Key points
- •
Multiple clinical guidelines, consensus statements, and expert management recommendations have been published for atopic dermatitis.
- •
Most prominent guidelines are from the American Academy of Dermatology and the College of Allergy, Asthma & Immunology Joint Task published in 2014 and 2012, respectively.
- •
Guidelines discuss diagnostic and grading severity criteria, which are generally used in clinical trials rather than clinical practice.
- •
There is a commonality among most guideline documents in terms of recommendations of standard interventions with moisturization, topical corticosteroids, topical calcineurin inhibitors, and phosphodiesterase-4 inhibitors.
- •
Systemic therapy recommendations are actively changing with introduction of new systemic immunomodulatory agents and other emerging therapies.
Introduction
Treatments for atopic dermatitis (AD) have evolved over the years. As our understanding of the underlying pathophysiology grows, treatment options broaden and treatment strategies are refined. This chapter provides a bird’s-eye view on the general therapeutic guideline, taking into account the recently published consensus. Following this chapter, the section Clinician’s Corner will then be divided into discussions of therapeutics from the perspectives of encountering the external factors (see Chapter 20, Chapter 21, Chapter 22 ), the internal factors (see Chapter 23 ), and the combined approaches (see Chapter 24, Chapter 25, Chapter 26, Chapter 27 ).
Overview of recently published guidelines
This chapter serves to provide a unified, consolidated overview of recently published therapeutic guidelines for the management of AD. Guidelines and similar documents, including consensus statements and expert management recommendations, are pursued and published to assist in the clinical care and management of AD patients. Many of these are produced through professional organizations that relate to specialty societies, including dermatology and allergy organizations, as well as pediatric and other primary care health care professional groups. Guidelines may vary by region, country, and continent; others are international initiatives. This chapter aims to review and emphasize the most essential current guidelines related to caring for patients with AD across the age groups, conscious that it can only relate a selection of them with cursory sampling of the actual recommendations.
This chapter will draw most of its focus from the American Academy of Allergy, Asthma & Immunology/American College of Allergy, Asthma & Immunology Joint Task Force 2012 AD Practice Parameter (JTF) and the 2014 American Academy of Dermatology (AAD) guidelines. The JTF Practice Parameter is an update of the 2004 parameter on AD; it is a single document with an executive summary, followed by evidence-based summary statements and an annotated flowchart of the diagnosis and management of AD. The AAD guidelines are organized into four separate publications. The AAD work group and JTF ranked their recommendations in similar manners, AAD from “A” to “C” and JTF from “A” to “D.” Both of these are based on the grade of evidence available for these clinical practices.
Diagnostic and severity grading criteria for atopic dermatitis
The diagnosis of AD is clinically based. In making this diagnosis, physicians should consider patient history, lesion morphology and distribution, and associated clinical signs. Various groups have created formal guidelines to aid in making a correct and reliable diagnosis of AD, though guideline criteria vary in sensitivity, specificity, and applicability to epidemiologic or clinical studies versus utility in clinical practice.
The 1980 Hanifin and Rajka criteria is one of the earliest and most recognized sets of diagnostic criteria for AD. The diagnosis of AD requires that one must meet 3 of 4 major criteria and 3 of 23 minor criteria ( Table 19.1 ). Though comprehensive and commonly used in clinical trials, the criteria can be difficult to incorporate into clinical practice—additionally, several of the minor criteria are nonspecific or uncommon ( ). Several international groups have proposed modifications to address these limitations (e.g., Kang and Tian criteria, International Study of Asthma and Allergies in Childhood [ISAAC] criteria) ( ). A systematically distilled version of the Hanifin and Rajka criteria created by the UK Working Party is often utilized for epidemiologic studies. The core set consists of one mandatory and five major criteria, all of which do not require laboratory testing. Both the Hanifin and Rajka and the UK Working Party diagnostic schemes have been validated in studies and tested in several different populations. The original UK criteria cannot be applied to very young children, although revisions to include infants have since been proposed ( ).
| Major criteria (3 of 4 must be present) | Minor criteria (≥3 must be present) |
|---|---|
|
|
A 2003 consensus conference directed by the American Academy of Dermatology suggested diagnostic schemes based on revised Hanifin and Rajka criteria that are simplified and applicable to the wide age range that can be affected by this disease ( Table 19.2 ) ( ). This version has been formally validated in children, but not adults ( ); however, it has been accepted by many for use in clinical trials, as well as considered a pragmatic approach for the clinical setting. The JTF and AAD guidelines both define AD as a chronic pruritic inflammatory disease that more commonly affects the pediatric population and is a diagnosis that is clinically based on patient’s history, characteristic clinical findings, and exclusion of other dermatoses. Both of these guidelines agree that the disease can be familial; however, the AAD guidelines associate AD with history of type 1 allergies, allergic rhinitis, and asthma ( ). The JTF guidelines assert the necessity of an atopic history, whereas the AAD guidelines only distinguish atopy as an important feature. The JTF guidelines outline the typical appearance according to the chronicity of AD lesions as pruritic, erythematous papulovesicular lesions associated with excoriation and serous exudate in the acute setting, whereas findings of lichenification, papules, and excoriations can be seen in patients with chronic AD. In contrast, AAD guidelines focus more on the standardized criteria based on the revised Hanifin and Rajka diagnostic schemes discussed earlier (see Table 19.1 ) ( ).
|
|
|
|
Multiple guideline’s criteria discuss the differential diagnosis and suggest judicial use of skin biopsy specimens or other tests (such as serum immunoglobulin E [IgE], potassium hydroxide preparation, contact allergy patch testing, and/or genetic testing), which may be helpful to rule out other or associated skin conditions. The AAD guidelines mandate the exclusion of other common cutaneous disorders before the diagnosis of AD; however, the JTF guidelines suggest this through thorough reevaluation. Other considerations discussed by the AAD work group include the lack of a requirement for specific biomarkers for a diagnosis or severity assessment and recommend against routine IgE levels ( ).
Severity grading criteria
There are a myriad of disease severity scales, the most common being the Eczema Area and Severity Index (EASI), Scoring Atopic Dermatitis (SCORAD), Investigator’s Global Assessment (IGA), and the Six Area, Six Sign Atopic Dermatitis (SASSAD) severity score.
As there are currently more than 20 different named instruments to measure the severity of AD, all of which assess different aspects of AD in different ways, a major obstacle exists in advancing evidence-based treatment. Because of this, the global Harmonising Outcome Measures for Eczema (HOME) initiative was created with the aim to standardize and validate a core set of outcome measurements for AD. As outlined on the HOME for eczema group webpage, the HOME outcome domains are divided into four categories: clinical signs measured by using a physician-assessed instrument, symptoms measured by using a patient-assessed instrument, health-related quality of life, and long-term control of flares ( ).
The EASI score evaluates the AD involvement based on the extent of dermatitis and the degree of erythema, papulation, excoriation, and lichenification by body regions. The SCORAD takes into consideration both the physician’s objectives on extent and severity as well as the subjective patient assessment of itch and sleeplessness. The Patient-Oriented Eczema Measure (POEM) is a short questionnaire solely based on the patient’s perspective in relation to symptoms and frequency. The Three Item Severity Scale (TISS) is another simplified scale that shows promise for future use in clinical practice, although it needs further testing.
The available literature suggests that the SCORAD index, the EASI score, and POEM severity scale have been adequately tested and validated, and therefore their use can be considered when practical ( ). For clinical studies, the HOME group states that all trials should measure the core domains (as referenced earlier) as this will enable trials to be compared and combined in meta-analyses. Worldwide consensus was reached that these core outcome domains should be used in all future AD trials. However, the inclusion of core outcomes does not preclude the use of any additional domains, scales, or instruments. The HOME group recommends the use of EASI score for the clinical signs core outcome, and POEM for patient reported symptoms core outcome. For the quality of life core outcome, they recommend the Dermatology Life Quality Index (DLQI) for adults, Children’s Dermatology Life Quality Index (CDLQI) for children, and the Infants’ Dermatitis Quality of Life Index (IDQoL) for infants. The RECAP (Recap of Eczema Control for Atopic Eczema) and the ADCT (Atopic Dermatitis Control Tool) are recommended as the long-term control outcome measures ( ).
These scales are primarily used in clinical trials and only rarely in clinical practice. IGA, as its name reflects, uses investigational global assessments (IGAs) in its studies. Many different IGAs have been utilized, and recently a validated IGA has been developed and recommended for use by the .
Body surface area (BSA) is a standard assessment in clinical studies and may be a useful measure in clinical practice.
The AAD guidelines state that for the general management of patients with AD, available disease severity measurement scales are not recommended for routine clinical practice because they were not designed for this purpose. Additionally, they recommend that clinicians ask general questions about itch, sleep, impact on daily activity, and persistence of disease, and currently available scales should be used mainly when practical ( ).
Nonpharmaceutic agents: moisturization, wet wrap therapy, and bleach baths
Topical therapies are the mainstay of AD treatment and are commonly used with other interventions to combat different components of this complex disease. In this section we focus on the current guidelines with reference to the JTF and AAD recommendations on these common nonpharmaceutic therapies: moisturizers, wet wrap therapy, and bleach baths.
Moisturization
Xerosis is one of the cardinal clinical features of AD. Dryness, essentially ubiquitous in AD patients, is secondary to a dysfunctional epidermal barrier and transepidermal water loss—factors that moisturizers help alleviate to some degree. Moisturizers may include emollients (i.e., glycol, soy sterols), occlusive agents (i.e., petrolatum, dimethicone, mineral oil), and humectants (i.e., glycerol, lactic acid, urea).
Moisturizers help to hydrate the skin—this has been supported both subjectively by patients and objectively by means of microscopy to assess changes in the skin’s capacitance/conductance. A number of clinical trials have shown that moisturizers lessen the symptoms and signs of AD, including pruritus, erythema, fissuring, and lichenification. Additionally, multiple randomized clinical trials have demonstrated that moisturizer use decreases the amount of prescription antiinflammatory treatments needed for disease control.
Most all guideline documents state that regardless of severity, moisturizers should be incorporated in all AD patient regimens and continued as part of a maintenance plan for prevention of flares due to strong evidence that it can reduce disease severity and the need for pharmacologic intervention. The JTF and AAD guidelines concur that moisturizers increase skin hydration by improving skin barrier function and reducing transepidermal water loss. Both agree that moisturizers are a main primary treatment for mild disease and an important adjunctive therapy in moderate and severe disease ( ).
It is expert consensus that moisturizers should be applied soon after bathing to improve skin hydration, though there is not an optimal amount or frequency of application defined by systematic studies. In the JTF and AAD guidelines, liberal and frequent application is suggested. The AAD and JTF guidelines acknowledge that most ointments carry the advantage of being preservative and fragrance free; however, no specific recommendations regarding vehicle systems and types of moisturizers have been made ( ).
Prescription emollient devices (PEDs) are generally agents with distinct ratios of lipids that mimic normal skin. They are designed and proposed to target defects in skin barrier function seen in AD patients. They have not been shown to be superior to other moisturizing products, and therefore AAD guidelines do not recommend their use ( ).
Among the few trials that have been conducted to compare and contrast specific moisturizing products, a specific moisturizer has not been deemed superior—this includes the PEDs. Choice of moisturizer is largely based on patient and provider preference, though ideally the chosen agent is effective and free of additives, fragrances, and other sensitizing agents. Despite the lack of specifics, both the JTF and AAD guidelines recommend incorporating moisturizers as part of the AD patient regimen in times of active disease, maintenance, and flare prevention.
Wet wrap therapy
AAD guidelines recommend that the use of wet wrap therapy with or without a topical corticosteroid (TCS) can be advised for patients with moderate to severe AD to decrease disease severity and water loss during flares ( ).
Both the JTF and AAD guidelines recommend wet wrap therapy with use of TCS in the setting of significant flares/refractory disease. This method tends to involve applying TCS followed by wet gauze and a second layer of dry gauze to seal in the medication. Some experts utilize two layers of clothing over TCS as an alternative. Wet wraps help to occlude and increase penetration of the therapeutic agent; additionally, it decreases water loss and provides a barrier against scratching. It should be mentioned that caution should be taken when using mid- to high-potency steroids as there is an increased chance of systemic side effects particularly if used widely on skin, though only the JTF guidelines recommend against overuse of wet wrap therapy ( ).
Bleach baths
In patients with moderate to severe AD and clinical signs of secondary bacterial infections, antimicrobial bleach baths may be recommended to reduce disease severity. For the population of AD patients that have frequent, recurrent bacterial infections both the JTF and AAD guidelines support the consideration of incorporating bleach baths in both active and maintenance periods. The effect of bleach baths on microbial colonization has been challenged by some experts ( ).
As an aside, there are no standards regarding frequency or duration of bathing appropriate for patients with AD, though both JTF and AAD guidelines recommend bathing with warm water and applying moisturizer shortly after bathing ( ). Studies have supported the utility of more frequent bathing with moisturization to follow versus infrequent bathing ( ). Limited use of nonsoap cleansers that are neutral to low pH, hypoallergenic, and fragrance free is recommended. There is not sufficient evidence to support adding oils or emollients to bath water; that being said, JTF supports additives such as baking soda or oatmeal for symptomatic relief while the AAD advises against such additives with the exception being bleach for reasons discussed earlier ( ).
Topical modalities: corticosteroids, calcineurin inhibitors, and phosphodiesterase-4 inhibitors
This section will focus on topical antiinflammatory therapies used in the treatment of AD. We focus on the current guidelines with reference to the JTF and AAD recommendations on pharmaceutic therapies, including TCS, topical calcineurin inhibitors (TCI), and phosphodiesterase-4 (PDE4) inhibitors. The choice of which topical therapy to use as maintenance therapy will depend on patient/caregiver and provider preference, access to medications (including formulary status and cost of medication), lesion location (TCS use in sensitive skin areas such as the face and eyes should be limited), and the effectiveness and tolerability observed with a particular agent ( ).
Topical corticosteroids
TCS products are used in the management of AD in both adults and children and are the mainstay of antiinflammatory therapy when nonpharmacologic interventions have failed ( ). According to both AAD and JTF guidelines, low-potency TCS are generally suggested for maintenance therapy, whereas intermediate and high-potency TCS are recommended for the acute control of AD. Twice-daily application of TCS is commonly recommended for the treatment of acute AD ( ), while some studies state that daily TCS may be as effective ( ).
Once healing is achieved (regression of eczema and improvement or resolution of itching), the goal of therapy should be long-term disease control, extending the relapse-free time for as long as possible and minimizing signs and symptoms. The use of moisturizers alone as maintenance therapy without a topical antiinflammatory may be sufficient for mild AD. For this purpose, emollients can be effective, but patients or family members should be educated to reapply topical antiinflammatory therapies with signs of relapse.
Proactive therapy with TCS may be started after eczema heals ( ). For proactive maintenance therapy, the AAD suggests once- to twice-weekly application of TCS in commonly flaring areas to prevent relapses, whereas the JTF discusses long-term control with twice-weekly TCS. Although no optimal quantity of TCS has been definitively recommended, AAD guidelines highlight the fingertip unit method applied over an area equivalent to 2 palms in addition to the use of charts that propose quantities that are age and area based ( ). Providers may also find it helpful to prescribe specific amounts of a topical agent to be used over the course of 1 week or month to ensure proper use of TCS, TCI, and/or moisturizers ( ). Both AAD and JTF guidelines caution use of TCS on areas of thin skin, such as the face, neck, and skin folds, because adverse effects are directly related to the surface area of affected skin, skin thickness, use of occlusive dressing, and potency and duration of TCS administered ( ).
Topical calcineurin inhibitors
TCI, including tacrolimus and pimecrolimus, are a distinct class of steroid-sparing, antiinflammatory agents that have been shown to be efficacious in acute flares and maintenance therapy of AD in both adults and children 2 years and older ( ). Tacrolimus ointment (0.03%) and pimecrolimus cream (1%) are approved as short-term and noncontinuous chronic treatments of AD in the United States, Canada, and Europe for children 2 years of age and older with mild to moderate AD. In addition, 0.1% tacrolimus ointment is approved for treatment of moderate to severe AD in children 16 years of age and older. The AAD and JTF guidelines agree that use of TCI at sites of sensitive or thin skin offers an advantage over use of TCS ( ). TCI are usually offered as a second-line therapy for acute and chronic treatment of AD in patients who have not responded adequately to other topical treatments or when those treatments are not recommended ( ).
Twice-daily application of either tacrolimus ointment or pimecrolimus cream is efficacious in treating inflamed AD lesions and improving or resolving pruritus. Proactive or maintenance therapy for AD that includes intermittent application of TCI twice daily or two to three times weekly to recurrent sites of involvement has been shown to reduce relapse and is recommended per AAD and JTF guidelines although not per US Food and Drug Administration (FDA) guidelines. Both AAD and JTF guidelines emphasize the importance of counseling patients on potential side effects to prevent premature discontinuation of treatment. Although not discussed in the JTF practice parameter, the AAD guidelines recommend the preceding use of TCI with TCS, where appropriate, to lessen the severity of local skin reactions with recommendation to initially use TCS to control a flare, whereas TCI can be applied as maintenance therapy to prevent relapse. In addition to informing patients about immediate site reactions with TCI, both guidelines advocate for proactive guidance regarding the FDA’s boxed warning for potential risk of malignancy ( ).
According to the European guidelines, although TCS remain the mainstay of therapy, tacrolimus and pimecrolimus are preferred in sensitive skin areas and for long-term use. Per European guidelines for children with AD, twice-weekly treatment with tacrolimus 0.03% ointment is recommended as it has been observed to reduce the number of flares and to prolong flare-free intervals and may be cost saving in children with moderate or severe AD ( ).
Topical phosphodiesterase-4 inhibitors
Crisaborole 2% topical ointment is a nonsteroidal antiinflammatory PDE4 inhibitor approved in 2016 by the FDA in the United States to treat mild to moderate AD in patients 2 years or older. As crisaborole was introduced after their publication, there are no current AAD or JTF guidelines available on this particular drug. When considering step-up therapy in mild to moderate AD when symptoms are not well controlled with a low to medium potency TCS, either TCS dose or potency can be increased, or a TCI or crisaborole can be added. Based on safety and efficacy profiles, it has been suggested that TCS be used to treat symptom exacerbations, followed by long-term maintenance therapy with a lower dose of a TCS and/or a TCI or crisaborole. Crisaborole can be used as a first-line agent in patients based on their established adverse event profiles with TCS or TCI use with the goal to proactively prevent relapses ( ).
Phototherapy
The AAD and JTF guidelines both recommend phototherapy as a treatment for AD refractory to topical treatments. Consideration of availability, cost, patient skin type, skin cancer history, and patient use of photosensitizing medications might help with the selection of phototherapy. Although AAD guidelines outline the multiple forms of light therapy without a definitive recommendation of a particular therapy, the JTF guidelines consider narrow-band ultraviolet B (UVB) as the most effective phototherapy option, given its low-risk profile, relative efficacy, availability, and provider comfort level. JTF guidelines additionally suggest using UVA1 for acute exacerbations, UVB modalities for chronic AD, and photochemotherapy with psoralen and UVA only for patients with severe widespread AD. AAD guidelines state that phototherapy can be used as maintenance therapy in patients with chronic disease. Dosing and frequency of phototherapy are dependent on minimal erythema dose, Fitzpatrick skin type, or both ( ).
Systemic therapies
Adults with severe recalcitrant AD that is not well controlled by intensive topical treatment or phototherapy and those for whom phototherapy is not a viable option may be offered systemic therapies. The choice of a systemic treatment will depend upon severity of AD, impact on the patient’s quality of life, the patient’s age and sex, family planning issues, presence of comorbidities, adverse effects, the patient’s preferences, and costs. This section will focus on current immunomodulating agents, systemic steroids, the biologic dupilumab, and emerging therapies.
Systemic immunomodulatory agents
Systemic immunomodulatory agents are indicated for the subset of adult and pediatric patients in whom optimized topical regimens and/or phototherapy do not adequately control the signs and symptoms of disease ( ). Both AAD and JTF guidelines recommend immunomodulatory agents in a subset of patients with severe AD refractory to topical regimens and phototherapy or when quality of life is severely affected ( ).
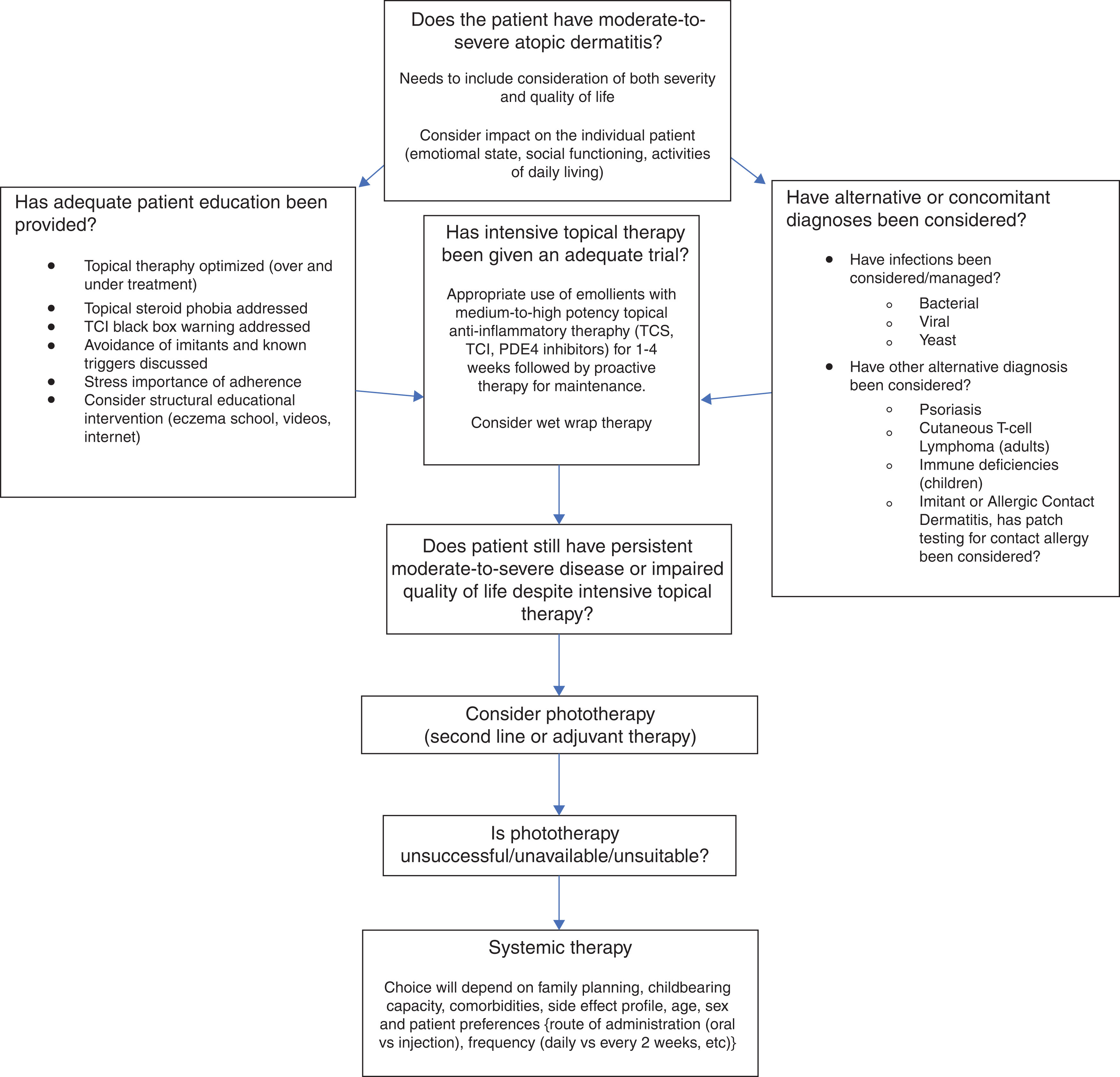
Physicians should optimize topical therapy before considering systemic medications for AD. Patients who have inadequate response should be evaluated for exacerbating factors such as cutaneous infection and for alternative diagnoses such as allergic contact dermatitis. Adequate education needs to be delivered to improve adherence to topical therapy. The general approach should be an intensive clearance period with a TCS followed by a safe and individualized regimen of intermittent TCS, TCI, or emollients to prevent flares. Phototherapy should be considered before the use of other systemic therapy if accessible and practical.
The recommends a systematic approach ( Fig. 19.1 ) to assess patients with severe signs and/or symptoms of AD and/or impact on quality of life before starting systemic therapy. One approach for identifying a candidate for systemic therapy is to utilize a disease severity score. The two most extensively validated disease severity scores are SCORAD, which incorporates the intensity of disease signs and extent along with the patient-reported sleep loss and itch, and EASI. Documentation of severe, extensive disease and/or QoL impairment at several time points with adequate topical therapy enables a holistic rationale for moving to systemic therapy ( ).