Abstract
With the steadily advancing age of living populations and the concomitant increase in the frequency of chronic diseases such as diabetes and atherosclerosis, chronic ulcers have become one of the most common and costly dermatologic ailments. Chronic ulcers often represent significant therapeutic challenges and sometimes diagnostic challenges. Accurate diagnosis is key to successful treatment as similar clinical presentations can differ dramatically in their response to a given therapeutic modality. For decades, chronic ulcer treatment relied primarily upon standard and advanced dressings as well as surgical interventions. Recently, our approach has dramatically changed, taking advantage of both medical therapeutics and advanced technologies. Lastly, multiple studies have clearly established the critical role of preventive protocols in decreasing the incidence of this epidemic of modern times.
Keywords
ulcer, venous ulcer, arterial ulcer, hypertensive ulcer, neuropathic ulcer, diabetic ulcer, chronic ulcer, wound healing, lymphedema, diabetic foot, pressure sores, pressure ulcers, mal perforans, ABI
▪ Ulcer – wound, sore ▪ Venous ulcer – ulcer due to venous insufficiency/venous hypertension ▪ Arterial ulcer – ulcer due to atherosclerosis ▪ Neuropathic ulcer – mal perforans ▪ Livedoid vasculopathy – atrophie blanche, livedoid vasculitis ▪ Pressure ulcer – decubitus ulcer, pressure sore, bed sore
- ▪
The history should include onset, course, symptoms, exacerbating and alleviating factors, past medical history, family history, social history, travel, and medications
- ▪
Important aspects of the physical examination include location, size, shape and depth of the ulcer, characteristics of the ulcer edge and base, and associated physical findings
- ▪
Helpful laboratory evaluation includes vascular studies, blood tests (e.g. for thrombophilia), microbial cultures, and biopsy
- ▪
A biopsy of non-healing chronic ulcers is indicated to exclude carcinoma or other underlying diseases
- ▪
In addition to local care, therapy of ulcers should aim to correct or treat the underlying cause:
- •
Venous insufficiency: compression
- •
Arterial insufficiency: revascularization
- •
Neuropathic or pressure ulcer: relief of pressure
- •
Infection: antibiotics
- •
Neoplasm: surgery, chemotherapy, radiotherapy
- •
Vasculopathy: treat the underlying cause of the occlusive disorder
- •
Vasculitis or other inflammatory process (e.g. pyoderma gangrenosum): corticosteroids, immunosuppressive agents
- •
- ▪
A key factor in therapy is the promotion of wound healing:
- •
Debridement: mechanical, enzymatic, autolytic
- •
Dressings: maintain a moist wound environment
- •
Infection control: antibiotics
- •
Skin grafting or skin substitutes
- •
Growth factors
- •
Introduction
A chronic wound is defined as one that fails to progress through a normal orderly and timely sequence of repair or a wound that passes through the repair process without restoring a functional result . A cutaneous ulcer is a wound that is associated with loss of both epidermal and dermal tissues. Chronic ulcers, which are due to a variety of systemic and/or local factors, can represent both a diagnostic and therapeutic challenge to the dermatologist. The majority of ulcers occur on the lower extremities, and most are related to venous insufficiency/venous hypertension, peripheral artery disease, or peripheral neuropathy ( Table 105.1 ). Less common causes are outlined in Fig. 105.1 and sites other than the legs may be involved (e.g. pressure-induced ulcers).
| COMPARISON OF CLINICAL FINDINGS IN THE THREE MAJOR TYPES OF LEG ULCERS | |||
|---|---|---|---|
| Venous | Arterial | Neuropathic/mal perforans * | |
| Location | Medial malleolar region |
| Pressure sites |
| Morphology |
|
| “Punched out” |
| Surrounding skin |
| Shiny atrophic skin with hair loss | Thick callus |
| Other physical examination findings |
|
|
|
Wound therapy has advanced considerably over the past few decades with the advent of innovative dressings and technologies, but the key factor in the management of chronic ulcers remains proper patient evaluation and correction of the underlying cause, when identified.
Venous Ulcers
Chronic venous disease is a common problem in adults, with its prevalence increasing with age. Due in large part to a lack of standardized diagnostic criteria, the prevalence varies considerably, from <1% to 17% in men and from <1% to 40% in women .
Risk factors for chronic venous disease include family history, age, female gender, obesity, pregnancy, prolonged standing, and height . Venous hypertension and venous insufficiency represent the most common cause of chronic leg ulcerations. Venous ulcers tend to recur, are sometimes painful, and clearly have an impact on quality of life . Because of their prevalence and chronicity, there are significant healthcare costs, estimated to be ~$3 billion per year in the US .
Pathogenesis
The venous system of the lower extremities consists of an interconnected network of superficial veins, deep veins, and perforator or communicating veins which connect the superficial and deep systems (see Ch. 155 ). Throughout the superficial, communicating and deep veins, one-way bicuspid valves ensure unidirectional flow toward the deep system, thus allowing blood to flow in a cephalad direction and preventing reflux ( Fig. 105.2A ). It is primarily the contraction of calf muscles that drives blood from the leg toward the heart .
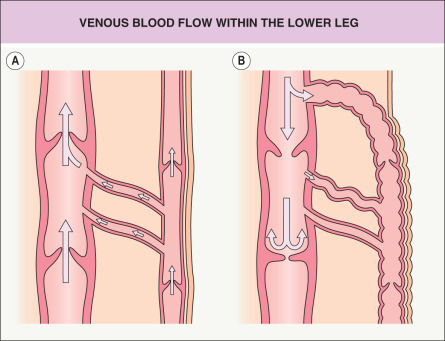
During standing, as leg muscles relax, venous pressure in the legs may reach up to 80 to 90 mmHg . Calf muscle contractions, e.g. when walking, transiently increase pressure within the deep leg veins, propelling blood towards the heart. When pressure rises within the deep system, the valves close, preventing retrograde flow and transmission of high pressure to the superficial system . When the deep system empties, the deep vein pressure abruptly falls to <30 mmHg and the valves open, enabling flow from the superficial into the deep system .
In the absence of competent valves, however, the decrease in venous pressure with leg movements is attenuated, and high pressures generated in the deep veins by calf muscle contraction are transmitted to the superficial venous system and to the microcirculation within the skin . A sustained elevation in venous pressure is responsible for the typical clinical picture seen in chronic venous disease and is termed venous hypertension or venous insufficiency .
In most patients, venous hypertension is caused by reflux through incompetent valves ( Fig. 105.2B ), whether primary (congenital) or secondary to deep vein thromboses or recurrent trauma. Additional causes of venous hypertension include venous outflow obstruction and failure of the calf muscle pump due to obesity or leg immobility .
Although venous hypertension appears to be central to the skin changes seen in chronic venous disease, the exact pathogenic cascade from valvular incompetence to frank ulceration remains obscure. A few pathomechanisms have been suggested, but it is still unclear whether they represent causative factors or epiphenomena . One proposal was that cuffs of fibrin around dermal capillaries (due to leakage of fibrinogen) could impede the diffusion of oxygen and nutrients to surrounding tissues and lead to degenerative skin changes . Other investigators suggested that venous pooling induced inter-endothelial space widening and deposition of fibrin and other macromolecules that then “trapped” growth factors, rendering them unavailable for wound repair .
Nowadays, however, the predominant theory is that chronic inflammation plays a key role in chronic venous ulcers . In the setting of elevated venous pressure, there is entrapment of leukocytes within small blood vessels and the skin of the lower extremity (referred to as the microvascular leukocyte-trapping hypothesis; Fig. 105.3 ) . These trapped leukocytes become activated and they initiate an inflammatory response that leads to cellular and tissue dysfunction, resulting in the dermal changes often observed in patients with chronic venous insufficiency.
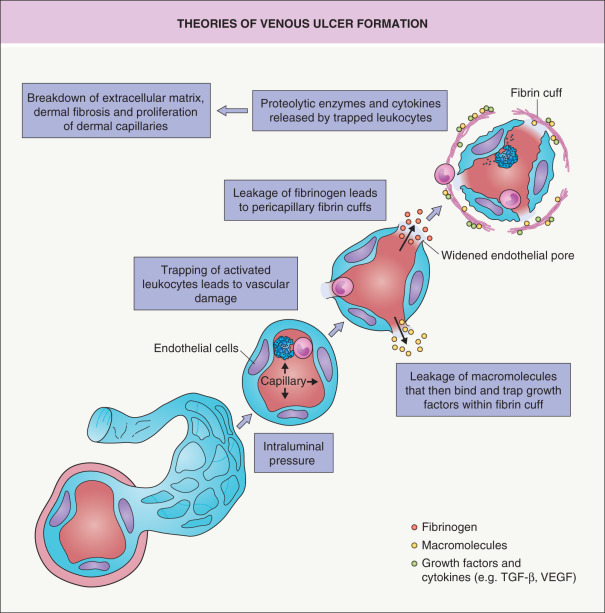
Expression of specific adhesion molecules on endothelial cells attracts various leukocytes (e.g. macrophages, T lymphocytes, mast cells) into the dermis via diapedesis (see Ch. 102 ), where they further promote inflammatory responses via the release of cytokines and activated proteinases . Specifically, increased expression and activity of matrix metalloproteinases (MMPs) contribute to the breakdown of the extracellular matrix, which promotes the formation of ulcers and impairs healing. Elevated levels of cytokines such as transforming growth factor-beta (TGF-β) and vascular endothelial growth factor (VEGF) translate into increased dermal fibrosis and proliferation of dermal capillaries, respectively (see Fig. 141.3 ). Capillary hyperpermeability and extravasation of red blood cells lead to deposition of ferric iron within affected skin, resulting in brown discoloration. A persistent inflammatory response can eventually give rise to ulcer formation, as continued production of MMPs and cytokines (e.g. interleukin-1 and tumor necrosis factor-alpha [TNF-α]) leads to impaired fibroblast function and increased degradation of growth factors necessary for wound healing. In addition, oxidative stress-induced premature aging and dysfunction of wound fibroblasts contribute to the chronicity of venous ulcers .
Clinical Features
Chronic venous disease is associated with a wide spectrum of clinical findings, ranging from uncomplicated telangiectasias and varicose veins to lipodermatosclerosis and large chronic ulcerations . Patients with venous ulcers often complain of swelling and aching of the legs, typically worsened by dependency and prolonged standing but improved by leg elevation, use of gradient support stockings or walking, all of which decrease venous pressure. Additional symptoms sometimes ascribed to chronic venous disease include muscle cramps, pruritus, and restless legs .
Most commonly, venous ulcers are located above the medial malleolus ( Fig. 105.4 ). The ulcer is usually shallow with a border that is typically irregular; the wound bed is often covered by a yellow, fibrinous exudate. If the latter is appropriately debrided, a base of healthy, non-ischemic granulation tissue is usually seen. When untreated, venous ulcers tend to become larger than most other chronic wounds and can even become circumferential on the lower extremity . Pedal pulses are usually palpable. If they are not palpable because of induration or edema, an ankle–brachial index (see below) should be obtained to exclude concomitant peripheral artery disease .
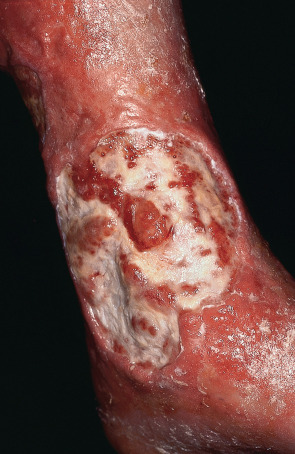
Characteristic associated features include ( Fig. 105.5 ): (1) varicose veins of varying severity (see Ch. 155 ); (2) pinpoint petechiae superimposed on a yellow–brown discoloration due to extravasation of red blood cells and deposition of hemosiderin within macrophages (see Fig. 22.6 ), which is sometimes accompanied by hyperpigmentation; (3) edema; and (4) stasis dermatitis with pruritus, erythema, scaling, and weeping (see Ch. 13 ). Of note, stasis dermatitis is sometimes aggravated by allergic contact dermatitis to topically applied medications, e.g. neomycin. The smooth, ivory-white, sclerotic plaques of atrophie blanche that are often surrounded by punctate telangiectasias and/or brown discoloration are observed in up to 40% of patients with chronic venous insufficiency .
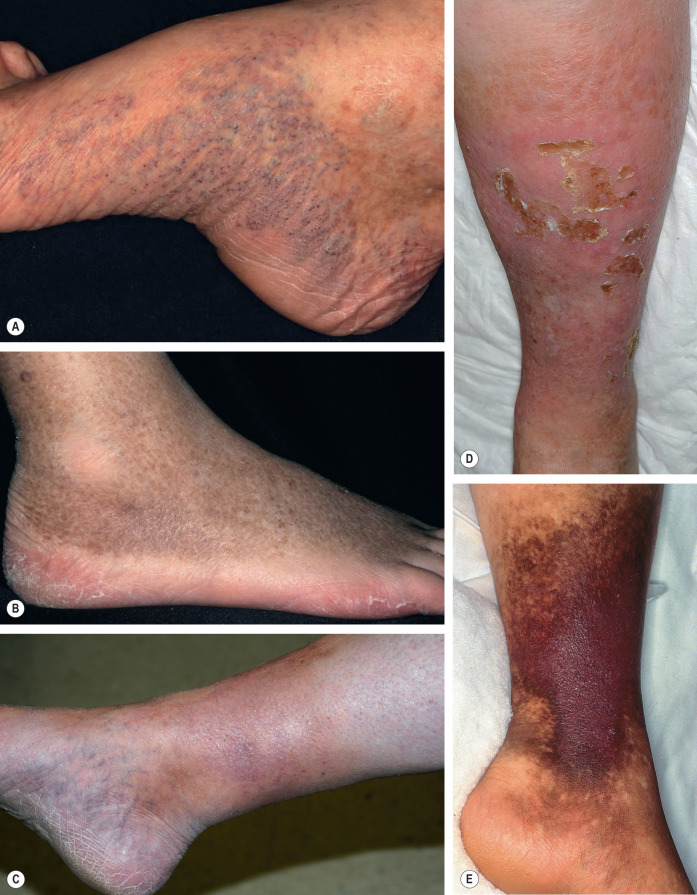
Patients with longstanding venous hypertension and insufficiency may develop chronic lipodermatosclerosis (sclerosing panniculitis) with induration and fibrosis that begins on the lower medial aspect of the leg above the malleolus (see Fig. 105.5C, D ). Eventually, the entire lower third of the leg may become involved, leading to an appearance that has been likened to an “inverted champagne bottle”. Some authors believe the degree of lipodermatosclerosis correlates with delayed wound healing. Chronic lipodermatosclerosis may be preceded by an acute inflammatory stage characterized by warm, erythematous, firm plaques that are tender and painful and are often misdiagnosed as cellulitis or another type of panniculitis, especially erythema nodosum .
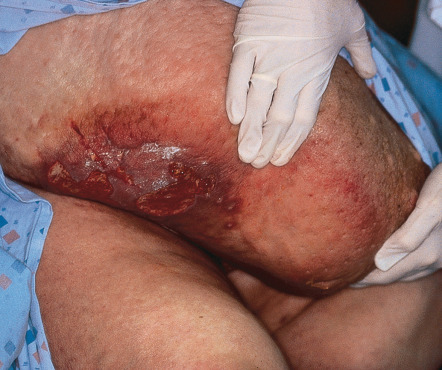
Lipodermatosclerosis, along with ulcerations, is not limited to the lower extremities, as these changes can be seen in other sites such as the most dependent portion of the pannus ( Fig. 105.6 ).
Laboratory Evaluation
Standardized evaluation of patients with chronic venous disease is based on the CEAP classification system which generates scores derived from objective clinical signs (C), etiology (E), anatomic (A) location of the affected veins, and pathophysiology (P) (reflux, obstruction or both) . Table 155.1 outlines the details of the CEAP classification system.
Duplex ultrasonography, preferably performed with the patient in a standing position, is useful for assessing both reflux and obstruction within the deep, superficial and perforating veins, from the inferior vena cava to the calf veins. It provides information that is necessary for determining the CEAP score. Some clinicians perform venography if an intervention is planned. Ascending venography is recommended for patients with post-thrombotic disease as it gives a detailed anatomic map of the venous return of the lower extremity, whereas descending venography is helpful in assessing valvular disease and estimating reflux severity. Experience with high-resolution magnetic resonance venography and computed tomography venography is still limited, but should increase over the next decade .
Pathology
In the skin surrounding the ulceration, nonspecific features are seen including reactive epithelial hyperplasia, dermal fibrosis and an inflammatory infiltrate, along with a proliferation of capillaries, extravasation of red blood cells, and deposits of hemosiderin within macrophages. Biopsies are occasionally performed to exclude a neoplastic, vasculitic or vasculopathic process.
Differential Diagnosis
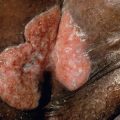
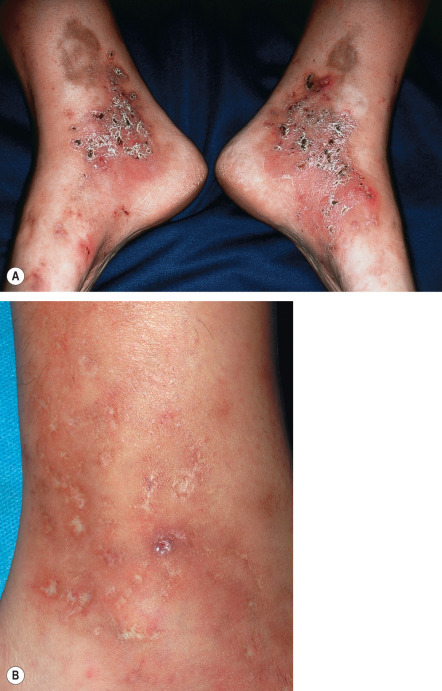
A significant number of patients may have a combination of venous and arterial insufficiency, so before treatments such as compression stockings are prescribed, the latter should be excluded (see below). In patients with livedoid vasculopathy ( Fig. 105.7 ) or recurrent deep vein thromboses, the possibility of thrombophilia (hypercoagulability) as a contributing factor needs to be considered (see Table 105.9 & Ch. 23 ). Additional clues from the history, e.g. rate of expansion, and physical examination aid in narrowing the differential diagnosis ( Table 105.2 ). Histologic examination of a longstanding or recalcitrant ulcer is recommended to exclude other diagnoses, such as squamous cell carcinoma or atypical mycobacterial infection (see Fig. 105.1 ), and the biopsy specimen should include surrounding non-ulcerated skin.
| POINTS TO INCLUDE IN THE HISTORY AND PHYSICAL EXAMINATION OF PATIENTS WITH A LEG ULCER | |
|---|---|
| History | Physical examination |
| General, emphasizing the following:
|
Treatment
Most of the therapeutic principles described in the following sections can be applied to the care of other types of cutaneous ulcers, unless otherwise specified. In general, normal wound healing requires adequate perfusion, good nutritional and immune status, and avoidance of deleterious mechanical forces . The initial step in the approach to an ulcer should be to address all systemic and local factors that may impair healing.
Moisture and occlusion
In the past, the mainstay of wound management was to dry the wound bed with absorptive gauzes. A major breakthrough in the management of chronic wounds was the realization that a moist environment is critical for wound healing and occlusive dressings do not increase the risk of infection . Moist retentive wound dressings stimulate collagen synthesis, promote angiogenesis by creating a hypoxic environment, encourage re-epithelialization, and decrease pain. By serving as a barrier against external contamination and by lowering wound pH, occlusive dressings actually decrease wound infection rates .
Dressings should be changed based on their potential for absorbency and the amount of exudate, with attention to the delicate balance between overhydration and excessive dryness. For example, occlusive dressings applied to highly exudative wounds may cause maceration and erosion of the surrounding skin, possibly leading to further deterioration of the wound. In addition, chronic wound fluid can perpetuate cytokine and MMP production (see above), thereby affecting the function of fibroblasts and growth factors and contributing to chronicity. It is therefore important to protect the skin surrounding the wound bed with an ointment in order to preserve barrier integrity .
Debridement
Debridement consists of the removal of necrotic, non-viable, or infected tissue from the wound bed in order to promote healing. In a sense, debridement is an attempt to shift the wound from a chronic state (with a milieu characterized by an abnormal extracellular matrix, an excess of proteolytic enzymes, and senescent cells) into an acute state. Debridement can be surgical (sharp debridement), mechanical (wet-to-dry), autolytic, chemical (enzymatic), and biologic (maggot therapy) .
Sharp debridement with surgical instruments represents the most rapid method for debriding a wound and is most appropriate for large necrotic and/or infected wounds. It also enables the clinician to estimate the extent and severity of a wound. Although damage to viable tissue is possible, sharp debridement performed by a skilled physician is usually highly selective . Caution is advised in the case of arterial ulcers because of the potential risk for tissue desiccation and ulcer enlargement . Sharp debridement may be associated with bleeding and pain and may necessitate analgesia.
Wet-to-dry (mechanical) debridement consists of placing gauze moistened with saline over the wound, allowing the gauze to dry, and then removing the dry gauze along with adherent non-viable tissue. It is relatively fast and easy to perform, but is painful and does not discriminate between viable and non-viable tissue. It is indicated for wounds with large amounts of necrotic tissue.
Autolytic debridement is the process by which the body uses its own proteolytic enzymes and phagocytic cells to clear necrotic debris. The process is enhanced by moist retentive wound dressings and may require weeks to accomplish. Autolytic debridement is selective, painless, and suited primarily for wounds with minimal debris. It is contraindicated if the wound is infected .
Enzymatic (chemical) debridement utilizes topically applied proteolytic enzyme preparations such as papain–urea or collagenase to digest necrotic tissue, collagen, fibrin, and wound exudate. Chemical debridement is a gradual process. The papain–urea preparation contains papain, which is derived from the plant Carica papaya and is a non-selective cysteine protease. It is active over a broad pH range and is associated with an intense inflammatory response and digestion of viable tissue; local pain or a burning sensation is often described. Therefore, the product should be applied only to the wound bed and not to the surrounding intact skin. Collagenase preparations are derived from Clostridium histolyticum ; they specifically target native collagen and are active within a narrow pH range of 6 to 8. These preparations are highly selective and do not harm viable cells . Prior to application, it is advisable to cross-hatch the eschar, if present, in order to increase penetration .
For therapy-resistant, chronic ulcers with marked necrosis and slough, maggot debridement therapy can be considered. Essentially, it exploits the ability of maggots to decompose necrotic tissue (benign myasis), but the debridement is done in a controlled manner so as to avoid undesirable effects on healthy tissues . Degradation via proteinases that are in larval excretory/secretory products is thought to contribute to wound debridement . It is important to select larval strains that feed preferentially on necrotic tissue and to avoid applying too many larvae to a wound. The flies most often employed are facultative calliphorids, in particular the green bottle blowfly, Lucilia sericata .
Maggots are used to treat various types of chronic ulcers, including pressure ulcers, venous ulcers, and neurovascular diabetic ulcers, as well as traumatic and postsurgical wounds. In a multicenter, randomized, open study that compared the clinical effectiveness of larval therapy to a standard debridement technique (hydrogel) for “sloughy” or necrotic leg ulcers, larval therapy did not improve the rate of healing or reduce bacterial load, but it did significantly reduce the time to debridement and increase ulcer pain .
Novel approaches to wound debridement are currently under investigation, including both hydrosurgery and ultrasound- and radiofrequency-based techniques .
Wound dressings
The ideal wound dressing should: (1) provide a moist retentive environment; (2) absorb excess exudate without causing maceration of surrounding skin; (3) protect against bacteria; (4) leave no residual debris but cause no trauma when removed; (5) decrease odor; and (6) relieve pain. In addition, the ideal wound dressing should be cost-effective, easy to handle, and be neither irritating nor allergenic. Obviously, there is no single dressing that can fulfill all these criteria throughout the entire healing process. A vast array of dressings exists and the choice of a dressing depends upon the wound type and its characteristics at a given time ( Table 105.3 ) .
| TYPES OF WOUND DRESSINGS | |||
|---|---|---|---|
| Dressing type | Properties | Disadvantages | Indications |
| Gauzes |
|
|
|
| Films |
|
|
|
| Hydrogels |
|
|
|
| Hydrocolloids |
|
|
|
| Alginates |
|
|
|
| Foams (polyurethane) |
|
|
|
| Collagens |
|
|
|
Management of wound infection
The presence of a microbial infection is always detrimental to wound healing. However, all chronic open wounds are colonized with bacteria and it is important, although often difficult, to distinguish true infection from bacterial colonization. Of note, colonization is defined by the presence of replicating bacteria and adherent microorganisms, but without evidence of tissue damage.
Critical colonization is a relatively new concept in which the bacterial burden within a chronic wound does not elicit the typical symptoms or signs of an infection, but does delay healing. The presence of ≥10 6 colony-forming units of bacteria/gram of tissue predicts delayed wound healing and a high risk for developing an infection . More recent studies have demonstrated that in many chronic wounds, bacteria persist in the form of biofilms. The latter represent communities of bacteria, protected by an adhesive polymeric matrix cover, that communicate with each other via water channels. Through these communication channels, bacteria are able to regulate the transcription of genes and protein products in a manner that is beneficial to them, but which can delay healing (quorum sensing) . Biofilms are particularly resistant to antimicrobial therapy.
Wounds become clinically infected when host defenses are overwhelmed. Signs and symptoms of infection include an increased exudate, purulent discharge, pain, and surrounding erythema and swelling. However, sometimes an increase in ulcer size and malodorous or friable granulation tissue are the only clues to the presence of an active infection within a chronic wound. Systemic signs of infection such as fever, chills, tachycardia, and leukocytosis may suggest that the infection has progressed to a bacteremia or septicemia, although the former may be subtle in elderly patients, necessitating a high index of suspicion.
Most chronic wounds have a polymicrobial flora. Bacterial cultures usually grow aerobic Gram-positive cocci, which are often mixed with Gram-negative bacilli and sometimes anaerobes . Clinically infected wounds should be cultured and treated initially with broad-spectrum systemic antibiotics. The antibiotic regimen is then adjusted based on the results of sensitivity testing.
In properly treated, non-healing wounds that demonstrate critical colonization, topical antimicrobials should be considered. Topical antimicrobial agents are divided into two major categories: antiseptics and antibiotics. Antiseptics have a broad antimicrobial spectrum as well as multiple microbial targets, but they are often toxic to host tissues. Topical antibiotics have specific cellular targets and a narrower spectrum of activity; they are not toxic to human cells but are more likely to induce bacterial resistance. Commonly used topical antibiotics include bacitracin, neomycin, mupirocin, retapamulin, gentamicin, and fusidic acid. However, topical antibiotics can cause allergic contact dermatitis, in particular neomycin and bacitracin. It is also preferable to avoid topical antimicrobials that have a systemic counterpart .
Commonly used antiseptics include hydrogen peroxide, chlorhexidine, iodine-based preparations (e.g. povidone-iodine, cadexomer iodine), and silver-releasing agents. Hydrogen peroxide and povidone-iodine have a broad antimicrobial spectrum with minimal resistance but may be cytotoxic. Cadexomer iodine is a complex of 0.9% elemental iodine plus cadexomer microbeads which are composed of a modified starch-based polymer. Upon absorption of exudate, the cadexomer beads swell and slowly release iodine into the wound bed, thus maintaining non-toxic iodine concentrations .
Silver is an effective antiseptic agent with broad-spectrum activity, including against methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and extended-spectrum β-lactamase producers. Resistance has rarely been reported, primarily with Gram-negative species . Only the ionized form of silver (Ag + ) has antimicrobial properties, so exposure to wound fluid or exudate is required if the source is metallic silver . Silver ions kill bacteria by damaging bacterial cell walls and intracellular and nuclear membranes as well as denaturing bacterial DNA, RNA, and key enzymes (e.g. respiratory enzymes) . Silver may also promote cellular proliferation and re-epithelialization by inducing the production of metallothionein by epidermal cells. Metallothionein increases zinc- and copper-dependent enzymes required for cellular proliferation and matrix remodeling . Silver has been incorporated into various dressing products (e.g. gauzes, hydrocolloids, alginates, foams) in addition to creams, gels, and barrier protectants which differ in their solubility and the rate at which silver ions are released into the wound bed . Disadvantages of silver products include potential irritation, permanent blue–gray discoloration (localized argyria), and cost. Topical silver sulfadiazine has induced neutropenia in children which was reversible once the cream was discontinued .
Lastly, as chronic wounds may serve as a portal of entry for Clostridium tetani , it is recommended that the tetanus immune status of patients with chronic leg ulcers be assessed .
Adjuncts to wound care
Compression
Compression therapy is a mainstay in the treatment of venous insufficiency. Compression stockings improve venous return, reduce edema, stimulate healthier granulation tissue within venous ulcers , and improve quality of life . A Cochrane meta-analysis of 39 randomized controlled trials concluded that compression therapy increased ulcer healing rates when compared to no compression .
Elastic stockings, preferably with graduated compression, are available in a wide range of compressive pressures (15 to 60 mmHg), lengths, and materials ( Table 105.4 ). Lower levels of compression (20 to 30 mmHg) are sufficient for preventing mild edema, whereas higher levels of compression (30 to 40 mmHg) are required to control stasis dermatitis or lead to the healing of ulcers. Compression stockings also prevent the recurrence of venous ulcers once healed. Therefore, lifelong use of compression is recommended for patients with a history of venous ulcers . In addition to stockings, there are other forms of compression such as bandages and wraps, as outlined in Tables 105.5 and 105.6 . Of note, surgical correction of superficial venous reflux (plus compression) does not improve ulcer healing but does reduce the recurrence of ulcers .
| CLASSES OF COMPRESSION STOCKINGS | ||
|---|---|---|
| Class | Pressure at the ankle | Indication |
| I | 20–30 mmHg | Simple varicose veins |
| Mild edema | ||
| Leg fatigue | ||
| II | 30–40 mmHg | Moderate edema |
| Severe varicosities | ||
| Moderate venous insufficiency | ||
| III | 40–50 mmHg | Severe edema |
| Severe venous insufficiency | ||
| Post-thrombotic lymphedema | ||
| IV | 50–60 mmHg | Elephantiasis |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree