( Fig. 6.5 ).
Resistant Telangiectasia (Matting/Second-Generation Vessels)
- •
0.1–0.2 mm in diameter: Vessels in this size range which remain or occur after previous treatments, are often refractory to treatment within a given time frame, and are the number one cause of dissatisfaction following small-vessel sclerotherapy ( Fig. 6.9 ). This type of vasculature is most common on the inner and outer thighs within 25 cm of the knees. The author’s experience suggests that vessels of this type are best treated by tincture of time, being treated no more than two to three times yearly. With sufficient time these vessels may resolve spontaneously or become responsive to sclerotherapy and other modalities. Once again, compression and injection of reticular veins or elimination of reflux do not appear to result in predictable benefits.
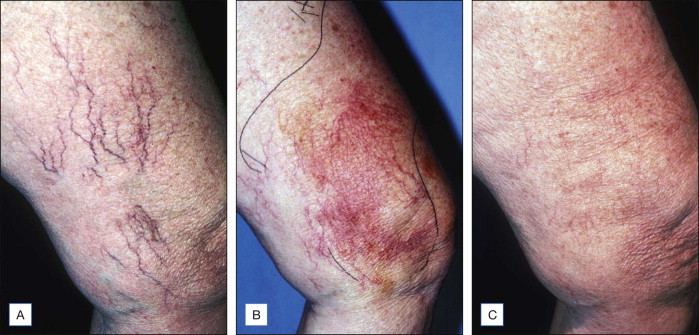
Figure 6.9
( A ) Pretreatment photographs reveal vessels varying in size from 0.2–0.8 mm in diameter. ( B ) This photograph, taken approximately 1 year later reveal extensive neovascularization resistant to several treatments carried out over a 6-month period. ( C ) 1 year later these neovascular vessels had resolved spontaneously. Deferred treatments are often the best way to approach neovascularization (matting)
- •
0.3–0.5 mm in diameter: Good news begins at 0.3 mm. These vessels usually require several treatments and fade slowly over several months without pigmentation or thrombi. The usual treatment interval for vessels in this size range is every 4–6 weeks, whether or not the patient has been treated before. As vessels become fractionally larger, a second (large-vessel pattern) supervenes. At 0.4 mm and 0.5 mm in diameter, pattern overlap occurs. Vessels of this size can undergo the small-vessel fading pattern or be destroyed rapidly, a process sometimes associated with pigmentation and thrombosis. About 25% of vessels measuring 0.4 mm and about 50% of those measuring 0.5 mm will respond rapidly. Changes in concentration may affect rapidity of this process. Higher sclerosant concentrations are routinely associated with more rapid results associated with increased thrombosis and pigmentation ( Figs 6.8 , 6.10 ). In some cases, injection of reticular veins that are in direct communication with clusters of larger telangiectasia produce dilutional effects that may reduce hyperpigmentation in fragile telangiectasia. This technique also permits fewer needle sticks. In other patients, injecting the reticular veins does not appear to modify outcomes one way or another. Although experts ‘feel’ that sclerosing reticular (feeder) veins is a uniformly good way to treat telangiectasia. No one has ever carried out long-term studies to determine if reticular veins have been sclerosed and if so, how long.
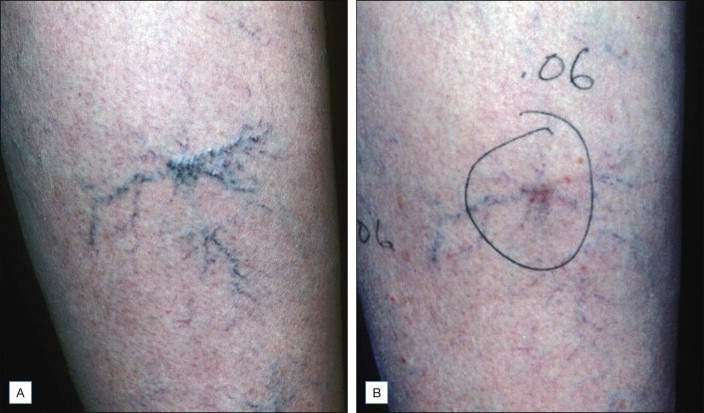
Figure 6.10
( A ) Pretreatment photographs, vessels 0.5–0.8 mm in diameter elevated and tortuous. ( B ) This post-treatment photograph taken 3 months after two treatments using 0.06% polidocanol produced a more gradual destruction with less thrombosis than a single injection of 0.5%, an eightfold difference in concentration.
- •
0.6–0.9 mm in diameter: Vessels in this size range are often purple or blue–green in color. They can be extremely fragile and often have been present for many years ( Fig. 6.6 ). Clusters of elevated and tortuous vessels of this size suggest longstanding venous hypertension or reflux. Their presence should prompt a thorough evaluation. These vessels usually require only one treatment. It may also be beneficial to employ more dilute sclerosants and/or compression to minimize pigmentation and clotting that routinely follows treatment. A constellation of bruising, thrombi, and pigmentation often makes treated veins of this type look worse before they look better. Patients may be reassured using postoperative photographs of patients with similar veins.
- •
1–5 mm in diameter: Large greenish-blue vessels (i.e., the color of the veins on the backs of hands) are usually destroyed in one or two treatments, a process that is often followed by clotting and pigmentation. Vascular fragility is a highly individual matter. Certain individuals will experience rapid destruction of vessels not associated with thrombosis or pigmentation using a wide range of sclerosant concentrations ( Figs 6.11 , 6.12 ). Compression dressing and hosiery are used to control thrombus size and hopefully reduce hyperpigmentation. All patients with vessels larger than 0.6 mm in diameter and less than 3 mm in size can be expected to respond quickly and accordingly can be treated more often, at 1–2-week intervals.
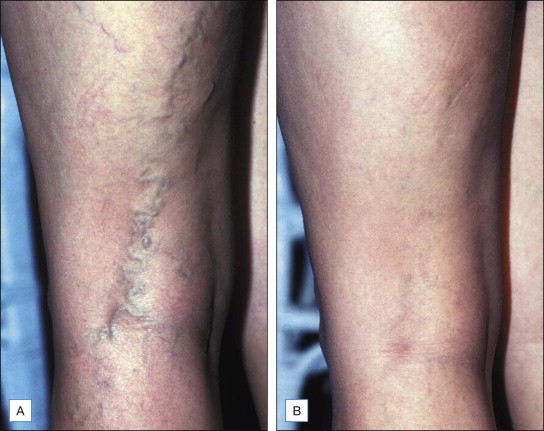
Figure 6.11
Although pigmentation is common following the treatment of large vessels, these two photos reveal complete destruction of a vessel in the 3 mm in diameter range without pigmentation. Variability such as this makes it difficult to determine the true impact of any treatment strategy
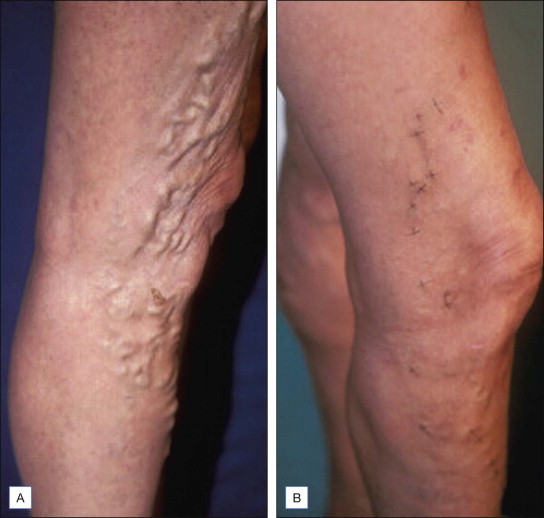
Figure 6.12
( A ) This pretreatment photograph demonstrates varicose veins 4–5 mm in diameter. ( B ) 1 week after treatment, almost complete resolution of these veins has occurred associated with minimal pigmentation. These results are a function of intrinsic variability and not particular strategies
Varicose Veins (4mm and Larger)
When they occur in specific anatomical locations (great and short saphenous vein), or they are tortuous size really is important and the larger they are the better the chance there is some degree of reflux or venous hypertension. Rapid results or a complete resistance are the rule with vessels of this size. Recurrences and treatment failures as a function of treatment methods have provoked a great deal of controversy.
Measuring Veins: Mechanics
Perhaps the handiest tool for the phlebologist who treats telangiectasia is the number 30 needle, which at 0.3 mm in diameter is exactly the size above which resistance rarely occurs when treating telangiectasias, and below which resistance is relatively common. A number 25 needle, at 0.5 mm in diameter, is the size of the vessels that are the bridge between vessels which fade slowly following multiple treatments, and larger vessels that are destroyed suddenly with pigmentation. The ability to predict the occurrence of pigmentation and reassure patients about its transient nature and the unimportance of small thrombi (we have euphemistically described them as ‘trapped blood’). This allays patient concerns about ‘clots breaking loose’. Patients are told to expect palpable thrombi when treating certain types of veins they are also told to regard the presence of these thrombi as an excellent measure of the completeness of vein destruction. They are also told that recurrences are rare but new telangiectasia may appear over time which will require maintenance ‘touch ups’.
•
Pitfalls in measuring vessels
All measurements are best carried out with the patient recumbent using minimum tension to avoid effacement of very small vessels or increases in vessel diameter produced by tension. A generous dab of alcohol renders the skin more transparent, permitting better vessel visibility. A number of devices have been devised that use light to trans-illuminate deeper vessels, but these are generally not necessary.
•
Measuring devices
Biersdorf-Jobst Inc. Charlotte, NC has fabricated an extremely useful clear plastic ruler ( Fig. 6.13 ). Bioform Medical Inc. (San Mateo, CA) is currently developing a measuring device of a more convenient size along with instructions on how to employ it.

For measuring very small vessels precisely, some form of magnification will be needed, which also make it easier to cannulate telangiectasia which are often smaller than the needle employed. The author prefers Optic-Aids ((Mattingly International Inc.); available at optometry or specialty shops) ( Fig. 6.14 ) at about 5×, but other alternatives are feasible, depending on operator preference. Loupes, some with polarizers, and a variety of expensive and more cumbersome devices are also available.

Sclerosants
‘The perfect sclerosant’, complication-free and 100% effective has not been discovered, nor is there a gold standard for accurately comparing different types of sclerosants. Although complications such as matting and hyperpigmentation have been attributed to the type of sclerosant employed, the author’s experience suggests that sclerosant concentration and individual variability, exert the most important influences. Allergies and tissue necrosis are related to both sclerosant type and concentration.
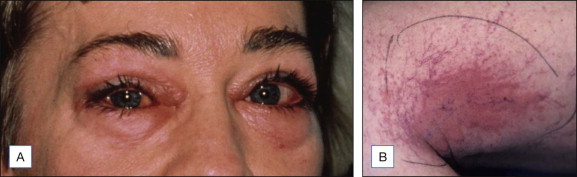
Sclerosants are categorized by their primary mechanism of action: detergent, osmotic (dehydrating), and chemical. There is no gold standard for comparing sclerosants. Although there is a great deal of controversy regarding relative efficacy and potential of various agents to cause specific complications there is general agreement that the use of more potent sclerosants is associated with an increased efficacy and a concomitant increase of adverse events. Sclerosants destroy veins by inducing thrombosis and fibrosis. Contraindications to the use of all types of sclerosants include hypercoagulable states, inability to ambulate, and significant peripheral vascular disease. Patients with serious allergies should be excluded from treatment using detergent sclerosants. Allergies are a great rarity following the use of modern sclerosants. Figure 6.15 documents clinical findings when serious allergies occur. Sclerosant choices are often made on the basis of previous experience and the biases which develop from it. Most importantly, when variables affecting treatment outcomes are understood, and accurate comparisons of relative sclerosant potency are established, there will be fewer arguments regarding the putative benefits or drawback of specific types of sclerosants. Tables 6.1 and 6.2 present a brief comparison of sclerosants.
| Sclerosant | Clinical application | Dosage | Advantages | Disadvantages | Complications | Animal studies |
|---|---|---|---|---|---|---|
| Hypertonic saline (US) Legal “off-label” use | Abortifacient, spider, small varicose veins (10–30%) | Individual; restrict volume in patients with salt-restricted diets | Absolute absence of allergies | Painful, weak sclerosant high risk of tissue necrosis | PE, deaths not reported CTN in concentrations >5%, NCTN | LD50 In mice NA |
| Sodium tetradecyl sulfate (Sotradechol, Bioniche Pharma Group) US/worldwide FDA approved 1946 | Varicose and spider veins 1–3%. Other concentrations “off-label” | Maximum single dose tx in one day not to exceed 10 mL of 3% (varies with publication) | Good empirical record; can be used in large volumes; can be foamed | Painful on extravasation; concentration-dependent tissue necrosis; contraindicated in polyallergic patients | PE, reported anaphylaxis .003%, DVT, fatalities rare can occur with low concentrations and volumes; CTN ≥1% concentrations, NCTN | LD50 in mice 90 ±5 mg/kg |
| Polidocanol (Aethoxysklerol, Merz Bioform) USA/Asclera™ FDA approved 2010 For reticular/spider veins only | Varicose and spider veins 0.5% and 1%; other concentrations “off-label” | Not to exceed 2 mg/kg (10 mL 1% for 50 kg patients) | Painless, allergies rare (0.1–0.3%); can be foamed. Incidence of NCTN .0001%. No CTN at 3% | Transient urticaria and pruritus; 1/3 potency of STS; contraindicated in polyallergic patients; contraindicated for patients with peripheral arterial disease. | PE, reported anaphylaxis .003%, lowest rate of fatalities of any detergent sclerosant extremely rare; cardiotoxicity | LD50 in mice 118–145 mg/kg varies with publication |
| Three forms of glycerin; Pure, Chromated (Scleremo™) and with epinephrine and xylocaine | Pure glycerin for neurologic applications; chromate and with additives for spider veins | Not to exceed 10 mL of pure glycerin; severe reactions never reported using low doses | Mild sclerosant; allergies rare; useful for fragile small vessels; no CTN, rare NCTN, pigment rare. | Painful, viscous, treatment failures. Chromated glycerin illegal to use. Pure glycerin ± additives “off-label” | Chromated glycerin 1 death; intravascular hemolysis, hemoglobinuria, renal damage, hyperglycemia, hyperosmolality | LD50 in mice NA |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree