Capsular contracture is one of the most common complications following breast surgery with implants and is a common cause for reoperation. Many techniques have been described to treat or prevent recurrent capsular contracture with varying success. Acellular dermal matrix (ADM), in combination with periprosthetic capsulectomy, is a powerful tool to delay or prevent recurrent contracture. Excellent results have been obtained when this approach has been used in patients with capsular contracture, but at increased cost.
- 1.
Capsular contracture is the most frequent cause for device-related reoperation in patients undergoing breast augmentation and revision augmentation, and the second most common reason for reoperation following reconstruction.
- 2.
Many established techniques have been described to reduce or prevent recurrent capsular contracture, but they are not effective in all cases.
- 3.
Acellular dermal matrix (ADM) in combination with capsulectomy can prevent or delay recurrent capsular contracture.
- 4.
The mechanism by which recurrent contracture is inhibited is not clear but may involve a delay of inflammatory cell infiltrate into the matrix (including myofibroblasts) modulating the foreign body reaction around the implant or a biomechanical interruption of the spherical contracture process.
- 5.
More than 1 type of ADM has been shown to be effective at preventing recurrent capsular contracture.
- 6.
More data are needed to determine whether the inhibition of capsular contracture in the setting of ADM and implants is permanent, and to identify the critical accompanying steps necessary to ensure lasting success from recurrent contracture.
Etiology of capsular contracture
All implanted materials failing to acquire a blood supply incite a prototypical foreign body reaction characterized by the development of a scar interface between the foreign body and its biologic environment. Because breast implants are compliant as opposed to other implanted devices such as pacemakers or total joints, an exuberant foreign body reaction that becomes contractile around the implant will lead to deformity and pain. The resulting capsular contracture may prompt a need for reoperation, complicating the result and leading to additional recovery and expense.
Several factors have been associated with an increased risk for capsular contracture, including:
- •
Subclinical infection with biofilm
- •
Silicone versus saline implants
- •
Smooth versus textured surfaces
- •
Subglandular versus subpectoral positioning
- •
Postoperative hematoma
- •
Silicone breast implant ruptures
- •
Reoperative implant surgery
- •
Radiation therapy proceeding or following reconstruction with implants.
Pathogenesis of Capsular Contracture
Capsular contracture appears to appear at two different times:
- 1.
Early, thought to result from poor sterility or surgical technique
- 2.
Late, as a result of a chronic inflammatory process.
In either case, the exact biologic processes leading to the development of a thick, contracting capsule is unclear. Evidence from a variety of sources has implicated the activation of the fibroblast into a contractile myofibroblast as the critical event in the contracture process.
Myofibroblasts
Myofibroblasts have been noted in the capsules around breast implants since the 1970s. Early in vitro studies of capsular tissue demonstrated a prototypical response to smooth muscle agonists and antagonists similar to that expected from smooth muscle preparations. In Hinz’s description, fibroblasts differentiate into activated myofbroblasts with potent contractile properties conferred from α smooth muscle actin (SMA) in a 2- step process under the direction of transforming growth factor beta (TGFβ1), specialized extra cellular matrix (ECM) proteins like fibronectin, and the mechanical microenvironment.
Smad Signaling
Smad signaling is the major pathway through which TGFβ1 regulates expression of α-SMA in fibroblasts. In an experimental model comparing wild-type and knockout mice lacking Smad, irradiation caused thick, distorted capsules in the wild-type mouse but not the knockout mice, presumably due to inhibition of TGFβ. Zinman and collegues demonstrated that rats treated with the angiotension-converting enzyme inhibitor enalapril, a potent inhibitor of TGFβ1, had significantly less peri-prosthetic fibrosis than untreated animals presumably due in part to the role TGFβ1 plays in activation of fibroblasts to myofibroblasts.
Stress Shielding
The mechanical microenvironment also plays a role in myofibroblast differentiation due to alteration in ECM stiffness. According to Hinz, fibroblasts residing in intact tissues are stress shielded by normal ECM. With trauma or repetitive injury, increasing ECM stiffness provides a strong mechanical signal, inducing the formation of α–SMA negative stress fibers that persist until the normalization of matrix compliance. Factors that antagonize TGFβ1, normalize matrix compliance, and improve cell-to-cell contact down-regulate α–SMA in vitro; decreasing gel stress in vitro, a model for stress shielding, causes myofibroblast apoptosis.
Evidence that Acellular Dermal Matrix Plays a Useful Role in the Treatment of Capsular Contracture
Salzberg demonstrated an incidence of capsular contracture of 0.4% in review of his direct-to-implant immediate breast reconstruction in 466 breasts using ADM with 21 month follow-up including 0% contracture following irradiation.
Maxwell showed a 0% incidence of capsular contracture at 1 year postoperatively following ADM use for revisionary breast surgery.
Stump demonstrated the absence of capsule formation around implants covered in ADM in a primate model at 10 weeks with a significant decrease in myofibroblast staining in the ADM-covered implants as compared with the capsules in implants not covered with ADM.
Basu demonstrated significantly decreased inflammatory response in biopsies of ADM as compared with native capsule around expanders at the time of second-stage exchange, suggesting that ADM potentially delayed the process of capsule formation.
Technique for Correction of Capsular Contracture with ADM
There is no consensus as to the required steps to optimize the results for preventing or delaying the recurrence of capsular contracture when ADM is used. In a retrospective review, Collis demonstrated a significantly lower incidence of recurrent subglandular contracture when total capsulectomy was performed rather than anterior capsulectomy only; this seems a reasonable approach when possible, especially when considering the biofilm theory of capsular contracture or retained foreign material (as in ruptured silicone gel devices) as causative for capsular contracture.
All the following may play a role in enhancing success.
- •
Meticulous technique
- •
Precise hemostasis
- •
Conversion to a subpectoral plane
- •
Use of textured devices
- •
Ample washing with triple antibiotic solutions.
By the same token, the ideal type (human versus animal), thickness, size, or shape of ADM required to create the optimal effect has not been demonstrated. If ADM exerts its effects through stress shielding of myofibroblasts or a delay in the inflammatory response due to the slow repopulation of the matrix, it stands to reason that larger and thicker pieces might perform better than smaller and thinner ones.
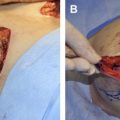
After capsulectomy, the ADM may be inset anteriorly with interrupted or running absorbable sutures or fixed in place using parachute sutures that are exited through the skin, tied loosely, covered with an occlusive dressing and removed one week later. Drains are a necessity and are discontinued when the drainage is less than 20 ccs over a 24 hour period.
Patient examples
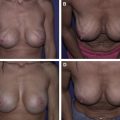
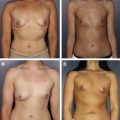
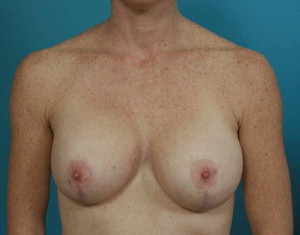
A 38-year-old patient who was 5 ft 8 in and 125 lbs underwent subpectoral augmentation with smooth-walled silicone gel devices and circumvertical mastopexy. At 1 year postoperatively, she developed a grade 3 capsular contracture of the right breast with high riding implant. At reoperation, a dense capsule was noted on the right. A total periprosthetic capsulectomy was performed; 6 × 16 cm piece of thick ADM was inset with running sutures to the anterior flap, and new implants were placed. Two years following revisionary surgery, a grade 1 capsule was present on the right (Case 1, Figs. 1–4 ). FLOAT NOT FOUND FLOAT NOT FOUND FLOAT NOT FOUND FLOAT NOT FOUND

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree