Introduction
Skin is the first immune defense mechanism and functions as a barrier against microorganisms. Infections are a significant problem once open wounds compromise this barrier. According to the US National Burn Repository, the four leading causes of burn morbidity are pneumonia, cellulitis, urinary tract infections, and burn wound infections. Infections are a primary factor contributing to mortality, accounting for 51% of deaths in burn patients, as discussed in both Chapters 24 and 26 , on multisystem organ failure and critical care, respectively.
A cutaneous burn is initially sterile as commensal skin flora are killed with the skin in the thermal event. Unfortunately, the burn wound provides optimal bacterial growth conditions due to a reduced blood supply and a nutrient-rich environment, leading to rapid wound colonization.
Interestingly, in a recent porcine study of standard burn wounds, there was no difference in bacterial counts between wounds treated with negative-pressure wound therapy, silver sulfadiazine, or normal saline. These data further demonstrate the need for early wound excision and coverage.
The 2007 American Burn Association (ABA) Consensus Conference defined wound colonization as follows: (1) low concentrations of bacteria on the wound surface, (2) absence of invasive infection, and (3) less than 10 5 organisms per gram of tissue. In a superficial burn, skin flora can survive in hair follicles and sebaceous glands in the same manner as keratinocytes to repopulate a physiologic microbiome. However, burns are customarily colonized by pathogens from the environment, the patient’s gut, or the nasooropharyngeal tract. Ladhani et al. have demonstrated poor specificity for identification of sepsis and septic shock in burn patients. They conclude the best approach is prevention (including wound care, early excision and grafting, antibiotic stewardship, nutrition) and a multidisciplinary support. Meze-Escobar demonstrated that the sepsis definitions in the Third International Consensus Definitions for Sepsis and Septic Shock have better sensitivity and specificity than the 2007 ABA consensus definitions. Ultimately, widely accepted definitions of burn wound infection and sepsis remain elusive, and further work is need to standardize research treatment and discussions. Indeed, Lee et al. found in a systemic review of definitions of burn wound infection 32 different definitions in 29 reviews, making data comparison impossible. This lack of effective definitions and diagnostic criteria to differentiate anticipated systemic inflammatory responses and sepsis continues to beset the field.
A race thus exists between the patient and the pathogen to dominate the wound surface. In the log phase growth, bacteria double two to three times per hour; consequently, a single bacterium can become 10 million in 1 day, far faster than any human cell can multiply. Therefore colonization can quickly become an infection capable of converting partial-thickness into full-thickness burns by causing vessel thrombosis and necrosis in viable tissues in the wound penumbra. Gram-positive bacteria tend to colonize an affected area first, with subsequent colonization by gram-negative bacteria. Delayed treatment risks colonization by extended-spectrum pathogens, bloodstream invasion, and the development of sepsis, all of which increase the likelihood of death.
The model of burn care advocated throughout this text is to rig the race for wound dominance in favor of the patient. Early wound excision eliminates the devitalized tissue that is the main reservoir for pathogen nourishment and habitat. Prompt autografting reestablishes skin barrier function and denies pathogens access to the host. Topical antimicrobials suppress bacterial growth and colony counts while allowing host fibroblasts and keratinocytes to proliferate and cover the wound. Assiduous washing technique allows 2-log reduction in colony counts, breaks down and removes biofilms, and purges the devitalized tissue that pathogens thrive upon. Systemic antimicrobials kill and suppress the invading pathogens accessing perfused areas of tissue. Quantitative wound culturing (QWC) enables effective diagnosis and directed application of the most efficacious antimicrobial with the lowest toxicity. Coordinated critical care, therapy, and nutrition ensure that the wounds have a sufficient supply of nutrients and immune cell–laden blood to clear pathogens, expand skin grafts, and reepithelialize wounds.
Prevention of infection
Prevention is the optimal way to minimize infection. Pathogens can be carried into or transmitted around the unit by staff, by visitors, or on equipment. Patients should have single rooms separated from other rooms by a door. Positive pressure in the rooms further aids in minimizing bacterial contamination. Patient rooms should undergo a daily cleaning in addition to deep cleaning upon patient discharge. This terminal cleaning should include washing the walls and ceiling and should be done 72 hours prior to admitting another patient to avoid the transfer of more virulent strains. Another important prevention measure is the use of contact precautions with all patients, including gowns and gloves. These items should be donned before entering and doffed prior to exiting the room, regardless of the bacteriologic status of the particular patient. Routine hand hygiene before and after patient interaction is also mandatory to prevent infection. Dressing materials, supplies, devices, and equipment must not be shared between patients. Bathing, showering, tubbing, mobile diagnostic equipment, and operating facilities should be decontaminated between each patient use. Furthermore, fomites, such as ties, rings, watches, and cell phones, should be prohibited because these are possible pathogen vectors. , Water and air filters ought to screen particles down to 0.2 and 0.3 μm, respectively, be changed monthly, and be cultured routinely as part of infection control monitoring. By maintaining these strict measures, transmission of infectious organisms between patients can be limited.
Diagnosis of burn wound infection
Diagnosis of burn infection in burn wounds can be complicated. The typical cytokine and immune cascades that create the typical presentations of infection known to all doctors are often initiated in the burn patient via the elaboration of damage- and pathogen-associated molecular patterns from burn wounds. This complicates diagnoses of infection and sepsis in the burn patient where clinical signs, such as elevated temperature or tachycardia, are normal components of burn pathophysiology (see Chapter 18 for more on hypermetabolism).
Early diagnosis of bloodstream infection in burn patients has been an issue of continuing research. Sadeq et al. performed a retrospective review of 100 pediatric burns over 20% total body surface area (TBSA) comparing events with positive blood cultures to negative cultures. They found a predictive value of bloodstream infection with spikes in heart rate and temperature in the preceding 24 hours with a sensitivity of only 57% and specificity of 44%. They also found that a combination of scored criteria can increase to 93% sensitivity and 81% specificity. In using such criteria, antibiotic stewardship may be improved, though it is as yet unclear if there would be an improvement in clinical outcomes.
Wound culturing remains a critical tool to guide the treatment of burn wounds and to determine what bacteria are colonizing therein. In patients with major burns, the wound usually becomes colonized 5 to 7 days postinjury. Because most initial infections in burn patients derive from endogenous bacteria flora, it is good clinical practice to perform initial wound cultures upon patient admission. This screening should include swabs of both sides of the groin and axilla as well as of the nose and throat. In addition, at each burn excision and whenever suspicion of invasive infection warrants, quantitative tissue cultures may be helpful.
Wound appearance and odor changes can help to diagnose wound colonization vs. wound infection ( Fig. 10.1 ). Kwei et al. described three main methods that exist for culturing a wound: qualitative (presence or absence of growth), semiquantitative (grading of the bacterial presence as scanty, few, moderate, or numerous), and quantitative (an absolute quantity is determined). , Swabs are useful but limited because they cannot distinguish between infection and colonization and are only accurate for the region sampled. These problems are mitigated by performing multiple tissue biopsies, which can reveal significant quantitative differences in different areas. Although quantitative cultures are more expensive, bacterial counts have been shown to correlate with histologic evidence of wound infection in approximately 80% of cases after biopsies are taken or after sterilization of the surface with antimicrobials. , Histologic examinations can be used to confirm invasive bacterial infection when counts exceed 10 organisms per gram of tissue. If histologic evidence of invasion is present, systemic antibiotics should be administered and the wound excised. , ,
Burn wound colonization. Flame burn to the left medial knee depicted postburn day 7. The eschar has degraded but is still present; however, the surrounding tissue is not cellulitic.
Studies by Robson and Krizek demonstrated that if burn wound colony counts from biopsies or after cleaning of the surface of the wound are greater than 10 5 /g tissue, the graft survival rate is only 19%, whereas colony counts of less than 10 5 /g tissue are associated with a 94% chance of graft survival. Sensitivities to available antibiotics, both systemic and topical, should instruct therapy. In the setting of resistant organisms, antibiotic synergy testing is advised. Thus the ultimate diagnosis of burn wound infection and the guidance of antimicrobials are directed by culture data.
In practice, the utility of quantitative culturing is limited by the time required for culture growth itself. Williams et al. demonstrated that improved outcomes for septic pediatric burn patients were best obtained by prompt antimicrobial drug administration and source control (as well as early recognition and resuscitation). In clinical practice, the operating surgeon must decide on wound viability and colonization based on experience and clinical judgment because it is impractical to delay source control for confirmatory data. As such, a practical approach is to excisionally treat burn wounds, as described in Chapter 10, while using semiquantitative cultures and patient response to guide postoperative antibiotic management.
Physical examination of the patient also provides valuable insight into the infectious nature of the burn wound. Burn wound erythema is a physiologic phenomenon produced sterilely by the liberation of inflammatory mediators from tissues surrounding the burn area and must not be confused with cellulitis. Normally, this erythema presents within 2 to 3 days of the burn injury and resolves by 1 week postburn ( Fig. 10.2 ). The best differential diagnosis comes from clinical palpation: erythema lacks significant induration or tenderness compared to an infectious process such as cellulitis.
Burn wound erythema. A hot oil scald burn on the lateral thigh is depicted with (A) surrounding erythema noted on postburn day 2. (B) Resolution of redness and healing on postburn day 12.
Cellulitis is a noninvasive infection of the tissues surrounding the burn wound. Cellulitis can be caused by a variety of pathogens. This infection is characterized by edema, hyperesthesia, erythema, induration, and tenderness detectable upon examination ( Fig. 10.3 ). The color of the wound contour and the odor from the wound may also raise suspicion of cellulitis. Furthermore, it can have a lymphangitic component. Particular attention should be given to patients who are elderly or diabetic because of the ease and speed with which infections progress in these populations. Cellulitic burn wounds benefit from systemic antimicrobials to cover likely causative agents in addition to standard burn treatments, such as topical antimicrobials or surgical excision and grafting. Progression of cellulitis despite antibiotics must always trigger suspicions of resistant organisms, such as methicillin-resistant Staphylococcus aureus (MRSA), and these organisms should be covered empirically.
Burn wound cellulitis. Cultures demonstrated Staphylococcus aureus . Clinical findings include increased pain, local inflammatory signs, and fever. Treatment includes systemic and topical antibiotics, excision, and autografting.
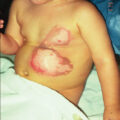
Graft ghosting (impetigo) is a wound infection that can cause late graft loss ( Fig. 10.4 ). This phenomenon can occur after spontaneous closure of a partial-thickness wound, following loss of previously adherent graft sites, and in skin donor sites. Characterized by multiple small abscesses, graft ghosting can lead to the complete destruction of the healed wound. The culprit typically responsible for this condition is S. aureus , particularly MRSA. The diagnosis is essentially clinical and can be confirmed by culturing. Treatment requires regular dressing changes, debridement of abscesses, local disinfection, and application of topical antimicrobials, such as mupirocin.
Burn wound impetigo. Right hand in a 13-year-old boy with 85% total body surface area burn, third degree to the hands shown (A) 4 months postburn at hospital discharge. (B) Three weeks later, the patient presented with reopening of the previously taken skin grafts to the hand and clinical impetigo. Cultures were positive for methicillin-resistant Staphylococcus aureus , and the patient was treated with vancomycin, mupirocin, and tub therapy.
Toxic shock syndrome (TSS) is a complication of severe soft tissue infection (SSTI), which occurs predominately in small burns. This syndrome results from colonization with TSS toxin-1–producing S. aureus . This disease is primarily seen in young children with burns covering less than 10% of the TBSA and that would otherwise be expected to heal without problems. The incidence is approximately 2.6% with a mean age of 2 years. TSS is clinically characterized by a prodromal period lasting 1 to 2 days with pyrexia, diarrhea, vomiting, and malaise. Although a rash is often present, at this stage the burn appears clean ( Fig. 10.5 ). Shock subsequently develops in untreated cases, but determining the cause of shock to be TSS can be complicated in this early phase of the patient’s presentation when a litany of potential and more common etiologies of shock exists. Once shock has developed, mortality can be as high as 50%. Awareness and aggressive action are the principal safeguards against the development and progression of TSS. A possible diagnosis of TSS should be considered in small burns when the patient is unexpectedly in shock. Because MRSA has emerged as the most commonly identified cause of SSTI, initiation of empiric anti-MRSA antimicrobials is warranted in all cases of suspected TSS. ,
Toxic shock syndrome (TSS) rash. Cutaneous rash commonly seen in patients with TSS. This rash does not universally appear in cases of TSS. Patients with small burns developing shock should be evaluated and treated for TSS with excision, grafting, and vancomycin. (A) A 13-year-old girl presenting TSS rash following a 10% total body surface area burn. (B) The typical macular erythroderma lesions. (C) A hematoxylin and eosin (H&E) 4× magnification micrograph of a TSS rash lesion showing an epidermal blister. (D) Further H&E 40× magnification micrograph with low inflammation.
(From Omar P. Sangüeza, MD; Professor and Director of Dermatopathology, Wake Forest University School of Medicine, North Carolina.)
Invasive wound infections manifest clinically with wound color changes, exudate, and odor. Within a short time, partial-thickness burns progress to full-thickness necrosis and begin expanding into unburned tissues. The 2007 ABA Consensus Conference defined invasive infections as follows: “the presence of pathogens in a burn wound at concentrations sufficient in conjunction with depth, surface area involved, and age of patient, to cause suppurative separation of eschar or graft loss, invasion of adjacent unburned tissue or cause the systemic response of sepsis syndrome.” Goal-standard diagnosis is made with histologic examination; however, clinical exam and quantitative cultures usually suffice ( Fig. 10.6 ). It should be noted that sepsis does not always develop during invasive infections ( Fig. 10.7 ). Treatment must be immediate and include aggressive surgical intervention augmented by the administration of systemic and topical antimicrobials. If no culture results are initially available, then a broad-spectrum empirical therapy against fungi and drug-resistant gram-positive and-negative organisms should be initiated until culture data become available. As discussed earlier, these definitions have been demonstrated to be somewhat inadequate in multiple series, but a standard replacement has not yet gained broad acceptance. Surgical extirpation must be aggressive and encompass excision of all necrotic and infected tissue, including fascia and muscle when warranted. Definitive wound coverage is not always indicated in this extirpative operation as dressing changes and hydrotherapy may be needed to further decrease heavy bacterial loads. In cases where tissue has already been excised or there is a life-threatening infection, limb amputation may be indicated. Topical antimicrobials and assiduous washing technique are indicated after extirpation to suppress microbial growth; however, the optimal methodology is to prevent infection, with swift action being taken should an infection arise.
Gram-negative bacilli invading deep viable tissue. Histopathologic confirmation is the gold standard test to diagnose burn wound infection by bacteria in viable tissue. Shown is a typical hematoxylin and eosin–stained section at 1000× magnification.
Invasive burn wound infection. This patient has 75% total body surface area full-thickness burns with wound sepsis due to Enterococcus faecalis and Enterobacter cloacae . Prompt excision, homografting, hemodynamic support, and systemic antibiotics controlled the infection. This patient was subsequently autografted and survived.
Sepsis and septic shock are complicated diagnoses in burn patients because large burns create a systemic inflammatory response syndrome (SIRS) and hypermetabolism of their own (see Chapter 7 on the etiology of shock). This hypermetabolism is a natural part of the body’s compensatory mechanism to burn injury and can last for up to 1 year following the insult, making it difficult to fit shock in burn patients into the definitions of sepsis and septic shock established by the Society of Critical Care Medicine. Patients with high concentrations of bacteria in the burn wounds in conjunction with delayed admission to a burn center or delayed removal of burned tissues are at the greatest risk of developing sepsis. Rapid and complete closure of deep burns is the best defense against this condition. An additional factor influencing the development of sepsis and increasing mortality during hospitalization in patients with equal burn sizes is decreased lean body mass. The identification of parameters associated with sepsis in burn patients is extremely important and ongoing. The 2007 ABA Consensus Conference provided criteria ( Box 10.1 ). These criteria are useful markers and indicators for sepsis but are by no means gospel. A skilled physician must take these factors into account along with changes in the patient’s clinical condition over time to make a presumptive diagnosis of sepsis. Aggressive treatment should be initiated and deescalated based on definitive diagnosis and patient response.
Box 10.1
From Greenhalgh DG, Saffle JR, Holmes JHT, et al. American Burn Association consensus conference to define sepsis and infection in burns. J Burn Care Res . 2007;28(6):776-790.
The 2007 American Burn Association Consensus Conference Definition of Sepsis
“Sepsis is a change in the burn patient that triggers the concern for infection. It is a presumptive diagnosis where antibiotics are usually started and a search for a cause of infection should be initiated. While there is need for clinical interpretation, the diagnosis needs to be tied to the discovery of an infection [defined below]. The definition is age-dependent with adjustments necessary for children.”
The trigger includes at least three of the following for sepsis:
-
I.
Temperature >39°C and <36.5°C
-
II.
Progressive tachycardia
-
A.
Adults >110 bpm
-
B.
Children >2 standard deviation (SD) above age-specific norms (85% age-adjusted max heart rate)
-
A.
-
III.
Progressive tachypnea
-
A.
Adults >25 bpm not ventilated
-
i.
Minute ventilation >12 L/min ventilated
-
i.
-
B.
Children >2 SD above age-specific norms (85% age-adjusted max respiratory rate)
-
A.
-
IV.
Thrombocytopenia (will not apply until 3 days after initial resuscitation)
-
A.
Adults <100,000/mcl
-
B.
Children <2 SD below age-specific norms
-
A.
-
V.
Hyperglycemia (in the absence of preexisting diabetes mellitus)
-
A.
Untreated plasma glucose >200 mg/dL or equivalent mM/L
-
B.
Insulin resistance; examples include
-
i.
>7 units of insulin/hour intravenous drip (adults)
-
ii.
Resistance to insulin (>25% increase in insulin requirements over 24 hours)
-
i.
-
A.
-
VI.
Inability to continue enteral feedings >24 hours
-
A.
Abdominal distension
-
B.
Enteral feeding intolerance (residual >150 mL/hour in children or 2cc residual per 24 hours)
-
C.
Uncontrollable diarrhea (>2500 mL/day for adults or >400 mL/day in children)
-
In addition, it is required that a documented infection (defined below) is identified
-
i.
Cultures positive for infection, or
-
ii.
Pathologic tissue source identified, or
-
iii.
Clinical response to antimicrobials
-
i.
-
-
A.
There are no objective laboratory tests to differentiate between inflammatory versus septic shock. To this day, white blood cell count is the most commonly monitored value to provide this insight, though it has low sensitivity and specificity. Procalcitonin (PCT) and C-reactive protein (CRP) are increasingly accepted markers in clinical use as predictors of sepsis in postoperative burn patients. Hassan performed a systematic review of 15 manuscripts finding that both CRP and PCT increase then gradually decrease after burn injury. In the setting of burn wound infection, both continue to rise, and PCT was found to be more reliable than CRP. In another study, it was demonstrated that CRP to albumin ratio at admission was an independent risk factor for sepsis and prognosis in severe burns. Recent multiomic analysis by Huang et al. evaluating genomic, proteomic, and transcriptomic responses in 60 severe burn patients has identified seven novel biomarkers as early warning signs in severe burn–associated sepsis. The protein S100A8 has both warning and therapeutic effects. Further studies continue to develop biomarkers and objective diagnostic panels, but there remains no accepted standard.
Treatment of burn wound infections
Treatment of burn wound infections is multimodal. Surgical debridement and assiduous washing technique decrease the bacterial and nutritive burden. Aggressive grafting denies the pathogens wound surface area to colonize and infect. Topical antimicrobial compounds reduce pathogen burden in the wound and periwound areas while allowing skin grafts to proliferate and cover the wound. Systemic antimicrobials are administered to treat invasive pathogens and prevent or reduce the systemic spread of infection.
Topical antimicrobial compounds
Topical antimicrobial compounds have significantly reduced burn mortality. , However, no single agent is entirely effective, and each possesses its own spectrum, advantages, and disadvantages (e.g., some retard wound healing, others have systemic metabolic effects on the patient). Recent studies have demonstrated that some agents used in the past are now ineffective in inhibiting bacterial growth. , Wounds may be dressed with any topical agent when quantitative culture counts persist at less than 10 2 /g tissue; however, higher colony counts warrant culture-directed topical antimicrobial selection. Topical antimicrobial agents fall into five major classes (soaps, oxidative halides, acids, heavy metals, antibiotics), each possessing different antimicrobial spectra, duration of action, penetration, and toxicities ( Box 10.2 ).
Box 10.2
Topical Antimicrobial Agents
| Class | Agents | Antimicrobial Spectrum | Graft Toxicity | Systemic Toxicity |
|---|---|---|---|---|
| Soaps | Johnson’s Baby Shampoo | Broad spectrum + biofilm | Low | None |
| Oxidative halides | Full-strength Dakin solution (0.5% NaOCl) | Broad spectrum + biofilm | High | Hyponatremia |
| 1/20 buffered Dakin solution (0.025% NaOCl) | Broad spectrum + biofilm | Low | None | |
| Oxychlorosene (Clorpactin) | Broad spectrum + biofilm | Low | None | |
| Hypochlorous acid | Broad spectrum + biofilm | Unknown | Unknown | |
| Povidone-iodine (Betadine) | Broad spectrum | High | High | |
| Acids | Acetic acid 0.5% | Bacteriostatic | Low | None |
| Acetic acid 2% | Bacteriostatic | Moderate | Moderate | |
| Acetic acid 3% | Bacteriostatic | High | High | |
| Heavy metals | Silver nitrate 0.5% | Broad spectrum | None | Electrolyte imbalance |
| Silver sulfadiazine (Silvadene) | Broad spectrum | None | Low | |
| Silver-releasing dressings | Broad spectrum | None | Low | |
| Xeroform (bismuth tribromophenate) | Limited bacteriostatic | Low | None | |
| BIPPS (bismuth subnitrate and iodoform) | Bacteriostatic | High | High if >1% total body surface area | |
| Antibiotics | Mafenide acetate (Sulfamylon) | Broad spectrum | Low | Metabolic acidosis |
| Gentamicin sulfate (Gentamicin) | Broad spectrum | Low | Low | |
| Bacitracin/polymyxin (Polysporin) | Broad spectrum | Nontoxic | Low | |
| Nitrofurazone (Furacin) | Broad spectrum, no Pseudomonas | Low | Low | |
| Mupirocin (Bactroban) | Broad spectrum, no Pseudomonas | Moderate | Moderate | |
| Nystatin 100,000 U/g (Mycostatin) | Weak antifungal | Low | Low | |
| Nystatin 6 million U/g | Strong antifungal | Low | Low |
Soaps
Soaps are the first form of topical antimicrobial and are used during washing. They are effective in disrupting biofilms and washing pathogens from the patient. Biofilms are coherent clusters of bacterial cells embedded in a biopolymer matrix that, compared with planktonic cells, resist host defenses and show increased tolerance to topically (antiseptics) and systemically (antibiotics) administered antimicrobials, thus creating the perfect environment for bacterial growth. Biofilms form in burn wounds from burn wound pathogens. Biofilm bacteria are extremely difficult to remove, requiring surgical or sharp instruments and/or mechanical wound debridement and washing with soap and water. According to recent recommendations, burn wounds not associated with clinical signs or symptoms of infection yet suspected of having biofilm should be treated with debridement, antimicrobial dressings, antiseptic soaks, and thorough cleansing with antiseptic products to prevent bacterial growth. Findings from Kennedy et al. support the significant role played by biofilms in burn wound sepsis and the importance of early excision and closure of the wound. Herndon et al. demonstrated that washing burn wounds with soap and water using assiduous washing techniques can cause a 2-log reduction in bacterial counts over 48 hours, improving autograft take by infected wounds.
Oxidative halides
Many oxidative halides are used as topical antimicrobials. Classically, sodium hypochlorite (Dakin solution) has been used because of its broad bactericidal range and effective dissolution of biofilms. Commercially available Clorox bleach is 5.25% NaOCl; the original Carrel-Dakin solution used in World War I was diluted with water to 0.5% and is described as full-strength Dakin solution. Studies have demonstrated full strength to be tissue toxic, as are the pharmacy-available half- and quarter-strength Dakin solutions, though they are effective antimicrobials. Investigations by Heggers and others established the efficacy of NaOCl at a concentration of 0.025%, or 1/20 Dakin. , , Buffered 1/20 Dakin was formulated to mimic normal human physiologic parameters, with the added benefits that it is a broad-spectrum antiseptic, yet nontoxic to fibroblasts and a noninhibitor to wound healing. It is bactericidal against Pseudomonas aeruginosa , S. aureus , methicillin-resistant staphylococci, enterococci, and other gram-negative and-positive organisms, and it may be used separately or in combination with other agents. Subsequently, Carrel and Dakins developed oxychlorosene (Clorpactin) to allow longer tissue half-life, neutral pH, and lower tissue toxicity. It is used intravesicularly and intrarenally to control hemorrhage in genitourinary cancer cases and has been recently resurging as a topical antimicrobial. It has been demonstrated to be nontoxic to skin grafts when tested as a wound irrigant. Currently, products are coming to market using hypochlorous acid as an active oxidizing agent in pH-neutral, isotonic formulations. , While of interest as topical antimicrobials for burn wounds, they lack the century-long experience of sodium hypochlorite solutions and so require further research. These agents are increasingly ubiquitous in burn care; however, concerns persist regarding effectiveness in the setting of dilution in draining wounds.
Povidone-iodine (Betadine) is another halide used as a topical antimicrobial; it is available as a liquid or an ointment in varying concentrations. It has a broad spectrum of activity, covering gram-positive and-negative bacteria, yeast, and fungi. Quantitative bacteriologic assessments imply that iodine is most efficacious when administered every 6 hours. Topical application of this agent can be painful. The iodine component of this topical agent may be absorbed more extensively in burn wounds, resulting in iodine toxicity, renal failure, acidosis, and dermatitis (in intact skin). Furthermore, povidone-iodine is cytotoxic to fibroblasts and keratinocytes; however, it remains a highly effective disinfectant when used on intact skin. ,
Acids
Acetic acid, also called ethanoic acid or vinegar, is a colorless topical agent used as a disinfectant for skin and soft tissue infections. It is effective against gram-negative bacteria, especially P. aeruginosa. Its clinical antibacterial efficacy requires a minimum concentration of 0.5%. Philips et al. reported the use of acetic acid as a topical agent for the treatment of superficial wounds infected by Pseudomonas ; later Sloss et al. investigated topical use of acetic acid at concentrations between 0.5% and 5%. Sloss showed all strains of Pseudomonas exhibited a minimum inhibitory concentration (MIC) of 2% in vitro, yet other studies determined a concentration of 3% acetic acid to have bacteriostatic activity, including against multiple antibiotic-resistant strains of Pseudomonas. , The results of in vitro studies indicate that acetic acid is toxic to fibroblasts and substantially decreases cell viability, effects that increase with the concentration. Although these results are not considered decisive for the treatment of burn wounds, a surgeon should always consider the eventual cytotoxic interaction, especially on fresh skin grafts, while using acetic acid.
Heavy metals
Silver ions are a common topical heavy metal antimicrobial. Silver is delivered as solutions, creams, or bound to dressing materials. The silver ions bind to proteins and enzymes, damaging those systems, and to DNA, resulting in an antimicrobial effect via a heavy metal oxidative pathway. In debrided wounds, a 0.5% silver nitrate (AgNO 3 ) solution is a potent disinfectant. It does not injure regenerating epithelium in the wound and is bacteriostatic against S. aureus , Escherichia coli , and P. aeruginosa . AgNO 3 has limited wound penetration because the silver binds rapidly to the surface proteins. Its hypotonic nature can cause osmolar dilution, resulting in hyponatremia and hypochloremia, so serum electrolytes must be monitored. AgNO 3 turns black when exposed to light or on contact with tissues or chlorine-containing compounds, but this is nontoxic. It can be combined with miconazole powder to yield an aqueous solution of 0.5% silver nitrate and 2% miconazole for greater efficacy in preventing bacterial and fungal overgrowth in burn wounds. Klebsiella spp., Providencia spp., and other Enterobacteriaceae are less susceptible to 0.5% AgNO 3 than other bacteria. Rarely, the combination of 0.5% AgNO 3 solution with Enterobacter cloacae or other nitrate-positive organisms can cause methemoglobinemia by converting nitrate to nitrite in the body.
Silver sulfadiazine (Silvadene, Thermazene, Flamazine, SSD), a 1% water-soluble cream, is a combination of sulfadiazine and silver with antimicrobial efficacy lasting up to 24 hours. While most effective against P. aeruginosa and the enterics, silver sulfadiazine has great utility against some yeasts, such as Candida albicans. However, recent reports of P. aeruginosa resistance and inadequacy against some strains of Klebsiella have emerged. More frequent dressing changes are required if a creamy exudate develops. Although this topical agent is facile in use and reduces pain, it retards wound healing. Unlike mafenide acetate, tissue penetration of silver sulfadiazine is limited to the surface epidermal layer, and it is not associated with acid-base disturbances or pulmonary fluid overload. It can be used separately or in combination with other antibacterials. An adverse drug reaction may be a reversible granulocyte reduction due to silver toxicity, although this is controversial and transient. , Over multiple applications, a pseudoeschar of silver sulfadiazine cream builds up on the wound, making assessment of the wound depth difficult because it then bears a strong resemblance to burn eschar, thus limiting the utility of this topical agent.
Over the past decades, there has been a proliferation of silver-containing dressings formulated to remain in place over a prolonged period. On superficial burns, these dressings are utilized as functional skin substitutes to permit reepithelialization in a bacterially suppressed, moist environment. Many centers use silver-containing dressings rather than applying solutions of topical antimicrobials, such as silver nitrate, because they are easier to manage, have more consistent antimicrobial levels, and are generally less messy. The antimicrobial actions of the dressings derive from the silver ions, so their spectrum is thus defined. Each product’s dressing substrate and particular silver formulation provide different dressing characteristics; however, the release and efficacy of silver against bacteria in wounds have not been largely studied or reported. Furthermore, there are to date no large-scale studies defining the supremacy of any particular product, so preference of the practitioner and cost typically define use. It is important to remember that no dressing, no matter how well marketed, replaces the tenets of burn surgery: aggressive debridement of devitalized tissue, coverage of wounds with skin, assiduous wound cleansing to remove pathogens and contaminants, and physical examination.
Bismuth is another heavy metal commonly used as a topical antimicrobial. It is bacteriostatic against enterics, but not cytotoxic to dermal fibroblasts and does not inhibit wound healing. It is often used on the commercially available dressing Xeroform, a gauze impregnated with bismuth tribromophenate and petroleum jelly. Bismuth is also delivered in conjunction with iodine in a compounded paste, bismuth iodoform paraffin paste (BIPP: 1 part bismuth subnitrate, 1 part liquid paraffin, and 2 parts iodoform). BIPP has been used effectively on thousands of patients treated inhospital for more than 50 years. Coated liberally on cotton gauze, this paste is used to dress small, debrided areas of exposed bone and tendons. , BIPP should not be applied to areas greater than 1% TBSA, and the course of treatment should be as brief as possible to limit the risk of bismuth toxicity. , In our experience, this compound prevents the development of infection and promotes the prodigious formation of granulation tissue, thereby permitting split-thickness skin grafting over surfaces conventionally described as ungraftable.
Antibiotics
Antibiotic-based topical antimicrobials define the fifth major class, and their mechanism of action is determined by the specific biochemistry characteristic of each particular agent. Mafenide acetate (Sulfamylon), available both as an 8.5% water-soluble cream and a 5% aqueous solution, is among the most commonly used topical agents. While broad spectrum, mafenide is particularly effective against all strains of Pseudomonas and Clostridium. The cream is applied twice daily and has the advantage that it does not require a dressing to adhere to wounds. Studies have reported that the use of 5% mafenide acetate solution in patients with major burns results in a 33% reduction in death. When used as a solution, an eight-ply dressing should be resaturated with fresh solution every 8 hours to remain above MIC. Mafenide has excellent tissue penetration, including eschar. This penetration makes it the topical agent of choice for deep ear burns because it effectively prevents invasive chondritis. It can cause pain on application and, like other sulfa drugs, can lead to allergic reactions. Since mafenide can inhibit carbonic anhydrase, metabolic acidosis can develop. Furthermore, protracted use may lead to the growth of C. albicans. While it can be used with other antimicrobials, mafenide acetate retards wound healing and reduces the breaking strength of healed wounds.
Gentamicin sulfate (Gentamicin) is an aminoglycoside available as a 0.1% water-soluble cream or solution. It has broad-spectrum bactericidal activity against aerobes and is often deployed against P. aeruginosa . However, resistance can develop, and sensitivities should be monitored.
Bacitracin/polymyxin (Polysporin) ointment is commonly used to prevent mechanical shear and suppress bacterial growth on newly grafted tissue. Both drugs are cell wall lytic agents, and polymyxin is a topical analogue of colistin (see Systemic Antimicrobials in Burn Patients, later). Drug concentrations available in the ointment do not treat infection. However, many surgeons rely on this topical agent for skin graft coverage as it is nontoxic and maintains the moist wound environment needed for epithelial growth. Effectiveness in contaminated or infected burns postexcision can be enhanced by use in combination with other agents, such as silver nitrate or mafenide. Prolonged use is associated with hypersensitivity development.
Nitrofurazone (Furacin), available as an ointment, solution, or cream, has been proved effective in the treatment of methicillin-resistant staphylococci. Furthermore, nitrofurantoin was demonstrated to be 75% effective against gram-negative bacterial isolates other than P. aeruginosa , whereas bacitracin/polymyxin was only 21% effective.
Mupirocin (Bactroban, pseudomonic acid A) is an antibiotic derived from the P. fluorescens capsule and inhibits isoleucyl tRNA synthetase and thus bacterial protein synthesis. It is the topical treatment of choice for MRSA infections, gram-positive microbes, and intranasal carriage. Mupirocin inhibits wound healing by a half-life of 2 days compared to controls, but the breaking strength of the wound is significantly enhanced. Due to rapid development of resistance, mupirocin should not be used for longer than 10 days.
Nystatin (Mycostatin, Nilstat) is an antifungal antibiotic produced by Streptomyces noursei . Nystatin is the highly potent topical equivalent of amphotericin-B; both exert antifungal activity by binding to ergosterol and lysing fungal cell membranes. Low-dose applications of 100,000 U/g as creams, lotions, or ointments are used as prophylaxis against fungal growth. Treatment with pure nystatin powder at a concentration of 6 million U/g on burn wounds has proved effective in eradicating invasive fungal infections. This novel application is not only effective superficially but also eradicates invasive clusters of fungi in deep wound tissues, as documented by pathologic examination. The application of the powder is easy and does not produce pain or discomfort, or impair wound healing. All previously autografted areas heal uneventfully. Liquid nystatin (swish and swallow) is used prophylactically to prevent the oral or perineal overgrowth of yeast and fungi in patients receiving multiple systemic antibiotics.
Systemic antimicrobials in burn patients
Long maligned, routine prophylactic use of antibiotics is well indicated for select burn patients. Whereas prophylactic antibiotics may be warranted for patients arriving from endemic areas, with penetrating traumas, open fractures, or from highly contaminated scenes, no prophylactic antibiotic treatments are typically initiated at admission or for routine perioperative prophylaxis. This is principally to avoid creation of antibiotic resistances with more difficult subsequent treatment. Additionally, thermal wounds are normally aseptic in the first hours following injury as the burn sterilizes the wound surfaces. ,
Empiric antibiotics are indicated in the setting of clinically suspected invasive wound infections, suspected sepsis, or septic shock. These antibiotics should broadly cover all likely infecting organisms and be instituted as a component of multimodal critical and surgical care of the burned patient. Clinical response should be followed to ensure adequate treatment, and antibiotics deescalated as soon as possible based on culture findings. After time and culture data allow definitive diagnosis of infection, culture-directed treatment antibiotics should be continued for a finite treatment course until cultures return negative or the wounds close.
Greatly altered in burn patients, pharmacokinetics and pharmacodynamics require regular evaluation by a skilled clinical pharmacologist to ensure safe and effective drug dosing. During the resuscitative phase of burn injury, which occurs within the first 48-hours posttrauma, burn shock can decrease blood flow to organs and tissues. Drug treatments during this phase result in a slower distribution rate, slower renal and hepatic elimination, and delayed absorption of enteral, subcutaneous, and intramuscular drugs. After 24 to 48 hours, the patient enters the hypermetabolic phase (see Chapters 23 and 26 for extensive discussion on hypermetabolic response, endocrine function, and critical care). During this period, burn patients exhibit increased blood flow to organs and tissues, an increased internal core temperature, and hypoproteinemia and edema formation. Intravenous (IV) drugs have a shorter half-life due to the enhanced glomerular filtration rate and elimination of renally excreted drugs. Antibiotic treatments in these patients must be administered at higher doses and/or frequency. Time-dependent, renally excreted antibiotics, such as vancomycin, must be carefully monitored to ensure they exceed the MIC of the bacteria. Oral drugs will also exhibit greater absorption from the gastrointestinal tract and a faster onset of action. The hypermetabolic phase causes hypoalbuminemia and raises the levels of acute-phase proteins. Albumin binds to acidic and neutral drugs, such as aminoglycosides, vancomycin, aztreonam, and cefotetan; consequently, in hypoalbuminemia, more free drugs are circulating, so a lower dose will be required for therapeutic effect. Conversely, acute-phase proteins bind tightly to basic drugs, such as penicillins and cephalosporins, resulting in less free drug and necessitating higher drug dosages to produce a therapeutic effect. The hepatic response occurring during the hypermetabolic phase will present as a decrease in phase 1 metabolism, such as oxidation, reduction, or hydroxylation of a drug by the cytochrome P450 system, affecting many antibiotics, such as the quinolones and the macrolides. The decreased activity of these hepatic drug–metabolizing enzymes, as well as their decreased hepatic clearance and prolonged half-life, may produce systemic toxicity. However, phase 2 metabolism in the liver, such as conjugation reactions between the drug and the endogenous substrate, will not be impaired. In light of the myriad of changes, care in the modern burn unit is greatly enhanced by the active participation of a pharmacist familiar with systematic drug-level monitoring in burn patients.
Selection of systemic antibiotics is based on the likely etiologic organism, local antibiogram, and systemic toxicity. In the setting of an outpatient with cellulitis around a burn wound, empiric gram-positive coverage to include MRSA is indicated. We routinely cover these outpatients with rifampin, a bactericidal antibiotic inhibiting RNA synthesis by binding to the β-subunit of the DNA-dependent RNA polymerase and blocking RNA transcription. Due to its high resistance pattern when used alone, rifampin must be used in conjunction with other antiinfectives, such as sulfamethoxazole-trimethoprim (Bactrim) or levofloxacin, in the treatment of MRSA. Linezolid is a bacteriostatic synthetic antibacterial agent of a newer class of antibiotics, the oxazolidinones, developed for MRSA, methicillin-resistant S. epidermidis , enterococci, and staphylococci. Linezolid inhibits bacterial protein synthesis by binding to the 50S ribosomal subunit to prevent translation. Adverse effects of linezolid include myelosuppression (e.g., anemia, leukopenia, pancytopenia, and thrombocytopenia), which is generally reversible on discontinuation of the drug, and Clostridium difficile colitis. A weak, nonselective, and reversible inhibitor of monoamine oxidase, linezolid may cause increased serotonin serum levels and serotonin syndrome in patients on various serotonin reuptake inhibitors, such as fluoxetine and sertraline. Prolonged use also carries a risk of polyneuropathy.
Invasive wound infections, graft loss, sepsis, and septic shock from suspected gram-positive bacteria should be treated empirically with IV vancomycin until culture-directed deescalation can occur. Vancomycin is bactericidal, preventing gram-positive bacterial cell wall glycopeptide polymerization and thereby producing immediate inhibition of cell wall synthesis and lysing of the cytoplasmic membrane. As a time-dependent antimicrobial, serum levels of this drug must unceasingly exceed the MIC to provide sufficient bactericidal activity. Due to the wide variability in vancomycin elimination among burn patients, the dosage must be individualized to optimize serum concentrations by serially following trough levels, with a typical goal of 10 to 15 μg/mL for most burn wound infections. Due to poor penetration, certain compartments, such as the lungs and central nervous system, require higher trough concentrations to achieve therapeutic levels of vancomycin; in cases of pneumonia or meningitis, concentrations of 15 to 20 μg/mL are recommended.
Alarmingly, gram-positive strains resistant to vancomycin, such as vancomycin-resistant Enterococcus (VRE) and vancomycin-intermediate S. aureus , have emerged. These bacteria are often susceptible to linezolid, as discussed earlier, as well as to tigecycline, daptomycin, quinupristin-dalfopristin, and dalbavancin, which have been developed specifically to tackle this problem. Working closely with a clinical pharmacist who can help establish an antibiotic and dosing regimen best fitting the specific resistance pattern in a given patient is critical.
Gram-negative infections more frequently require admission and IV antibiotics. While empiric therapy should be directed by local antibiogram, consideration must also be given to additive toxicity when combined with empiric gram-positive agents. Inhospital, we empirically use imipenem/cilastatin due to their low nephrotoxicity when used in conjunction with vancomycin. Deescalation and discontinuation of antibiotics should occur as soon as cultures, wounds, and physiology warrant. Because gram-negative bacteria frequently become multidrug resistant, testing for synergy between different classes of antibiotics is advisable.
Third- and fourth-generation cephalosporins and extended-spectrum penicillins are the antibiotics of choice for many burn centers for empiric coverage of gram-negative infections due to their broad coverage and low toxicity. Fourth-generation cephalosporins (e.g., cefepime), extended-spectrum β-lactamase inhibitor penicillins (e.g., piperacillin-tazobactam and ticarcillin-clavulanate), and most importantly the carbapenems (e.g., imipenem/cilastatin, meropenem, and ertapenem) are important tools in eradicating gram-negative infections. Newer fifth-generation cephalosporins were developed to treat resistant Pseudomonas ; unfortunately, novel resistance patterns are already emerging. These antibiotics are time dependent and most efficacious when serum concentrations between dosing intervals are maintained at 1 to 2 times the MIC; thus extended infusions over 3 to 4 hours or continuous infusion may be necessary to keep concentrations above MIC, particularly when a pathogen is near the resistance threshold. In perforating the cell wall, penicillins are often synergistic with intracellular antibiotics, such as aminoglycosides, and testing is warranted in the setting of highly or pan-resistant organisms. ,
Aminoglycosides remain effective for significant susceptible gram-negative infections. Much of critical care has moved to once-daily dosing of these concentration-dependent antibiotics because it is as effective and less toxic than conventional dosage intervals. Pooled data from randomized controlled studies in adults showed that once-daily administration of aminoglycosides is associated with similar or greater efficacy (e.g., bacteriologic and/or clinical cure), less nephrotoxicity, and no greater risk of ototoxicity than administration of multiple daily doses. Some clinicians remain concerned that traditional dosing intervals may be preferable in severe infections or in patients with unpredictable pharmacokinetics, as described in burn patients. Therefore monitoring aminoglycoside serum concentrations and/or the peak serum concentration to MIC ratio in burn patients with life-threatening infections, suspected toxicity or nonresponse to treatment, decreased or varying renal function, and increased aminoglycoside clearance is well indicated.
Gram-negative, multidrug-resistant organisms (MDRO) and pandrug-resistant organisms (PDRO) are increasingly prevalent. Many of these islets have sensitivities to polymyxins, a class of cell wall intercalating antibiotics that includes the topical agent polymyxin B and its IV analogue colistimethate sodium (colistin, polymyxin E). These antibiotics were largely abandoned in the 1970s due to concerns of toxicity when Kunin and Bugg showed polymyxin accumulation highest in the kidney and brain tissues, followed by liver, muscle, and lung. It was also reported that colistimethate appeared to increase the incidence of C. difficile colitis, renal dysfunction, and neuropathies proportionately to the duration of its use. , However, colistimethate use against MDRO has surged due to a lack of other systemic options. Branski et al. reported that, in 118 patients with life-threatening MDRO gram-negative infections, colistimethate provided an important salvage option for burn patients with otherwise incompletely treated infections. They further found that hepato-, neuro-, and nephrotoxicity were no different in matched patients treated with or without colistimethate, indicating concerns from 40 years ago might be unfounded with modern critical care.
Antifungal treatment is complicated by the limited number of agents and their relative toxicities. The most commonly used antifungal agent, fluconazole, has excellent activity against C. albicans and low toxicity. However, non– C. albicans spp. are increasingly frequent causes of invasive candidiasis, and these are resistant to fluconazole. The Infectious Disease Society of America advocates echinocandins as the best empiric treatment for yeast infections, as most yeast are susceptible to them. However, these are recommended only until cultures become available. Cultures should be performed frequently in these patients because resistance can develop quickly. , There is an increased prevalence of Candida spp. with greater antimicrobial resistance and higher mortality rates than C. albicans , such as C. tropicalis and C. krusei. , Posaconazole and voriconazole, both azoles, are treatments of choice for invasive Aspergillus and Fusarium . They are also effective against infections caused by Candida spp., including fluconazole-resistant ones. However, azoles have unpredictable, nonlinear pharmacokinetics with extensive interpatient and intrapatient variation in serum levels. Due to this and numerous drug–drug interactions, therapeutic drug monitoring is crucial. ,
For decades, amphotericin B deoxycholate (AmBD), an IV polyene analogue of nystatin, has been the standard choice for IV treatment of life-threatening invasive molds. This drug is associated with significant toxicity, including infusion-related events and dose-limiting renal dysfunction. Three new lipid formulations of amphotericin B (AmB lipid complex [ABLC], AmB colloidal dispersion, and liposomal AmB [LAmB]) offer several advantages over AmBD, including increased daily doses of the parent drug (up to 15-fold), high tissue concentrations in reticuloendothelial organs, a decrease in infusion-related events (especially with ABLC and LAmB), and a marked decrease in nephrotoxicity. These lipid drugs are more expensive, but their enhanced safety profiles make them the new standard for treating invasive molds, particularly Mucor . The most successful treatment for fungal infection is prevention via swift removal of all burned tissue and closure of wounds with autografts. In the presence of active mold infections, voriconazole is the first-line treatment, followed by ABLC. An echinocandin, such as caspofungin, can be considered for combination treatment of Aspergillus and Fusarium. Fusarium spp. have demonstrated innate resistance to amphotericin.
Dang et al. performed a systematic review of mucormycosis manuscripts representing 114 patients. They found disseminated mucormycosis with a mortality rate of 80% vs. 36% for patients with localized disease and found diabetes, time to diagnosis, and treatment were not predictors of survivorship. Total burn surface was an independent predictor of mortality. Standard treatment remains prompt as well as radical excision guided by frozen sections and amphotericin B greater than 5 mg/kg/day with posaconazole for salvage therapy.
Pathogens will continue to evolve novel resistance mechanisms more rapidly than researchers can develop antibiotics. In the dire case where no antibiotic susceptibility exists for an infecting pathogen, it is important to remember excision of infected tissue, graft coverage of wounds, and topical care remain effective treatment plans. Barret and Herndon noted that aggressive, early surgical treatment reduced wound culture counts from greater than 10 5 /g to less than 10 4 /g and yielded excellent skin graft take. In contrast, poorer skin graft take occurred in patients with initial counts greater than 10 6 /g but were reduced to 10 4 /g only after delayed surgical excision, thus underscoring the correlation between early and aggressive wound excisions and better patient outcomes in the treatment of severe burns. , While infections can become overwhelming, there is no bacterial resistance mechanism to the surgeon’s knife.
Specific pathogens in burn wounds
S. aureus, gram-positive cocci in clusters, remains the chief cause of burn wound infection and is a well-documented opportunistic pathogen in humans. , Colonization with these bacteria in an uninjured individual is usually asymptomatic, but they are a source of opportunistic infection that can lead to severe illness and death, especially in burn patients. Staphylococcus produces virulence factors, such as proteinases, coagulases, and hyaluronidases, that enable it to invade local tissues and disseminate hematogenously, causing generalized systemic infection and sepsis. The most common infections of Staphylococcus spp. are septicemia, cellulitis, impetigo, scalded skin syndrome, and postoperative wound infections. However, puerperal sepsis, pneumonia, osteomyelitis, endocarditis, and burn wound infection are the gravest. Exotoxins, which are produced by pathogenic strains of staphylococci, include a pyrogenic toxin, a dermonecrotizing toxin, and leukocidin. In addition to the exotoxin TSST-1, these organisms can produce enterotoxins A, B, and C, risk factors for TSS in susceptible patients. Staphylococcus spp. generally produce penicillinases that make natural penicillins ineffective and thus require treatment with penicillinase-resistant penicillins, such as oxacillin. MRSA is now the predominant isolate, with rates of infection greater than 50% in burn units. , Empiric coverage should be vancomycin for IV therapy or Bactrim and rifampin orally, as detailed earlier, based on the local antibiogram and patient cultures.
Streptococci were once the leading cause of burn wound infection but are now less prevalent. Arranged in chains, these gram-positive cocci are particularly virulent when infecting a burn wound and quantitatively do not require less than 10 5 colony-forming units (CFU)/g of tissue to prevent wound closure. A mere few β-hemolytic streptococci can cause wound infection, failure of a primary closure, and loss of a skin graft. The major infecting species are Streptococcus pyogenes (also referred to as group A streptococci and the most problematic) and S. agalactiae (group B streptococci). Natural penicillins, such as penicillin G and penicillin V, and first-generation cephalosporins are bactericidal to these species. While resistance to these penicillins or cephalosporins has not yet emerged, culture and antibiotic sensitivity data should be followed.
Enterococci are important inciters of gram-positive burn wound infection. Encouragingly, a recent review comparing sepsis mortality between consecutive decades (1989–1999 and 1999–2009) found a steep decline in the rate of infection with enterococci (25%–2%), perhaps stemming from the more liberal use of vancomycin in recent years. However, with the increasing prevalence of VRE, mortality rates from VRE are now greater than those of MRSA (58% and 33%, respectively). While most Enterococcus spp. respond to vancomycin, VRE is treated with linezolid, a combination of ampicillin and aminoglycosides, or quinupristin/dalfopristin (Synercid). Deescalation is advised as soon as indicated by culture data.
Pseudomonas is not just the most ubiquitous gram-negative burn wound pathogen, but also the most likely to be responsible for sepsis leading to burn-related death. , Both local environments and gastrointestinal tracts (via translocation of endogenous gastrointestinal flora) are believed to be the primary sources of this bacterium. This species has a predilection for moist environments, and human burn wound exudates have been shown to stimulate the expression of virulence factors of P. aeruginosa , the pathogen chiefly responsible for nosocomial respiratory tract infections. Additionally, it breeds invasive and troublesome wound infections in burn patients. A superficial wound infection caused by P. aeruginosa typically will have a yellow-green color and noxious fruity smell. This may become an invasive infection, ecthyma gangrenosum, causing purplish-blue, punched-out lesions in the skin; if local thrombosis of vessels is present, then the wound requires immediate treatment to remove newly necrotic tissues ( Fig. 10.8 ). Empiric treatment for P. aeruginosa infection has evolved from aminoglycosides to antipseudomonal β-lactams, such as piperacillin/tazobactam, cefepime, and carbapenems. The increasing prevalence of MDRO P. aeruginosa requires the expanding use of antibiotics based on culture data, employment of fifth-generation cephalosporins, use of synergistic antibiotic coverage, and use of colistimethate (see Systemic Antimicrobials in Burn Patients, earlier). This rapidly evolving and virulent pathogen is best eradicated by rapidly closing wounds to deny the bacterium access to any susceptible wound surface.
Ecthyma gangrenosa. A tissue-invasive burn wound infection here seen in a 7-year-old girl transferred to our hospital after a 6-week treatment at another hospital. (A) Note the extensive greenish and slime-coated wound consistent with an invasive wound infection. The patient died subsequently from an overwhelming hematogenously spread pneumonia. (B) The vessel wall at 40× magnification micrograph shows a clear bacterial infiltration. (C) A typical purplish ecthyma gangrenosa lesion in a 35-year-old man.
(C, From Omar P. Sangüeza, MD; Professor and Director of Dermatopathology, Wake Forest University School of Medicine, North Carolina.)
Acinetobacter spp. are gram-negative rods used commercially to convert wine to vinegar and are native flora of the respiratory tract, skin, gastrointestinal tract, and genitourinary tract. This organism may lead to numerous opportunistic infections, including pneumonia and infections of the surgical site and urinary tract. Second only to P. aeruginosa in prevalence, this pathogen has an enhanced capacity for transfer between patients due to its ability to survive in both dry and wet conditions and equally on animate and inanimate objects, whether metal or plastic, making nosocomial transmission a major concern. , Acinetobacter spp. have been isolated from diverse clinical sources, including the upper and lower respiratory tracts, the urinary tract, and surgical and burn wounds, as well as in bacteremias secondary to IV catheterization. An agent of low virulence, it has a predilection for infecting patients with dysfunctional host defense mechanisms. Although traditionally susceptible to ceftazidime and ciprofloxacin, Acinetobacter has developed resistance to such an extent that only carbapenems (e.g., imipenem and meropenem) can now be relied on to treat these infections. In cases of MDRO Acinetobacter , colistin has become the rescue treatment, as with Pseudomonas infections. ,
Stenotrophomonas maltophilia (also known as P. maltophilia or Xanthomonas maltophilia ) is an aerobic gram-negative bacillus responsible for nosocomial infections in immunocompromised patients. Increasingly reported in burn patients, this pathogen causes life-threatening infections that are very difficult to clear due to the particularly obturant biofilm Stenotrophomonas produces. Additionally, S. maltophilia is inherently resistant to a variety of antimicrobial agents, such as aminoglycosides, β-lactam agents, and carbapenems. The most common types of infections caused by S. maltophilia are wound and bacteremia; pneumonia and generalized infections associated with this pathogen are less common. , Infections are sensitive to trimethoprim-sulfamethoxazole alone or used with levofloxacin, and sensitivities should be monitored. Aggressive surgical excision and assiduous washing technique with soap and water are vital to combat the resultant biofilm.
Enterobacteriaceae, such as E. coli , Klebsiella spp., Enterobacter spp., Serratia marcescens , and Proteus spp., are often revealed as the cause of burn wound infections and other nosocomial infections in burn patients. Although these pathogens have greater sensitivity to antibiotics than other gram-negative bacteria, emerging resistance patterns to carbapenems and fourth-generation cephalosporins led to a larger array of MDRO and PDRO. Carbapenem-resistant Enterobacteriaceae are increasing in incidence and in many cases require treatment with colistin.
Anaerobic bacteria, such as Bacteroides spp. and Fusobacterium spp., are rarely a cause of invasive burn infection. These bacteria are normal flora from the oropharyngeal cavity to the gastrointestinal tracts. Anaerobic flora are responsible for 2% to 5% of surgical wound infections in the oropharyngeal area , and 10% to 15% of wound infections in the gastrointestinal and urogenital tracts. In burn patients, anaerobic infections are usually associated with avascular myonecrosis secondary to electrical injuries, frostbite, or cutaneous flame burns with concomitant crush-type injuries. With the advent of early excision and grafting, the incidence of anaerobic infections in thermal injury has been reduced significantly. If anaerobic infection is suspected, it is vital that collected specimens be placed in appropriate transport tubes void of oxygen. In these cases, broad-spectrum antibiotics covering anaerobes should be given until sensitivities are available to ensure the administration of the appropriate drug. ,
Fungal wound infection and colonization have become increasingly prevalent following the introduction of topical antibacterials and liberal use of broad-spectrum antibiotics. This has resulted in a surge in invasive fungal infection linked to higher death rates, regardless of the extent of the burn, coincident inhalation injury, or patient age. In a recent review of 15 burn units, fungi were isolated at least once from 6.3% (435/6918) of patients, with positive cultures being most commonly obtained from the wound itself followed by (in order of decreasing frequency) respiratory, urine, and blood specimens. Yeasts are identified primarily on the basis of specific biochemical tests, but both macroscopic and microscopic morphologies are also used to make final identification ( Fig. 10.9 ). Mold identification is based on growth rate, colony structure, microscopic appearance, dimorphism at different incubation temperatures, and inhibition of growth by cycloheximide, as well as various biochemical tests ( Fig. 10.10 ).