Introduction
The prevalence of venous disease makes it an everyday problem in clinical practice. In recent years, a surge in treatment options available has renewed interest in venous diseases. Phlebology, the study and treatment of venous disease, was recognized in the US as a self-designated medical specialty in 2005 by the American Medical Association, creating a venue for this disease complex. Novel minimally invasive treatment options for superficial chronic venous insufficiency include endovenous thermal ablation, endovenous chemical ablation, ambulatory phlebectomy, and ultrasound-guided foam sclerotherapy. Compelling successful outcomes, swift recovery, and low complication rates have ignited interest in using less invasive techniques in lieu of the traditional open surgical methods. Applied new understanding has established superficial chronic venous insufficiency as a medical problem/disease and not merely a cosmetic concern.
We must credit much of our new comprehension of venous disease directly to the development and refinement of duplex ultrasound instrumentation. As the equipment expands into digital sophistication, technical knowledge and experience must be applied proportionally to obtain accurate diagnostic results. Duplex ultrasound has become the gold standard for the evaluation of chronic venous insufficiency of the lower extremities. Venography of the lower extremities has largely been abandoned for less invasive techniques. Historically, venous duplex studies of the lower extremities were aimed at the diagnosis of deep vein thrombosis. The genesis of venous ‘mapping’ expanded the scope of ultrasound to examine the superficial venous system and study venous valvular hemodynamics. The expansion of minimally invasive therapies has renewed interest in venous hemodynamics as it relates to procedural and surgical intervention.
Ultrasound visualization during procedural interventions has provided numerous options for minimally invasive treatments. Accordingly, ultrasound training expertise must be expanded to include the applied skills for pre-, peri- and post-phlebologic treatments.
This chapter will provide a basic explanation of the use of an ultrasound device and its application to diagnostic and therapeutic use in phlebology ultrasound.
Ultrasound Instrumentation
Packaging design utilized by ultrasound manufacturers to promote ease of use, affordability and portability has consequently downplayed the underlying complexity of the ultrasound device. Ultrasound is both operator based and operator dependent, and is completely reliant on the individual user’s experience and skill to obtain accurate results. Additionally, the overall engineering design characteristics of the equipment will affect the results that can be obtained. Advanced duplex systems, regardless of size or specific use, require a thorough understanding of Doppler physics and the scientific principles applied to ultrasound within the human body. Diagnostic quality from ultrasound is obtained by users who have applied diligent study and practice. In addition, diagnostic results are equally dependant on the caliber of the instrument from which the study was obtained.
Much of the focus of this chapter will pertain to the use of ultrasound as a tool to gain information of the lower extremity venous system and its relation to structure (anatomy) and physiology (flow). Within a relatively short time span, ultrasound technology progressed from static images to today’s real-time imaging. Crude hand-held Doppler listening devices have also evolved. Revolutionary breakthroughs occurred when engineers discovered how to integrate the two technologies into one device; hence the term duplex was coined. Doppler physics combined with imaging created simultaneous information for the user, and is particularly useful in the application to blood vessel diagnosis. Further technology expanded with the introduction of color flow Doppler. An explanation of the ultrasound physics device is beyond the scope of this chapter. The author recommends further reading and physics study by noted authors Frank Miele and Sidney Edelman. A basic understanding of the scientific theories applied to an ultrasound scanner will provide a basis for obtaining best ease of use.
With the use of an acoustic medium (gel) and a transducer to touch the skin, sound waves are transmitted. Brightness of the returning echo forms the basis for B-mode, gray scale imaging. Such images (pixels) are displayed on a monitor and are subject to manipulation of the sound waves through soft tissue.
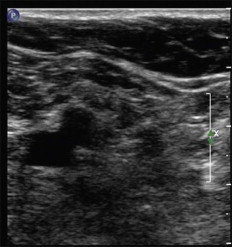
Color flow indicates presence, direction and relative magnitude of movement. In particular, when applied to the movement of blood or blood velocity, flow is displayed as a color pixel. The concept of movement detection, however, must be kept in the mind of the ultrasound user since transducer movement or vessel movement may be interpreted erroneously as blood flow. The display of the upper and lower segments of the color bar ( Fig. 4.1A ) serve as a reference to indicate flow direction, or movement, either toward or away from the transducer. Longitudinal images of vessels in axial positions commonly depict arteries in red (flow away from the heart) and veins in blue (flow towards the heart) ( Fig. 4.1A ). With this setting, red pixels inside the vein represent reverse flow. Color images can be displayed in longitudinal or transverse images. The transverse, oblique image facilitates (1) simultaneous visualization of multiple veins, and (2) assessment of diameter measurements. The standard documentation of reflux has been accepted to be displayed in a longitudinal format combined with Doppler spectral tracings.
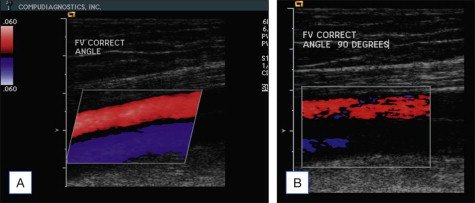
Color flow sensitivity is dependent on flow physics and is controlled by instrument settings. Velocity, blood volume and direction affect detection of color flow. Difficulties in detecting low velocity are well recognized. Flow directly toward or away from the transducer, representing an angle of insonation equal to zero, produces the highest sensitivity. An angle of insonation of approximately 60° is commonly employed to longitudinally image vessels parallel to the skin ( Fig. 4.1A ). Flow parallel to the transducer, representing an angle of insonation of 90°, results in lowest sensitivity ( Fig. 4.1B ). The three-dimensional images are displayed on a viewing monitor in a two-dimensional formatted image, which visually can create confusion for the untrained user.
In addition to the user’s angle of insonation, there are other settings, or ‘knobology’, that will impact flow sensitivity. Velocity scale (a preferred nomenclature to PRF, or pulse repetition frequency, a non-physiological term) and color flow gain must be counterbalanced to provide increased sensitivity without resulting in what is termed a ‘noisy’ image. A gain setting high enough to avoid artifacts onto the gray-scale image is recommended. The velocity scale must be adjusted for each patient’s unique flow conditions, though there are anticipated normal flow velocities common in patients. Another important control is persistence, or more specifically, the length of time the color flow persists on the display. Electronically, the flow signal detected in one time frame can persist on display for many subsequent frames. Therefore, fast signals, such as the ones created by valve closure, may persist on screen, giving the illusion of color flow flashes or wisps of bidirectional flow. Color flow findings observed should be confirmed with spectral display.
The duplex technology matches flow velocity detection to a specific anatomic region depicted in B-mode and color flow imaging. A graphic display of the velocities within a blood vessel is created by the instrument as a spectral waveform. Velocity values are calculated based on the Doppler frequency shift caused by the flowing blood, the frequency of the transducer, the angle of insonation, and the velocity of ultrasound through the tissue. A Doppler frequency spectrum is created within the instrument onto a graphic display of blood flow velocities within the sample volume at each instant of time. The sample volume control determines the region of flow detection within the vessel that will be sampled. Appropriate sample volume size selection is critical. Too large of a sample volume will increase artifact within the corresponding spectral tracing due to vessel wall or hand motion by the sonologist. A small sample volume produces a crisp waveform tracing without the noise caused by spectral broadening. This spectral broadening represents the detection of a wide range of velocity values at the same time, and has more general applications to arterial imaging. A larger sample volume within the vein is recommended to detect flow with a decentralized velocity profile. As is the case for color flow, Doppler gain settings and velocity scale affect the sensitivity to flow, or reflux detection. Increased gain and decreased velocity scale improve detection of low velocity, low volume reflux. The gain adjustment will be increased or decreased to provide bright flow velocity waveforms just below the level that causes a noisy speckle on the display.
The ultrasound imaging transducer, or probe, one selects will decide the depth range that can be examined. Higher frequency transducers, i.e. ranging 10.0–17.0 MHz, are designed to look in the near field for structures such as the superficial venous anatomy and/or facilitate needle access or guidance. Lower frequency transducers, i.e. ranging 4.5–7.5 MHz, permit visualization at lower depths and are selected when evaluating the deep veins. In most instances, when performing a diagnostic study you will need both a low and a high frequency transducer. For extremity venous applications, linear transducers are ideal.
Ultrasound equipment is designed for a variety of applications, thus manufacturers build in features to create simple steps to assist users. One helpful feature is a pre-set function of preferred settings for examination types, very similar to a short-cut button on a personal computer. Using these pre-sets as a starting point to begin the study may help eliminate some of the mystery a new user may have with ultrasound setups. These presets are only a suggestion and can be manipulated by the user anytime during the examination. The settings utilized will influence results, so familiarity with each setting’s impact on the image is crucial. These settings include depth, overall gain, time gain control (TGC), scale, and wall filters, to name a few. The presets utilized for a diagnostic study are unique in comparison to those selected for therapeutic applications. For example, during a diagnostic survey the dynamic range is set high to discover complexities of tissue and structures. During therapeutic interventions, decreasing the dynamic range will enable detection of high contrast objects, such as needles and catheters.
A variety of factors will influence which ultrasound instrumentation to purchase. Some of these considerations are intended use, cost, portability, flexibility, and ergonomics. Of greatest importance to this decision is consideration of diagnostic quality and the ease with which results can be obtained.
Ultrasound Training
The use of ultrasound technology has become a routine tool within the specialty of phlebology. The expertise and training necessary for individuals who utilize ultrasound will vary. The American College of Phlebology (ACP) offers ultrasound fellowship training opportunities specific to venous applications. Other training may include rotational fellowships and regional ultrasound courses. Study and practice are necessary to obtain experience and confidence in one’s ability.
Professional medical societies such as the American College of Radiology (ACR) recommend that ‘physicians responsible for diagnostic ultrasound examinations be able to demonstrate familiarity with the anatomy, physiology, and pathophysiology of those organs or anatomic areas that are being examined’. Though the intent may be to examine only the venous structures, these are not the only anatomical features seen on routine examination. Imaging from the skin line will demonstrate subcutaneous fat, muscle, tendons, arteries, nerves, joint spaces, periarticular bursae, and bone.
The American College of Surgeons (ACS) suggests that ‘physicians performing ultrasound examinations and ultrasound-guided procedures must be familiar with the principles of ultrasound physics, and the indications, advantages, limitations, performance, and interpretation of the ultrasound examinations. Further criteria of personnel performing ultrasound under the supervision of the surgeon must be appropriately trained and certified and their performance regularly evaluated within the framework of the quality improvement process.’
Ultrasound credentialing can be obtained through independent bodies such as Cardiovascular Credentialing International ( www.cci-online.org ) or the American Registry of Diagnostic Medical Sonographers ( www.ardms.org ). Certification examinations are strongly encouraged for individuals performing diagnostic ultrasound. The examination of RPhS (Registered Phlebology Sonographer) is directly linked to individuals performing phlebology ultrasound and would be a worthwhile achievement.
Government and State regulations exist regarding not only the individuals performing diagnostic ultrasound but also the accreditation of the ultrasound facility. Many private insurers also restrict reimbursement for ultrasound usage based on a variety of factors. Overall these variable policies may impact the use of ultrasound within an office or medical setting.
Diagnostic Applications
•
Patient assessment
Diagnostic considerations for patients who present with chronic venous disease will vary, since much depends on the clinical circumstances. Within previous chapters, great detail has been given about how to assess a patient with venous complaints. A summary of the patient’s medical, surgical and familial history is essential. Not to be overlooked is a simple question regarding the presence of birth marks. For example, port wine stains may initiate a multifaceted study in search of a vascular malformation diagnosis. Though the diagnostic complexity may fluctuate, a standard written protocol is imperative. The aim of the diagnostic study is to determine the function and status of the deep and superficial venous structures. Since this information may likely be the basis on which treatment is planned, an accurate description of any anatomical and physiological abnormality will be essential. Without this, the method of treatment considered necessary to improve the patient’s condition will be unclear. Patients also frequently request a copy of their medical records, and a thorough ultrasound report with documented hard copy findings will facilitate care elsewhere should the patient make this decision.
Traditionally, patients who are referred for a diagnostic ultrasound study are symptomatic, and the investigation will relate to the symptomatology described. For instance, unilateral edema will have a unique diagnostic aim compared to a patient with bilateral edema. If the patient presents with cosmetic concerns (such as ‘matting’ or new telangiectasias) and has failed sclerotherapy, the basis for examination is to identify an explanation for the surge of new vessel occurrence. Patients may present with the mildest form of skin changes (telangiectasias) to the most severe (ulceration). The primary aim of the diagnostic study should be to identify the pathologic phenomenon contributing to the patient’s disease. The role of the diagnostic study in the evaluation of patients presenting with symptomatic chronic venous disease would be to identify a reason commensurate with the patient’s complaints and/or to provide a basis for the distribution of varicose veins clinically manifested. Patients presenting with atypical diagnostic complexities may be better served if evaluated by a more experienced sonologist.
•
Diagnostic duplex evaluation
The diagnostic assessment is of utmost importance for good clinical outcomes. It must not be rushed, as a meticulous description of the anatomic and physiologic findings will be an essential aid in the patient’s course of treatment.
Proper diagnosis and identification of key anatomic elements will impact treatment planning of the great or small saphenous veins or tributaries of the superficial veins. Variations exist within the venous system, thus the sonologist must have a thorough and accurate knowledge of both deep and superficial venous anatomy and pathophysiology, as described within previous chapters of this text.
Consensus documents from the Union of International Phlebology (UIP) detailing the ultrasound anatomy and the principles of ultrasound diagnosis were published in 2006 ( , ). These documents are an excellent resource and are recommended by this author for further reading and study.
•
Reflux studies
The identification of venous valvular reflux (i.e. retrograde flow) is actually a composite of information. The veins in the lower extremity are divided into three compartments and are comprised of the deep, saphenous and superficial veins. As one begins to examine one vein or compartment, it is apparent that all the compartments are intimately interconnected, and it is impossible to segregate one without consideration of the other two. All three compartments of the venous system are suspended between the balances of flow determined by pressure gradients. A reflux examination will evaluate all three compartments, and the total of all findings should explain the patient’s clinical presentation and equal the sum of ultrasound conclusion.
Venous flow between the compartments is synchronized by a series of one-way valves intended to open and close, facilitating drainage towards the heart. Described as the hierarchy of drainage, blood flows from the microcirculation to superficial tributaries to saphenous to the deep venous system or from the superficial veins to the deep system via perforating veins. Duplex ultrasound examination of the venous system to study reflux is a search for any flow abnormality between the compartments, both at their beginning and at their end. Abnormal flow is the detection of flow disrupting the drainage hierarchy.
The study aim of venous valvular function is to identify reflux, defined essentially as retrograde flow for an abnormal duration of time. Venous valvular reflux can be observed using spectral Doppler and/or color flow, relying on various maneuvers to study flow in provoked response to valve closure/function (see Box 4.1 ). These methods impose force to generate a pressure gradient that will stimulate normal one-way valves to close. Valves failing to close permit abnormal retrograde flow (reflux). The duration of retrograde flow considered to be abnormal has been studied. According to noted author and scientist, Dr. Nicos Labropoulos, unique reflux values are applied to deep, superficial, and perforating veins (see Table 4.1 ). Though reflux values as an absolute time have been described, keep in mind the reported research values were obtained by experienced users. Artifact can occur at the site of reflux evaluation, particularly if obtained by less experienced users or if the calculation of valve closure timing is incorrect. A conservative reflux time measure, in the author’s opinion, is 1.5 second of retrograde flow in the deep system and 1.0 second of retrograde flow in the superficial system. New research and understanding of reflux values in perforating veins is evolving. As one studies flow in the perforating veins, retrograde (outward) flow may be discovered and conservatively measured at 0.5 seconds. Vein diameter and the venous reservoir capacity influence reflux duration as well, and relationships have been described between vein diameter and reflux.
Methods Utilized to Elicit Valve Closure to Observe Reflux
The transducer is held to the skin over the vessel of interest
- •
Valsalva maneuver: movement of blood occurs with inspiration and cessation of moving blood with forced contraction. Retrograde flow observed during forced contraction is positive.
- •
Compression of distal veins: movement of blood occurs with augmentation of the tissue distal to the transducer and cessation of blood movement upon augmentation release. Retrograde flow observed during release is positive.
- •
Use of rapid cuff inflation/deflation device: a standardized cuff is positioned onto the thigh or calf approximately 5 cm. below the ultrasound transducer. With rapid cuff inflation forward movement of blood is observed. With rapid deflation cessation of blood movement is observed. Retrograde movement of blood observed with deflation is positive.
- •
Parana maneuver (modified): movement of blood is provoked with the patient’s muscular contraction and relaxation. The patient swaying slightly forward (contraction) enhances blood movement (systole). As the patient balances back during relaxation (diastole), blood movement ceases. Observation of retrograde blood movement during relaxation is positive for reflux.
| GSV | SSV | Perforators | Femoral vein | Popliteal vein |
|---|---|---|---|---|
| >0.5 sec | >0.5 sec | >0.35 sec | >1.0 sec | >1.0 sec |
The amount of reflux necessary to produce clinical symptoms is yet to be established. However, patients with reflux consistently describe complaints similarly, including pain, aching, fatigue, heaviness, swelling, itching, or spontaneous venous rupture. Asymptomatic patients with large varicose veins are not uncommon.
Reflux studies are an ultrasound observation of venous blood flow movement within the vessel and from one compartment to the next, or movement towards the heart. Prior to inspection for ‘abnormal’ retrograde flow, or reflux, an understanding of ‘normal’ antegrade flow is fundamental. Otherwise, the significance of retrograde flow identified may be overvalued or misunderstood. Reading earlier chapters will explain the pathophysiologic role of the superficial, perforating, and deep veins. In earlier text, the hierarchy of venous drainage was described. In patients presenting with advancing stages of varicose veins, the blood is no longer sufficiently drained because there is a discrepancy between reflux volume and drainage volume. A reflux examination is an attempt to replicate or mimic dynamic blood flow. Gravity plays a role in the study of venous valvular dynamics, thus a standardized duplex study for reflux is obtained while the patient is standing. Other variables of reflux detection may include time of day, temperature, hormonal influences, and proximal obstruction.
Before beginning the study of ultrasound venous valvular function with duplex, first obtain an anatomic survey to visualize the venous system from the deepest to the most superficial. Too frequently the studies are performed in opposite fashion, without attention to what is of fundamental importance.
•
Examination of the deep venous system
The historical application of duplex ultrasound has been targeted at the deep venous system, and there are numerous resources and publications available detailing the deep venous examination. Further reading should include published standards by the Society of Vascular Ultrasound (SVU) or the Intersocietal Commission for the Accreditation of Vascular Laboratories (ICAVL). Appropriate flow-sensitive ultrasound instrumentation and transducers are necessary to achieve accurate results.
As emphasized above, the study of the three compartments will begin with the deep venous system, an essential step prior to evaluating the saphenous and superficial venous system. The principle of deep venous diagnosis relies on three methods: compression, color flow, and spectral Doppler. The following text explains a focused protocol for a patient presenting with symptomatic varicose veins without suspicion for vascular malformation, pelvic insufficiency, or deep venous obstruction. Though a normal study is anticipated in most patients presenting with primary varicose veins, the sonologist must be skilled at detecting abnormal ultrasound findings. The individual patient’s clinical presentation and/or consequent abnormal ultrasound findings may determine if a deviation from a standard protocol is warranted.
The study of the deep venous system is best performed with the patient supine, and with the hip rotated slightly outward. Begin with an anatomic survey in cross-sectional imaging with a transverse orientation of the transducer (described as the transducer rotated 90°). Identify the common femoral vein (CFV) at the level of the groin crease. Immediately adjacent to the CFV is the common femoral artery (CFA) ( Fig. 4.2 ). The arterial structures are an important deep venous ultrasound landmark for the examiner. The two vessels should be relatively equal in caliber and echogenicity. Marked vessel disparity may suggest aneurysmal dilatation, and the relevance of this finding will correspondingly have significance to either vessel. Additionally, identify the presence of any echogenic foci either within the vein ( Fig. 4.3A,B ) or the artery ( Fig. 4.4A,B ). Echogenicity within the vessel lumen are considered abnormal findings either as thrombus (venous) or atherosclerosis (arterial). With the patient relaxed, compression with the transducer at the skin’s surface should achieve complete coaption of the vein walls ( Fig. 4.5 ). The arterial structures do not collapse. The compression study is a careful full-length survey of the deep venous system and includes the common femoral (CFV), femoral (FV), deep femoral (DFV), popliteal (POP), posterior tibial (PTV), anterior tibial (ATV) and peroneal veins (PERO). This portion of the study should not be rushed, as there are variations to the anatomy. Compression and coaption of the veins is the mainstay of excluding deep vein thrombosis. Areas of venous noncompressibility suggest thrombosis within the lumen.