Abstract
Every day, more than 1 million people worldwide are newly infected with sexually transmitted infections (STIs), leading not just to an increase in morbidity and mortality, but also a higher risk of transmission of HIV. Existing prevention and management strategies have not reduced global incidence and prevalence, making STIs an ongoing public health problem. In addition, the increases in antibiotic resistance provide health professionals with new challenges. This chapter reviews important aspects of syphilis, gonorrhea, chancroid, lymphogranuloma venereum, and donovanosis (granuloma inguinale), including diagnostic tools, clinical manifestations, and treatment guidelines.
Keywords
sexually transmitted infections, sexually transmitted diseases, STI, STD, syphilis, lues, gonorrhea, GC, chancroid, lymphogranuloma venereum, LGV, donovanosis, granuloma inguinale, genital ulcer disease, gonorrhea antibiotic resistance
Introduction
The term “venereal diseases” was historically used to refer to infections that are sexually transmitted, i.e. syphilis, gonorrhea, chancroid, lymphogranuloma venereum, and granuloma inguinale. These disorders are now referred to as “sexually transmitted diseases” (STDs) or “sexually transmitted infections” (STIs), terms that directly reflect recognition that they are caused predominantly by sexual contact with an infected person. For infections caused by pathogens for which non-sexual routes of transmission predominate, e.g. yeasts or cytomegalovirus, the term “sexually transmissible infections” is used. Table 82.1 lists the names of commonly encountered sexually transmitted or transmissible pathogens.
| SEXUALLY TRANSMITTED AND TRANSMISSIBLE PATHOGENS | |
|---|---|
| Bacteria | Neisseria gonorrhoeae Treponema pallidum Haemophilus ducreyi Chlamydia trachomatis Mycoplasma hominis, M. genitalium Ureaplasma urealyticum Gardnerella vaginalis Atopobium vaginae Mobiluncus curtisii, M. mulieris Klebsiella (Calymmatobacterium) granulomatis Shigella spp. Campylobacter spp. Helicobacter cinaedi, H. fennelliae |
| Viruses | Human immunodeficiency virus, types 1 and 2 Herpes simplex virus, types 2 > 1 Human papillomavirus (several types) Hepatitis viruses, B > C and (via fecal–oral contact) A Cytomegalovirus Molluscum contagiosum virus Human T-cell leukemia/lymphotrophic virus, types I and II Human herpesvirus, type 8 |
| Protozoa | Trichomonas vaginalis Entamoeba histolytica Giardia lamblia |
| Fungi | Candida albicans |
| Ectoparasites | Phthirus pubis Sarcoptes scabiei |
Syphilis
▪ Lues
- ▪
Sexually acquired, chronic infection caused by Treponema pallidum and characterized by a variety of clinical manifestations and involvement of multiple organ systems
- ▪
Can also be transmitted before birth (congenital)
- ▪
Intermittently active disease with primary, secondary, and tertiary stages as well as a latent period of variable length that occurs before the onset of tertiary syphilis
- ▪
Mucocutaneous manifestations vary from genital ulcers to widespread papulosquamous eruptions to granulomatous nodules
- ▪
Syphilis facilitates the transmission of HIV, especially in countries with a high rate of both infections
History
Syphilis has been of great historical importance and has played a major role in medicine for centuries. The disease was named after an afflicted shepherd named Syphilus in 1530. Theories on the origin of the disease in the late fifteenth century are controversial. One theory proposes that Christopher Columbus and his crew acquired the disease from Native Americans living in the Caribbean islands and brought it back to a nonimmune population in Europe in 1493, as signs of syphilis have been found in skeletons of pre-Columbian Native Americans . Another theory is that venereal syphilis may have already been endemic but became more widespread and severe as a consequence of the wars at that time in Europe. Lastly, the environmental theory claims that venereal syphilis is a variant of other diseases caused by Treponema spp. and became modified by environmental factors, especially temperature .
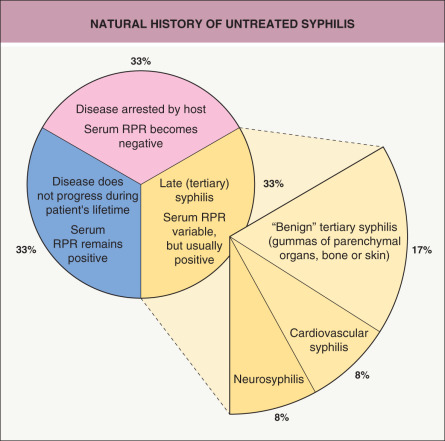
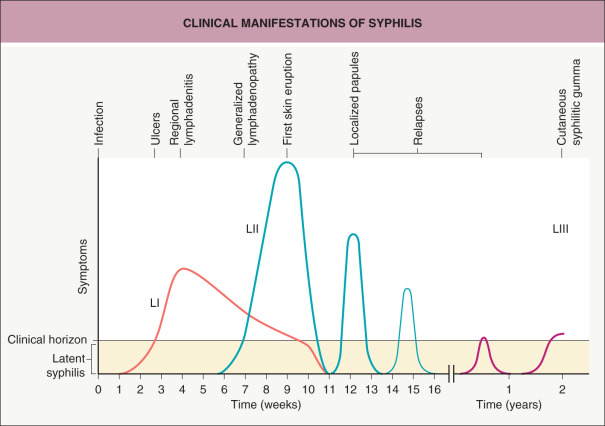
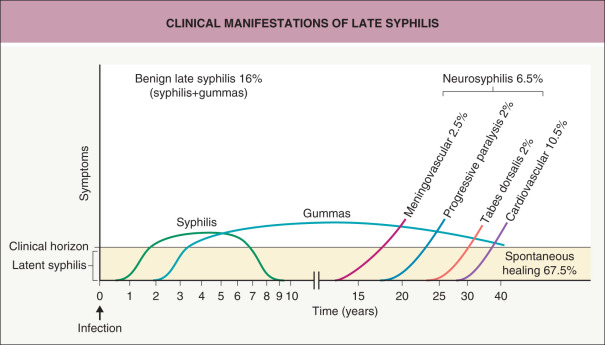
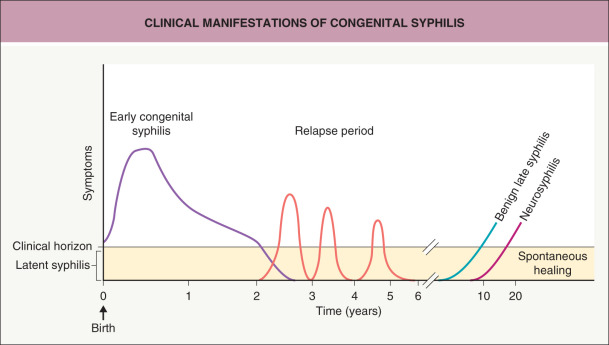
Information on the natural course of untreated syphilis ( Fig. 82.1 ) has come primarily from two large studies. The Oslo Study was a prospective investigation of infected, but untreated, individuals conducted between 1890 and 1910, with a follow-up period of 50 years. Approximately one-quarter of infected persons had at least one additional relapse of secondary syphilis, often occurring during the first year (90%). Approximately 15% of infected persons developed benign late syphilis with gummas of the skin (70%), bones (10%), and mucous membranes (10%). Cardiovascular syphilis was reported in ~14% of men and 8% of women, while neurosyphilis was observed in 10% of men and 5% of women. Altogether, the study concluded that 17% of men and 8% of women died as a result of untreated syphilis.
The Tuskegee Study was undertaken in 1932 in infected black men, with regular examinations for short- and long-term consequences of untreated infection. This represented a highly unethical investigation, as penicillin treatment was withheld without written consent. The principal finding was an increased mortality rate in the syphilitic group compared to the controls, with an ~20% loss-of-life expectancy at an interval of 12 years. Specific lesions of late syphilis were found in about 14% of infected men at the 20-year evaluation, and 12% after 30 years, with cardiovascular syphilis and neurosyphilis representing the primary causes of death .
Epidemiology
Syphilis is distributed worldwide and is particularly problematic in low-income countries, where it is a leading cause of genital ulcer disease. Worldwide, the rates of primary and secondary syphilis decreased dramatically with the introduction of penicillin treatment after the Second World War. In contrast to Western European countries, an increase in the infection rate was observed during the late 1980s in rural southern and urban regions of the US. Although the number of syphilis cases per year in the US subsequently declined and in 2000 fell to its lowest point since reporting began in 1941, over the past 15 years the number of primary and secondary syphilis cases diagnosed per year in men has more than quintupled ( Fig. 82.2 ). According to the Centers for Disease Control and Prevention (CDC), the incidence rate of primary and secondary syphilis in 2015 in the US was 13.7 cases per 100 000 population in men and 1.4 cases per 100 000 population in women. In the US, the incidence in black and Hispanic individuals is 2- to 5-fold higher than in other population groups, but the highest risk group is men who have sex with men (MSM) as they accounted for >60% of cases of primary and secondary syphilis in 2015 .
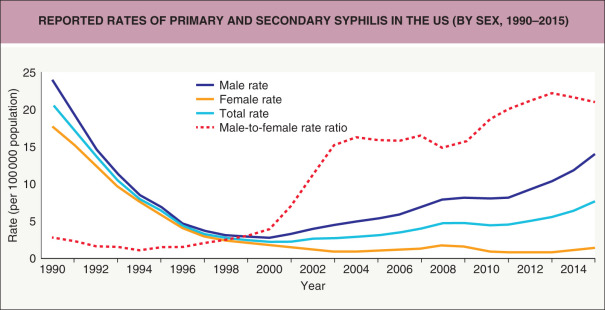
With the resurgence of syphilis in Eastern Europe in the 1990s and migration of sex workers from this region to Western Europe, syphilis has also been observed more frequently in Western European countries over the past 15 years.
Biology of T. pallidum
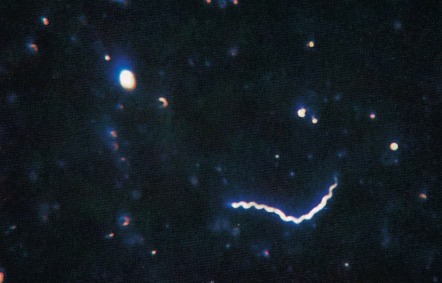
T. pallidum is a member of the genus Treponema of the order Spirochaetales, and it was identified in 1905 by Schaudinn and Hoffmann ( Table 82.2 ) . The characteristics of this microorganism are outlined in Table 82.3 . Fig. 82.3 represents a darkfield photomicrograph of T. pallidum .
| CLASSIFICATION OF TREPONEMA SPECIES | |||
| Order Spirochaetales Genus Leptospira Genus Borrelia Genus Treponema | |||
| Disease | Distribution | Transmission | |
| T. pallidum subsp. pallidum | Venereal syphilis | Worldwide | Sexual; maternal–fetal |
| T. pallidum subsp. pertenue | Yaws | Tropics | Non-venereal (all ages) |
| T. pallidum subsp. endemicum | Endemic syphilis (Bejel) | Desert | Non-venereal (all ages) |
| T. carateum | Pinta | Tropics | Non-venereal (all ages) |
| T. denticola, T. socranskii | Periodontal disease | Worldwide | Non-venereal |
| Common features shared by all spirochetes within the genus Treponema : morphology, antigenic properties, many DNA sequences, serologic detection, biochemical properties. Variable features for spirochetes within the genus Treponema : transmission mode, geography, age of patients, appearance of clinical lesions. | |||
| CHARACTERISTICS OF TREPONEMA PALLIDUM |
|
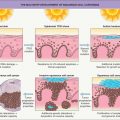
Pathogenesis of Untreated Syphilis
Syphilis is a chronic systemic infection that progresses through active and latent stages ( Fig. 82.4 ). Inoculation and penetration occurs via mucosal surfaces and abraded skin, followed by attachment to host cells and multiplication of the microorganism. Within a few hours, treponemes disseminate to the regional lymph nodes and internal organs .
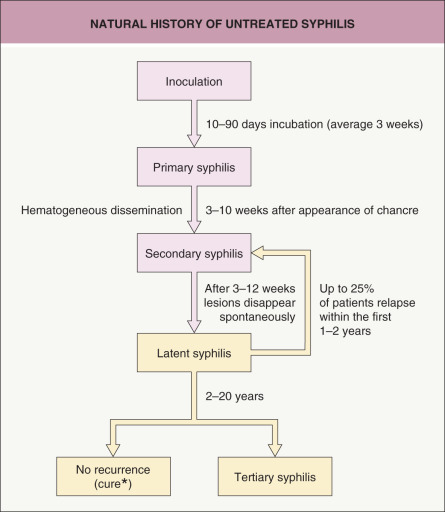
Primary stage
The primary lesion develops 10–90 days after infection (median of 3 weeks) as an indolent papule, followed by surface necrosis and the typical well-circumscribed ulceration that is firm to palpation (chancre). It is accompanied by enlarged regional lymph nodes. Histopathologically, the microorganism is observed among cells typical of a Th1-predominant cellular response; the latter leads to macrophage activation and destruction of a large number of treponemes. Several pathogenic mechanisms have been proposed, including an antigenically inert treponemal cell surface, resistance to phagocytosis, and premature down-regulation of the local host immune response.
Secondary stage
The secondary stage is characterized by dissemination and multiplication of the microorganism in different tissues, either simultaneously with or up to 6 months after healing of the primary local lesion. This stage follows primary syphilis in almost every patient who does not receive appropriate treatment. Circulating immune complexes (which contain treponemal outer membrane proteins), human anti-fibronectin antibodies, and complement are present during this stage of the disease and play a role in the pathogenesis of the different types of lesions .
Secondary syphilis is characterized by a broad spectrum of clinical manifestations involving the skin as well as systemic signs such as malaise, fever, and generalized lymphadenopathy. It lasts several weeks or months, with relapses in ~25% of patients. Pregnant women can infect a fetus via transplacental passage of the microorganism.
Latency
Latency is the period between healing of the clinical lesions and appearance of late manifestations, and it can last for many years. About 70% of untreated individuals will remain in this stage for the rest of their lives and are immune to new primary infection (see Fig. 82.1 ). Latent syphilis is divided into early (1 year or less) and late (more than 1 year) subsets, and it is characterized by positive serologic tests for specific antibodies (see below) without clinical signs or symptoms. Infectivity may occur intermittently due to the presence of treponemes in the bloodstream, and pregnant women with latent syphilis may infect the fetus in utero .
Tertiary stage
The tertiary stage is also called late syphilis and is characterized by the presence of a small number of organisms and a high cellular immune reactivity against the organism. Signs of late syphilis can be recognized in approximately one-third of untreated individuals several months to years after being infected with treponemes (see Fig. 82.1 ). The microorganisms may invade the central nervous and cardiovascular systems as well as the skin (and other organs), leading to damage related to host delayed-type hypersensitivity responses, which produce local inflammation and gummas in affected tissues.
Syphilis and HIV
Syphilis and other STIs that produce genital ulcers further increase the risk of acquiring HIV. Reasons for the increased risk of HIV transmission include:
- •
lack of an epithelial barrier due to ulceration of the skin or mucous membranes
- •
large numbers of macrophages and T cells with receptors for HIV
- •
production of cytokines by macrophages stimulated by treponemal lipoproteins.
Clinical Features
Syphilis is usually sexually acquired, but maternal–fetal transmission also occurs and can result in congenital syphilis. Syphilis is an intermittent disease with primary, secondary, and tertiary stages as well as a latent period of variable length (divided into early and late subsets) that precedes the onset of tertiary syphilis (see Fig. 82.4 ) . The definitions of early and late syphilis by the CDC and World Health Organization (WHO) differ slightly. Early syphilis includes the primary and secondary stages as well as early latency (CDC: acquired <1 year previously; WHO: acquired <2 years previously). Late syphilis extends from late latency (CDC: acquired >1 year previously; WHO: acquired >2 years previously) through the tertiary stage.
Primary syphilis
The chancre usually presents as a single, indolent, round or oval, indurated ulcer ( Fig. 82.5 ) that is associated with regional adenopathy. Some patients report a preceding painless papule that enlarged and ulcerated a few days later. The time of onset ranges from 10 to 90 days (average 3 weeks) after T. pallidum exposure. Untreated, chancres heal within a few weeks ( Fig. 82.6 ).
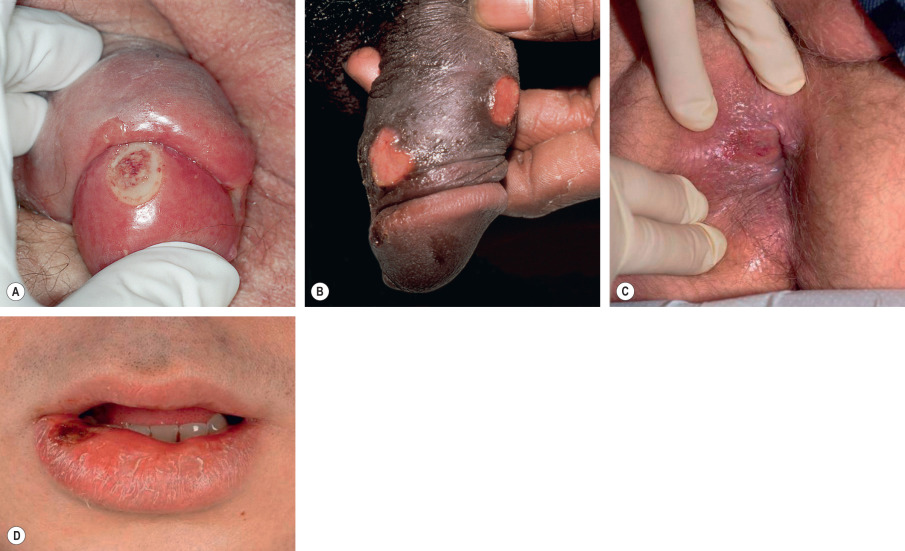
Asymptomatic infections are common due to unrecognized chancres, especially when located in the cervical region in women; in these cases, syphilis is more frequently diagnosed during the secondary stage. The same applies to chancres located in the anal, perianal, or rectal areas (see Fig. 82.5C ), seen more commonly in MSM. The mechanism of spontaneous healing without treatment is not well understood and seems to depend on local immunity.
Laboratory diagnosis of primary syphilis includes the following:
- •
The presence of treponemes by darkfield microscopic examination of fluid from the surface of the chancre is the most sensitive and specific method for the diagnosis of primary syphilis (see Fig. 82.3 ).
- •
Antibodies to cardiolipin are present in about 80% of individuals at the onset of clinical symptoms. Cardiolipin, a component of mammalian cells, seems to be modified by treponemes such that antibodies are produced by the infected person and can be measured by the rapid plasma reagin (RPR) or Venereal Disease Research Laboratory (VDRL) assay.
- •
Antibodies to surface proteins of T. pallidum as detected by hemagglutination assays ( T. pallidum h em a gglutination test [TPHA], m icro h em a gglutination assay for antibodies to T. pallidum [MHA-TP]), or f luorescent t reponemal a ntibody abs orption (FTA-ABS) assay are present in 90% of patients with clinical symptoms of primary syphilis. Since antibodies usually remain positive for life, a differentiation between primary syphilis and a prior infection may not be possible and darkfield examination should be undertaken.
Secondary syphilis
The secondary stage of the disease results from the hematogenous and lymphatic dissemination of treponemes after a few weeks or months (3–10 weeks). It is characterized by recurrent disease activity, with mucocutaneous as well as systemic manifestations. Prodromal symptoms include low-grade fever, malaise, sore throat, adenopathy, weight loss, muscle aches, and sometimes a headache from meningeal irritation ( Table 82.4 ).
| CLINICAL FEATURES OF SECONDARY SYPHILIS |
|
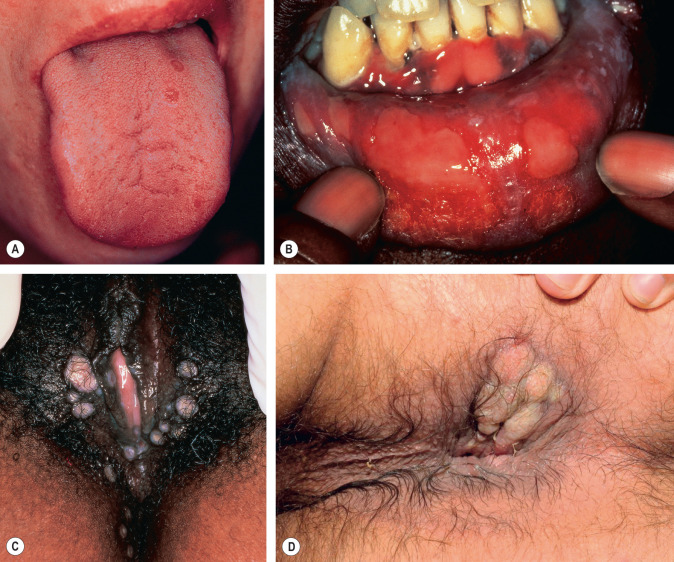
During the second stage of syphilis, the most commonly observed clinical presentation (80%) is a generalized, non-pruritic papulosquamous eruption ( Fig. 82.7 ). Lesions can range from 1–2 mm to 15–20 mm in diameter, and they vary in color from pink to violaceous to red–brown. Mucosal lesions range from small, superficial ulcers that resemble painless aphthae to large gray plaques ( Fig. 82.8A,B ). Condylomata lata are often observed in the moist regions of the anogenital area due to local spreading of the microorganisms ( Fig. 82.8C,D ). Lymph node enlargement is present in the majority of patients. Focal neurologic findings occasionally occur.
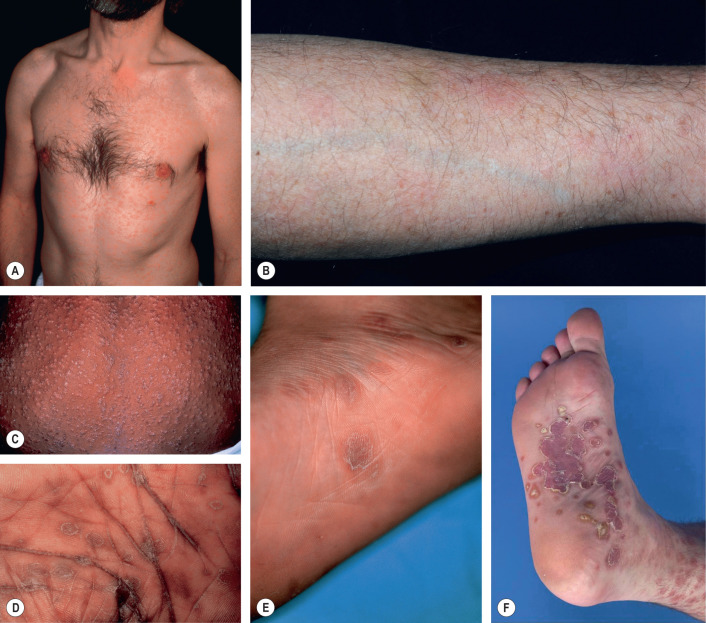
Additional clinical presentations of secondary syphilis include annular or figurate plaques with central hyperpigmentation on the face ( Fig. 82.9A ), non-scarring “moth-eaten” alopecia, split papules at the oral commissures ( Fig. 82.9B ), granulomatous nodules and plaques ( Fig. 82.9C ), and crusted necrotic lesions ( Fig. 82.9D ). Malignant syphilis (lues maligna) is extremely rare; the disseminated lesions resemble primary chancres ( Fig. 82.9E ).
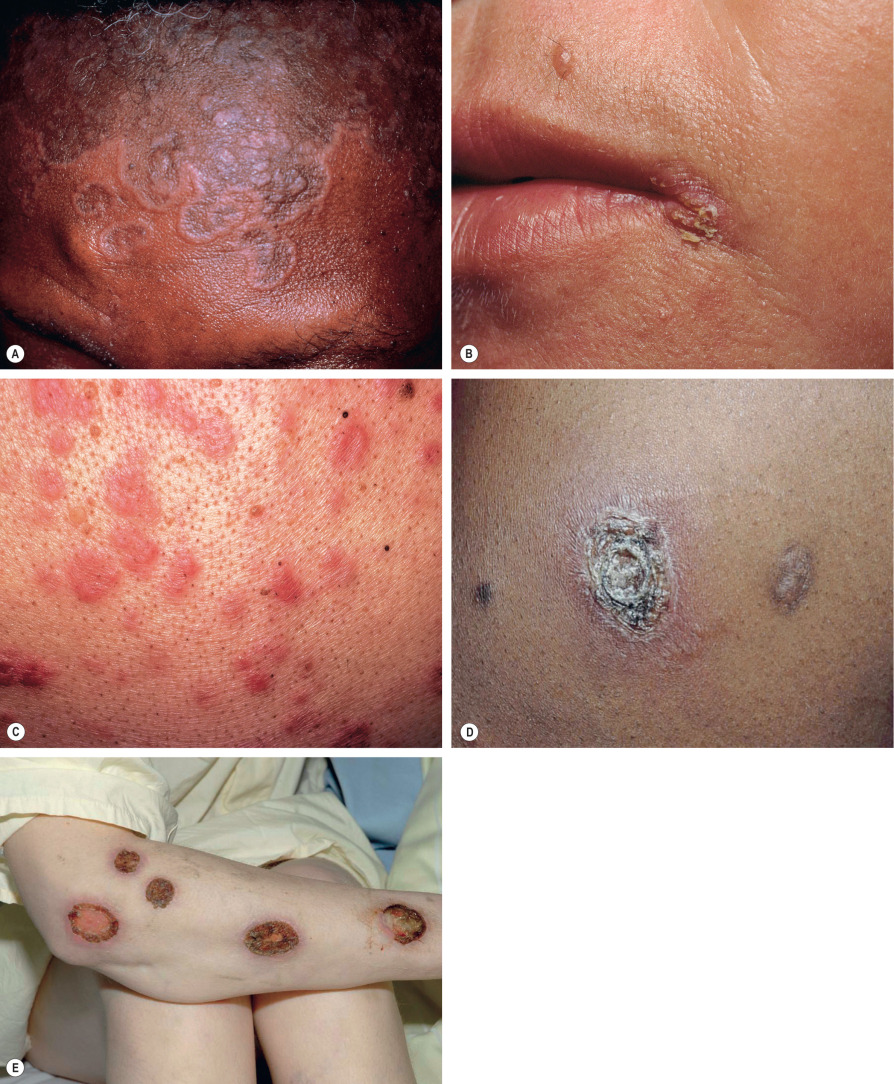
Without treatment, the lesions resolve over several weeks to months. Relapse occurs in ~20% of patients within 1 year, often with mucosal or mucocutaneous manifestations in the anogenital area.
Laboratory diagnosis of secondary syphilis includes the following:
- •
Presence of treponemes by darkfield examination of serous exudates from localized lesions of the skin and mucous membranes (the exception being the oral cavity; see below).
- •
Serologic tests are more useful in secondary, as compared to primary, syphilis. Cardiolipin antibodies (e.g. RPR or VDRL tests) as well as specific antibodies are always positive in patients with secondary syphilis; a mistakenly or temporarily negative non-treponemal test rarely occurs due to the prozone phenomenon or HIV infection, respectively (see Table 82.8 ).
- •
Spirochetes can be detected by immunohistochemistry in the majority of biopsy specimens.
- •
All patients should be tested for HIV infection, and testing should be repeated in those who fail to respond to treatment.
- •
Patients with ocular symptoms (e.g. photophobia, visual changes) should undergo ophthalmologic evaluation to assess for uveitis and neuroretinitis, including slit-lamp examination.
- •
A lumbar puncture and cerebrospinal fluid (CSF) analysis should be performed in patients with ocular or neurologic (e.g. headache, hearing loss, cranial neuropathies) signs/symptoms and when treatment failure is suspected (e.g. persistent/recurrent signs/symptoms or failure of non-treponemal test titers to decline fourfold within 6–12 months). Patients with HIV are at increased risk for neurosyphilis, especially if they have a CD4 count <350 cells/ml and/or a RPR titer of ≥1 : 32; however, unless neurologic symptoms are present, CSF examination in HIV patients has not been associated with improved clinical outcomes.
Latent syphilis
After a period of 3–12 weeks, untreated secondary syphilis typically resolves spontaneously, leaving the patient in an asymptomatic state. Diagnosis is based on a positive syphilis serology test result in the setting of no clinical evidence of treponemal infection. The two stages are:
- •
Early latent stage: within 1 year of the onset of latency (CDC definition), with the possibility of recurrence of the disease. About 90% of secondary relapses occur within the first year.
- •
Late latent stage: after 1 year from the onset of the disease (CDC definition); relapses after 1 year are very infrequent. This disease state may last for many months or years.
Diagnosis of latent syphilis is established by reactivity in an RPR or VDRL assay, a positive MHA-TP or FTA-ABS test, and the absence of any clinical signs of the disease. The diagnosis is easy to establish if there is a history of a chancre or skin manifestations that fit the clinical picture of syphilis. In many cases, however, the medical history provides no useful information regarding the presence of clinical symptoms in the previous weeks to months or whether the patient was previously treated. In such instances, differentiation between early and late latency is not possible and patients are regarded as having late latent syphilis from a management standpoint (see below). Response of latent syphilis to treatment is indicated by a decline in the RPR or VDRL titer. Persistently high (e.g. lack of a fourfold decline within 12–24 months) or rising titers are a sign of treatment failure, and examination of the CSF should be performed.
Tertiary (late) syphilis
Classic tertiary syphilis, as a consequence of untreated syphilis, is nowadays rare. It has a variable range of manifestations that appear months to years after initial infection ( Fig. 82.10 ). In particular, these include involvement of the skin, bones, CNS, heart, and great vessels. Approximately one-half of patients with tertiary syphilis have “benign” late syphilis with the development of gummas, about one-quarter develop cardiovascular manifestations, and one-quarter develop neurologic symptoms (see Fig. 82.1 ).
In tertiary syphilis, non-treponemal serologic tests are usually positive with high titers. Patients with tertiary syphilis (or any symptoms related to late syphilis) should undergo lumbar puncture and CSF examination to determine if neurosyphilis is present before therapy is initiated.
“Benign” late syphilis
The most common feature of late syphilis is gummas, which are locally destructive lesions in the skin, bones, liver, and other organs. The gummas in the skin are nodular or noduloulcerative lesions, often with an arciform pattern ( Fig. 82.11 ). They remain for weeks to months and then involute with scarring, but eventually new nodules reappear. With treatment, resolution of the lesions is prompt and complete. A solitary gumma that is located subcutaneously may become necrotic, similar to a “cold” abscess, resulting in ulceration of the skin or mucous membranes as well as destruction of underlying bones.
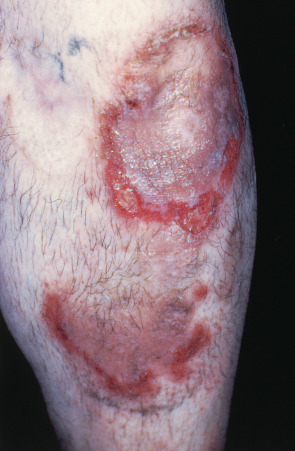
Bones are affected as frequently as the skin. Within the bone, a gumma is destructive and is typically accompanied by periostitis and osteitis. Clinical manifestations include pain, swelling, and limited range of motion. Other sites that can be affected by gummas include the tongue and oral cavity, upper respiratory tract, myocardium, and digestive as well as nervous systems.
Cardiovascular syphilis
Cardiovascular syphilis has a late onset, with a latent period of 15–30 years, and it occurs in about 8–10% of individuals with untreated infection. During the early stage of the disease, T. pallidum demonstrates a predilection for the vasa vasorum of the proximal aorta and produces transmural inflammatory lesions, which lead to endarteritis of the vessels. The organisms then remain there in a latent state for many years.
Neurosyphilis
Although classically a manifestation of tertiary syphilis, neurosyphilis can occur at any stage of the disease. Neurosyphilis is the reflection of a chronic (or sometimes acute) form of meningitis, with vascular and parenchymatous sequelae in the cerebrum and spinal cord ( Table 82.5 ) . Asymptomatic neurosyphilis is defined by the presence of abnormalities in the CSF (see below), but in the absence of any neurologic symptoms or clinical findings. While in some individuals the CSF abnormalities may resolve spontaneously or persist into advanced age, asymptomatic neurosyphilis can progress to late symptomatic neurosyphilis.
| CLASSIFICATION OF NEUROSYPHILIS |
|
The basic pathomechanism for cerebrovascular syphilis is an infarction secondary to syphilitic endarteritis; this can result in hemiparesis or hemiplegia. Parenchymatous neurosyphilis, including general paresis, dementia paralytica, and paretic neurosyphilis, is due to direct invasion of the cerebrum by treponemes. It is a rare and late form of neurosyphilis, with dementia and neurologic symptoms such as paralysis progressing, if untreated, until death occurs.
Tabes dorsalis, which affects up to a third of patients with neurosyphilis after a long latent period, has a number of typical clinical signs and symptoms, including diplopia, lightning pains (painful paresthesias, especially of the extremities), loss of vibratory and position sense, reduced reflexes in the legs, ataxia, sphincter dysfunction, visceral crisis (abdominal, rectal, and laryngeal pain), and Argyll Robertson pupils (abnormal pupillary light reflexes: loss of direct and consensual; retention of accommodation). Tabes dorsalis may burn out even without treatment.
Neurosyphilis in HIV-infected individuals
In the mid 1970s, the first cases of so-called “atypical neurosyphilis” were described. With the subsequent recognition of co-infections with syphilis and HIV, it became apparent that neurosyphilis is common in HIV-infected individuals, and patients present with manifestations of early-onset neurosyphilis.
Laboratory diagnosis of neurosyphilis
In addition to the clinical findings, the diagnosis of neurosyphilis (including acute syphilitic meningitis, meningovascular syphilis, and parenchymatous neurosyphilis) is based upon reactive blood and CSF serologies, which are almost always positive. CSF abnormalities include elevation of pressure, protein concentration, and immunoglobulin level as well as a mononuclear pleocytosis. The presence of specific antitreponemal antibodies in the CSF is mandatory, but it is not proof of the diagnosis of neurosyphilis, since IgG antibodies can diffuse into the CSF or result from contamination of the CSF by blood. Establishment of local antibody synthesis is possible by using the CSF-IgG index, obtained by dividing the CSF to serum IgG ratio by the CSF to serum albumin ratio. A result >0.7 is indicative of IgG synthesis in the brain due to local inflammation. The intrathecal T. pallidum antibody index is calculated by dividing the TPHA titer in the CSF by the CSF to serum albumin ratio multiplied by a factor of 10 3 , and an index >100 is indicative of the synthesis of treponemal-specific antibodies within the CNS. The presence of nonspecific antibodies, e.g. a positive VDRL test or RPR assay in CSF, is observed in most, but not all, cases, and a negative test does not exclude the presence of neurosyphilis.
Congenital syphilis
Congenital syphilis was initially described in 1497 and is therefore the oldest recognized congenital infection. In 1906, it was demonstrated via the Wassermann test that transmission of syphilis to the fetus required an infection in the pregnant woman.
There was an upsurge in congenital syphilis in Eastern Europe in the late 1990s, which paralleled the increased incidence of syphilis in adults and adolescents in this region. In the early 1990s, an increased frequency of congenital syphilis was also observed in some inner-city minority populations in the US . In the US, there are currently ~12 cases of congenital syphilis per 100 000 live births. The risk of mother-to-child transmission of untreated syphilis and its potential consequences are outlined in Table 82.6 .
| MOTHER-TO-CHILD TRANSMISSION OF UNTREATED SYPHILIS AND ITS CONSEQUENCES |
| Risk |
|
| Consequences of infection |
|
Early congenital syphilis
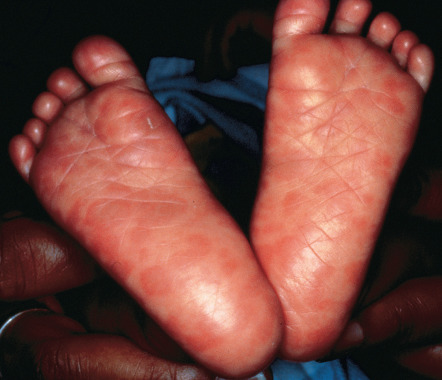
Infants generally present with symptoms during the neonatal period or within the first 3 months of life ( Fig. 82.12 ). At the latest, they develop symptoms within the first 2 years of life. Typical manifestations are marasmic syphilis (i.e. cachexia) and skin lesions similar to those of acquired secondary syphilis ( Fig. 82.13 ), except that they may be bullous (pemphigus syphiliticus) and tend to be more erosive. Additional clinical findings are “snuffles” (bloody or purulent mucinous nasal discharge), perioral and perianal fissures, lymphadenopathy, and hepatosplenomegaly. Skeletal involvement (i.e. osteochondritis) may result in pseudoparalysis of Parrot because of reduced movement of the extremities due to pain; other manifestations include anemia, thrombocytopenia, syphilitic pneumonitis (pneumonia alba), hepatitis, nephropathy, and congenital neurosyphilis.
Late congenital syphilis and stigmata
Late congenital syphilis in a child or adolescent corresponds to tertiary syphilis in an adult and is not infectious. The stigmata represent the delayed consequences of localized inflammation at the sites of treponemal infection ( Table 82.7 ). In about one-third of children, an interstitial keratitis is seen; this finding together with typical dental abnormalities (Hutchinson teeth) and neural deafness forms the Hutchinson triad.
| STIGMATA OF CONGENITAL SYPHILIS |
| Cutaneous |
|
| Dental |
|
| Skeletal |
|
| Other |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree