The recognition of burns and their treatment is evident in cave paintings that are more than 3500 years old. Documentation in the Egyptian Smith papyrus of 1500 bce advocated the use of a salve of resin and honey for treating burns. In 600 bce , the Chinese used tinctures and extracts from tea leaves. Nearly 200 years later, Hippocrates described the use of rendered pig fat and resin-impregnated bulky dressings, which was alternated with warm vinegar soaks augmented with tanning solutions made from oak bark. Celsus, in the 1st century ce , mentioned the use of wine and myrrh as a lotion for burns, most probably for their bacteriostatic properties. Vinegar and exposure of the open wound to air was used by Galen (130–210 ce ) as a means of treating burns, whereas the Arabian physician Rhases recommended cold water for alleviating the pain associated with burns. Ambroise Paré (1510–1590), who effectively treated burns with onions, was probably the first to describe a procedure for early burn-wound excision. In 1607, Guilhelmus Fabricius Hildanus, a German surgeon, published De Combustionibus, in which he discussed the pathophysiology of burns and made unique contributions to the treatment of contractures. In 1797 Edward Kentish published an essay describing pressure dressings as a means to relieve burn pain and blisters. Around this same time, Marjolin identified squamous cell carcinomas that developed in chronic open burn wounds. In the early 19th century, Guillaume Dupuytren ( Fig. 1.1 ) reviewed the care of 50 burn patients treated with occlusive dressings and developed a classification of burn depth that remains in use today. He was perhaps the first to recognize gastric and duodenal ulceration as a complication of severe burns, a problem that was discussed in more detail by Curling of London in 1842. In 1843 the first hospital for the treatment of large burns used a cottage on the grounds of the Edinburgh Royal Infirmary.
Guillaume Dupuytren.
Truman G. Blocker Jr. ( Fig. 1.2 ) may have been the first to demonstrate the value of the multidisciplinary team approach to disaster burns when, on April 16, 1947, two freighters loaded with ammonium nitrate fertilizer exploded at a dock in Texas City, killing 560 people and injuring more than 3000. At that time, Blocker mobilized the University of Texas Medical Branch in Galveston, Texas, to treat the arriving truckloads of casualties. The “Texas City Disaster” is still the deadliest industrial accident in American history. Over the next 9 years, Truman and Virginia Blocker followed more than 800 of these burn patients and published a number of papers and government reports on their findings. The Blockers became renowned for their work in advancing burn care, with both receiving the Harvey Allen Distinguished Service Award from the American Burn Association (ABA). Truman Blocker Jr. was also recognized for his pioneering research in treating burns “by cleansing, exposing the burn wounds to air, and feeding them as much as they could tolerate.” In 1962 his dedication to treating burned children convinced the Shriners of North America to build their first Burn Institute for Children in Galveston, Texas.
Truman G. Blocker Jr.
Between 1942 and 1952, shock, sepsis, and multiorgan failure caused a 50% mortality rate in children with burns covering 50% of their total body surface area (TBSA). Recently, burn care in children has improved survival such that a burn covering more than 95% TBSA can be survived in more than 50% of cases. In the 1970s, Andrew M. Munster ( Fig. 1.3 ) became interested in measuring quality of life after excisional surgery and other improvements led to a dramatic decrease in mortality. First published in 1982, his Burn Specific Health Scale became the foundation for most modern studies in burns outcome. The scale has since been updated and extended to children.
Andrew M. Munster.
Further improvements in burn care presented in this brief historical review include excision and coverage of the burn wound, control of infection, fluid resuscitation, nutritional support, treatment of major inhalation injuries, and support of the hypermetabolic response.
Early excision
In the early 1940s, it was recognized that one of the most effective therapies for reducing mortality from a major thermal injury was the removal of burn eschar and immediate wound closure. This approach had previously not been practical in large burns owing to the associated high rate of infection and blood loss. Between 1954 and 1959, Douglas Jackson and colleagues at the Birmingham Accident Hospital advanced this technique in a series of pilot and controlled trials starting with immediate fascial excision and grafting of small burn areas and eventually covering up to 65% of the TBSA with autograft and homograft skin. In this breakthrough publication, Jackson concluded that “with adequate safeguards, excision and grafting of 20% to 30% body surface area can be carried out on the day of injury without increased risk to the patient.” This technique, however, was far from being accepted by the majority of burn surgeons, and delayed serial excision remained the prevalent approach to large burns. It was Zora Janzekovic ( Fig. 1.4 ), working alone in Yugoslavia in the 1960s, who developed the concept of removing deep second-degree burns by tangential excision with a simple uncalibrated knife. She treated 2615 patients with deep second-degree burns by tangential excision of eschar between the third and fifth days after burn and covered the excised wound with skin autograft. Using this technique, burned patients were able to return to work within 2 weeks or so from the time of injury. For her achievements, in 1974, she received the ABA Everett Idris Evans Memorial Medal and, in 2011, the ABA lifetime achievement award.
Zora Janzekovic.
In the early 1970s, William Monafo ( Fig. 1.5 ) was one of the first Americans to advocate the use of tangential excision and grafting of larger burns. John Burke ( Fig. 1.6 ), while at Massachusetts General Hospital in Boston, reported unprecedented survival in children with burns of more than 80% TBSA. His use of a combination of tangential excision for the smaller burns (Janzekovic’s technique) and excision to the level of fascia for the larger burns resulted in a decrease in both hospital time and mortality. Lauren Engrav et al., in a randomized prospective study, compared tangential excision to nonoperative treatment of burns. This study showed that, compared with nonoperative treatment, early excision and grafting of deep second-degree burns reduced hospitalization time and hypertrophic scarring. In 1988 Ron G. Tompkins et al., in a statistical review of the Boston Shriners Hospital patient population from 1968 to 1986, reported a dramatic decrease in mortality in severely burned children that he attributed mainly to the advent of early excision and grafting of massive burns in use since the 1970s. In a randomized prospective trial of 85 patients with third-degree burns covering 30% or more of their TBSA, Herndon et al. reported a decrease in mortality in those treated with early excision of the entire wound compared with conservative treatment. Other studies have reported that prompt excision of the burn eschar improves long-term outcome and cosmesis, thereby reducing the amount of reconstructive procedures required.
William Monafo.
John Burke.
Skin grafting
Progress in skin grafting techniques has paralleled the developments in wound excision. In 1869 J.P. Reverdin, a Swiss medical student, successfully reproduced skin grafts. In the 1870s, George David Pollock popularized the method in England. The method gained widespread attention throughout Europe, but because the results were extremely variable it quickly fell into disrepute. J.S. Davis resurrected this technique in 1914 and reported the use of “small deep skin grafts,” which were later known as “pinch grafts.” Split-thickness skin grafts became more popular during the 1930s, due in part to improved and reliable instrumentation. The “Humby knife,” developed in 1936, was the first reliable dermatome, but its use was cumbersome. E.C. Padgett developed an adjustable dermatome that had cosmetic advantages and allowed the procurement of a consistent split-thickness skin graft. , Padgett also developed a system for categorizing skin grafts into four types based on thickness. In 1964 J.C. Tanner Jr. and colleagues revolutionized wound grafting with the development of the meshed skin graft ; however, for prompt excision and immediate wound closure to be practical in burns covering more than 50% of the TBSA, alternative materials and approaches to wound closure were necessary. To meet these demands, a system of cryopreservation and long-term storage of human skin for periods extending up to several months was developed. Although controversy surrounds the degree of viability of the cells within the preserved skin, this method has allowed greater flexibility in the clinical use of autologous skin and allogenic skin harvested from cadavers. J. Wesley Alexander ( Fig. 1.7 ) developed a simple method for widely expanding autograft skin and then covering it with cadaver skin. This so-called sandwich technique has been the mainstay of treatment of massively burned individuals.
J. Wesley Alexander.
In 1981 John Burke and Ioannis Yannas developed an artificial skin that consists of a silastic epidermis and a porous collagen–chondroitin dermis and is marketed today as Integra. Burke was also the first to use this artificial skin on very large burns that covered more than 80% of the TBSA. David Heimbach led one of the early multicenter randomized clinical trials using Integra. Its use in the coverage of extensive burns has remained limited partly because of the persistently high cost of the material and the need for a two-stage approach. Integra has since become popular for smaller immediate burn coverage and burn reconstruction. In 1989 J.F. Hansbrough and S.T. Boyce first reported the use of cultured autologous keratinocytes and fibroblasts on top of a collagen membrane (composite skin graft; CSG). A larger trial by Boyce revealed that the use of CSS in extensive burns reduces the requirement for harvesting of donor skin compared with conventional skin autografts and that the quality of grafted skin did not differ between CSS and skin autograft after 1 year. The search for an engineered skin substitute to replace all of the functions of intact human skin is ongoing; composite cultured skin analogs, perhaps combined with mesenchymal stem cells, may offer the best opportunity for better outcomes. ,
Topical control of infection
Infection control is an important major advancement in burn care that has reduced mortality. One of the first topical antimicrobials, sodium hypochlorite (NaClO), discovered in the 18th century, was widely used as a disinfectant throughout the 19th century, but its use was frequently associated with irritation and topical reactions. In 1915 Henry D. Dakin standardized hypochlorite solutions and described the concentration of 0.5% NaClO as most effective. His discovery came at a time when scores of severely wounded soldiers were dying of wound infections on the battlefields of World War I. With the help of a Rockefeller Institute grant, Dakin teamed up with the then already famous French surgeon and Nobel Prize winner Alexis Carrel to create a system of mechanical cleansing, surgical débridement, and topical application of hypochlorite solution, which was meticulously protocolized and used successfully in wounds and burns. Subsequently concentrations of NaClO were investigated for antibacterial activity and tissue toxicity in vitro and in vivo, and it was found that a concentration of 0.025% NaClO was most efficacious because it had sufficient bactericidal properties but fewer detrimental effects on wound healing.
Mafenide acetate (Sulfamylon), a drug used by the Germans for treatment of open wounds in World War II, was adapted for treating burns at the Institute of Surgical Research in San Antonio, Texas, by microbiologist Robert Lindberg and surgeon John Moncrief. This antibiotic would penetrate third-degree eschar and was extremely effective against a wide spectrum of pathogens. Simultaneously, in New York, Charles Fox developed silver sulfadiazine cream (Silvadene), which was almost as efficacious as mafenide acetate. Although mafenide acetate penetrates the burn eschar quickly, it is a carbonic anhydrase inhibitor that can cause systemic acidosis and compensatory hyperventilation and may lead to pulmonary edema. Because of its success in controlling infection in burns combined with minimal side effects, silver sulfadiazine has become the mainstay of topical antimicrobial therapy.
Carl Moyer and William Monafo initially used 0.5% silver nitrate soaks as a potent topical antibacterial agent for burns, a treatment that was described in their landmark publication and remains the treatment of choice in many burn centers today. With the introduction of efficacious silver-containing topical antimicrobials, burn-wound sepsis rapidly decreased. Early excision and coverage further reduced the morbidity and mortality from burn-wound sepsis. Nystatin in combination with silver sulfadiazine has been used to control Candida at Shriners Burns Hospital for Children in Galveston, Texas. Mafenide acetate, however, remains useful in treating invasive wound infections.
Nutritional support
P.A. Shaffer and W. Coleman advocated high caloric feeding for burn patients as early as 1909, and D.W. Wilmore supported supranormal feeding with a caloric intake as high as 8000 kcal/day. P. William Curreri ( Fig. 1.8 ) retrospectively looked at a number of burned patients to quantify the amount of calories required to maintain body weight over a period of time. In a study of nine adults with 40% TBSA burns, he found that maintenance feeding at 25 kcal/kg plus an additional 40 kcal/% TBSA burned per day would maintain their body weight during acute hospitalization. A.B. Sutherland proposed that children should receive 60 kcal/kg body weight plus 35 kcal/% TBSA burned per day to maintain their body weight. D.N. Herndon et al. subsequently showed that supplemental parenteral nutrition increased both immune deficiency and mortality and recommended continuous enteral feeding, when tolerated, as a standard treatment for burns.
P. William Curreri.
The composition of nutritional sources for burned patients has been debated in the past. In 1959 F.D. Moore advocated that the negative nitrogen balance and weight loss in burns and trauma should be met with an adequate intake of nitrogen and calories. This was supported by many others, including T. Blocker Jr., C. Artz, and later by Sutherland.
Fluid resuscitation
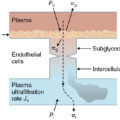
The foundation of current fluid and electrolyte management began with the studies of Frank P. Underhill, who, as professor of pharmacology and toxicology at Yale, studied 20 individuals burned in a 1921 fire at the Rialto Theatre. Underhill found that the composition of blister fluid was similar to that of plasma and could be replicated by a salt solution containing protein. He suggested that burn patient mortality was due to loss of fluid and not, as previously thought, from toxins. In 1944 C.C. Lund and N.C. Browder estimated burn surface areas and developed diagrams by which physicians could easily draw the burned areas and derive a quantifiable percent describing the surface area burned. This led to fluid replacement strategies based on surface area burned. G.A. Knaysi et al. proposed a simple “rule of nines” for evaluating the percentage of body surface area burned. In the late 1940s, O. Cope and F.D. Moore ( Figs. 1.9 and 1.10 ) were able to quantify the amount of fluid required per area burned for adequate resuscitation from the amount needed in young adults who were trapped inside the burning Cocoanut Grove nightclub in Boston in 1942. They postulated that the space between cells was a major recipient of plasma loss, causing swelling in both injured and uninjured tissues in proportion to the burn size. Moore concluded that additional fluid, over that collected from the bed sheets and measured as evaporative water loss, was needed in the first 8 hours after burn to replace “third space” losses. He then developed a formula for replacement of fluid based on the percent of the body surface area burned. M.G. Kyle and A.B. Wallace showed that the heads of children were relatively larger and the legs relatively shorter than in adults, and they modified the fluid replacement formulas for use in children. I. E. Evans and his colleagues made recommendations relating fluid requirements to body weight and surface area burned. From their recommendations, intravenous infusion of normal saline plus colloid (1.0 mL per kg/% burn) along with 2000 mL dextrose 5% solution to cover insensible water losses was administered over the first 24 hours after burn. One year later, E. Reiss presented the Brooke formula, which modified the Evans formula by substituting lactated Ringer’s for normal saline and reducing the amount of colloid given. Charles R. Baxter ( Fig. 1.11 ) and G. Tom Shires ( Fig. 1.12 ) developed a formula without colloid, which is now referred to as the Parkland formula. This is perhaps the most widely used formula today and recommends 4 mL of lactated Ringer’s solution per kg/% TBSA burned during the first 24 hours after burn. All these formulas advocate giving half of the fluid in the first 8 hours after burn and the other half in the subsequent 16 hours. Baxter and Shires discovered that after a cutaneous burn, not only is fluid deposited in the interstitial space, but marked intracellular edema also develops. The excessive disruption of the sodium-potassium pump activity results in the inability of cells to remove excess fluid. They also showed that protein, given in the first 24 hours after injury, was not necessary and postulated that, if used, it would leak out of the vessels and exacerbate edema. This was later substantiated in studies of burn patients with toxic inhalation injuries. After a severe thermal injury, fluid accumulates in the wound and, unless there is adequate and early fluid replacement, hypovolemic shock will develop. A prolonged systemic inflammatory response to severe burns can lead to multiorgan dysfunction, sepsis, and even mortality. It has been suggested that, for maximum benefit, fluid resuscitation should begin as early as 2 hours after burn. , Fluid requirements in children are greater with a concomitant inhalation injury, delayed fluid resuscitation, and larger burns.
Oliver Cope.
Francis D. Moore.
Charles R. Baxter.
G. Tom Shires.
Inhalation injury
During the 1950s and 1960s, burn wound sepsis, nutrition, kidney dysfunction, wound coverage, and shock were the main foci of burn care specialists. Over the past 50 years, these problems have been clinically treated with increasing success; hence, a greater interest in concomitant inhalation injury evolved. A simple classification of inhalation injury separates problems occurring in the first 24 hours after injury, which include upper airway obstruction and edema, from those that manifest after 24 hours. These include pulmonary edema and tracheobronchitis, which can progress to pneumonia, mucosal edema, and airway occlusion due to the formation of airway plugs from mucosal sloughing. , The extent of damage from the larynx to tracheobronchial tree depends on the solubility of the toxic substance and the duration of exposure. Nearly 45% of inhalation injuries are limited to the upper passages above the vocal cords, and 50% have an injury to the major airways. Less than 5% have a direct parenchymal injury that results in early acute respiratory death.
With the development of objective diagnostic methods, the incidence of an inhalation injury in burned patients can now be identified and its complications identified. Xenon-133 scanning was first used in 1972 in the diagnosis of inhalation injury. , When this radioisotope method is used in conjunction with a medical history, the identification of an inhalation injury is quite reliable. The fiberoptic bronchoscope is another diagnostic tool that, under topical anesthesia, can be used for the early diagnosis of an inhalation injury. It is also capable of pulmonary lavage to remove airway plugs and deposited particulate matter.
K.Z. Shirani, Basil A. Pruitt ( Fig. 1.13 ), and A.D. Mason reported that smoke inhalation injury and pneumonia, in addition to age and burn size, greatly increased burn mortality. The realization that the physician should not underresuscitate burn patients with an inhalation injury was emphasized by P.D. Navar et al. and D.N. Herndon et al. A major inhalation injury requires 2 mL per kg/% TBSA burn more fluid in the first 24 hours after burn to maintain adequate urine output and organ perfusion. Multicenter studies looking at patients with acute respiratory distress syndrome have advocated respiratory support at low peak pressures to reduce the incidence of barotrauma. The high-frequency oscillating ventilator, advocated by C.J. Fitzpatrick and J. Cortiella et al., has the added benefit of pressure ventilation at low tidal volumes plus rapid inspiratory minute volume, which provides a vibration to encourage inspissated sputum to travel up the airways. The use of heparin, N -acetylcysteine, nitric oxide inhalation, and bronchodilator aerosols have also been used with some apparent benefit, at least in pediatric populations. Inhalation injury remains one of the most prominent causes of death in thermally injured patients. In children, the lethal burn area for a 10% mortality without a concomitant inhalation injury is 73% TBSA; however with an inhalation injury, the lethal burn size for a 10% mortality rate is 50% TBSA.