The use of acellular dermal matrices (ADMs) has become a useful adjunct to implant-based breast reconstruction and revision of the augmented breast. In both instances, the goal is replacement or reinforcement of thinned or missing tissues for implant support and control of the implant pocket. This article reviews the factors that contribute to periprosthetic tissue thinning, and the advantages and limitations of the use of ADMs for revision breast surgery and breast reconstruction. Proof of concept for the use of ADMs in the periprosthetic space is detailed from early clinical experience and histologic analysis documenting vascular ingrowth and cellular repopulation.
- 1.
Acellular dermal matrices (ADMs) have been used for more than 10 years for revision breast surgery and primary reconstructive and aesthetic breast surgery.
- 2.
ADMs may act as a template for tissue regeneration via vascular ingrowth, cellular repopulation, and tissue remodeling.
- 3.
ADMs in revision breast surgery address problems stemming from periprosthetic tissue thinning such as implant malposition and visible rippling.
- 4.
Implant-based postmastectomy reconstruction may be more stable and secure with the use of ADMs.
- 5.
Breast implant–related problems often occur in combination.
- 6.
More predictable and stable outcomes for revision breast implant surgery may be achieved with the use of ADMs in some circumstances.
Overview of breast augmentation and breast reconstruction
Breasts are important in disproportion to their specific functional role in reproduction. Why this should be so, and why the human is the only species in which breasts are permanently enlarged is a question for anthropologists and social scientists, but the evidence is clear. For example, patients who undergo mastectomy experience measurable benefits in quality of life and overall well-being after reconstruction. Among these benefits are improved feelings of sexuality and body image, particularly in, but not limited to, younger women. A line of evidence is developing suggesting that patients who undergo mastectomy plus reconstruction fare better than patients who are treated with lumpectomy plus radiation in both quality of life and physical measures (arm edema, shoulder range of motion). This suggests that the characteristics of the breast, and not simply the presence of a breast mound, are important; a breast hardened by radiation treatment may not necessarily be a preferable option to a reconstructed breast, provided that the reconstruction can be done with minimal morbidity and aesthetically pleasing results. Trends in recent years toward early reconstruction with implants supported by ADMs indicate that a shift in approaches to breast cancer treatment is underway.
Patients who undergo breast augmentation similarly accrue tangible rewards in life. For these reasons, and others less easily discernible, cosmetic and reconstructive breast surgery occupies a central role in plastic surgery. The drive to build better breasts has fueled significant innovation in the 50 years since silicone breast implants were introduced. The number of breast augmentations and postmastectomy reconstructions have dramatically increased, with augmentation becoming the most popular cosmetic surgery operation in the United States in recent years and breast reconstruction moving up to the top 5 for reconstructive procedures. Concerns about complications focused initially on the implants and led to improved design such as the use of a barrier layer to minimize gel bleed, more cohesive silicone gels, and greater variety of profiles for better matching of implant dimensions to each patient’s anatomy. These improvements led to a more favorable attitude about breast implant safety.
Surgical analysis and planning have simultaneously advanced in sophistication, including tissue analysis, optimization of muscle coverage with the dual plane, and modifications. However, certain problems inherent to breast implant surgery are inevitably magnified with the increase in the numbers of women undergoing implant surgery and a larger pool of long-term implant recipients. Despite advances in implant technology and surgical techniques, as many as 1 in 4 patients who undergo augmentation will have a reoperation within 4 years. In patients who undergo reconstruction, reoperation rates are even higher, a point emphasized by the US Food and Drug Administration in a 2011 status update. Of particular note are rates of reoperation for revision surgery, estimated at more than 1 in 3. Revision often begets further revisions, pointing to the challenges of reoperative breast implant surgery and highlighting the inadequacy of traditional approaches. Although some would argue that problems of capsular contracture, implant malposition, visible rippling, animation deformity, and other issues are largely avoidable with proper technique and implant selection, the need for solutions to these problems remains. Given that changes in tissue-implant relationships can occur over a period of years, the need for revision surgery is unlikely to be reducible beyond a minimum that is yet to be determined.
The use of ADMs has become a useful option for many of these problems. Originally conceived as a dermal substitute for burn reconstruction, ADMs did not find early acceptance for that application, but began to be used for implantation for soft tissue augmentation or replacement in other clinical settings. Volume persistence in the range of 80% to 85% at 22 months was reported for facial augmentation, and static slings for correction of facial paralysis were noted to be stable even after radiation treatment. Success with lip augmentation, septal perforation repair, and periodontal surgery suggested a range of other possible uses. The concept of stable tissue support and/or enhancement could be extrapolated to applications in breast implant surgery.
Clinical experience with ADM s
With increasing exploratory use of ADMs in revision breast surgery, the indications for their use expanded (See cases in Boxes 1 through 4 ). The examples begin with early experience and progress to more recent examples of various clinical problems. Although they represent different applications, the underlying issues highlight the common denominator of inadequate soft tissue support.
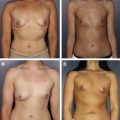
A 49-year-old woman presented after bilateral mastectomy for ductal carcinoma in situ and delayed reconstruction with an expander-implant sequence using saline implants. The expanders had been placed following satisfactory healing after mastectomies, using a dual-plane technique with muscle coverage superiorly and medially and pectoralis release inferiorly. The expansion phase was 6 weeks, reportedly without complications. A revision was done with capsulotomies and implant replacement several weeks after the expander-implant exchange. The patient’s concerns were visible rippling, asymmetry, wide intermammary space, and unnatural contours. Because of an athletic lifestyle and personal preferences, the patient refused muscle flap procedures.
Directed examination showed a tall thin woman with a body mass index (BMI) of 20.5 kg/m 2 . The implants were asymmetrically positioned with inframammary fold malposition on the right side and an intermammary distance of 6 cm. Tissue coverage was thin, with a pinch test of 5 mm on the upper pole ( CASE Fig.1 A).
Given the patient’s concerns, options were limited. A lack of tissue for implant support and coverage was determined to be the underlying problem, a condition that may be termed periprosthetic atrophy. In this case, the problem was amplified by implants of inadequate diameter (the options for implant profiles were limited at that time, which was before the introduction of high-profile or low-profile shapes.) Fat grafting was considered but there was a lack of donor volume and, although coverage may have been improved, this would not have added support. The use of acellular matrices in the periprosthetic setting had not yet been documented. However, based on limited but successful personal experience with augmentation revisions using ADMs, it seemed to be a reasonable choice among few options.
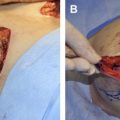
In conjunction with implant exchange and capsule revisions including bilateral medial capsulotomies and a right inferolateral capsulorrhaphy, a series of thick AlloDerm ® grafts (Lifecell Corp., Branchburg, NJ, USA) was placed. The grafts were affixed with resorbable sutures, with 1 edge of the graft across the junction of the anterior and posterior capsule. The concept was to define the boundaries of the capsule, smooth the transition from chest wall into the breast mound, and improve coverage of the implant. Long-term results have been stable, with now more than 10 years of follow-up (see CASE Fig. 1 B).
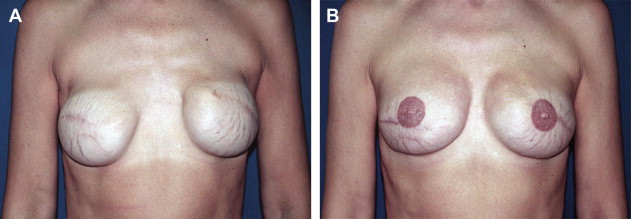
A 26-year-old woman presented with significant asymmetry after transaxillary augmentation with saline implants (left 225 mL, right 410 mL). Her concerns further included symmastia (medial fold malposition) and bottoming out (lower fold malposition) ( CASE Fig. 2 A). Analysis of the problem determined a complex tissue deficit consisting of both a severe chest wall deformity, pectus excavatum, and a thin, lax soft tissue envelope for the breast, with a pinch test of 5 mm over the central mound despite submuscular placement.
Given the multiple problems in combination, traditional options were again determined to be inadequate. A supportive tissue envelope needed to be created to prevent relapse of the right implant across the midline, but muscle flap procedures were deemed inappropriate and capsule flaps insubstantial. ADMs had proved useful for more routine cases of medial fold malposition and so the concept was extended to this more challenging situation.
To have the option of adjusting fill volumes with patient input, the revision was done in stages using expander implants (Spectrum, Mentor Corp., Santa Barbara, CA, USA) and a solid silicone block behind the right implant. In addition, extensive capsule revisions were done including a superiorly based rectus abdominis fascia flap and two 4 × 7 AlloDerm® sheets placed medially and inferiorly as onlay grafts on the right side forming a sling to the pectoralis muscle. Fill ports were removed with a final volume of 375 mL on each side. Results are shown at 1 year (see CASE Fig. 2 B).
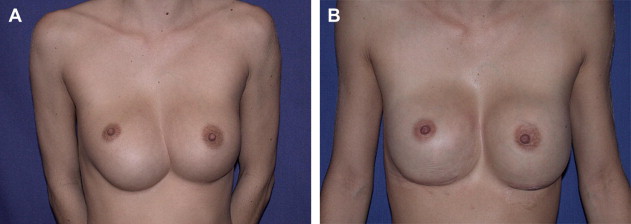
A 29-year-old fitness instructor presented for revision surgery after having subpectoral saline implants placed 7 years earlier. A year later, revision surgery had been done for malposition issues (details unknown). A deflation 3 years later prompted a replacement of both implants with 425-mL silicone gel high-profile implants. A capsulorrhaphy had been performed because of lower fold malposition with the first implants. The patient expressed concerns about severe rippling but was generally pleased with the size.
Directed examination showed a thin patient (BMI 21.7 kg/m 2 ) and significant traction ripples over both beasts ( CASE Fig. 3 A, B). Implant coverage over the upper pole was estimated at less than 1 cm by pinch test. Base diameter was measured at 13 cm.
Analysis suggested a problem of both coverage and support. With the implants already in the submuscular plane, additional options for coverage were needed, and with improved lower pole support the traction effect could be minimized. In a case of this severity, onlay grafts to the capsule in the upper pole alone are not certain to provide a definitive correction, and support to the lower pole alone may similarly be inadequate.
The surgical plan included replacement with moderate-profile implants (Allergan Style 15, 421 mL) and ADM grafting to all 4 quadrants (AlloDerm ® thick, 4 × 7 cm). This created an implant pocket separate from the skin envelope. Six-month follow-up confirmed stable correction (see CASE Fig. 3 C, D). The larger pieces of porcine-derived ADM available now would likely be a better choice.
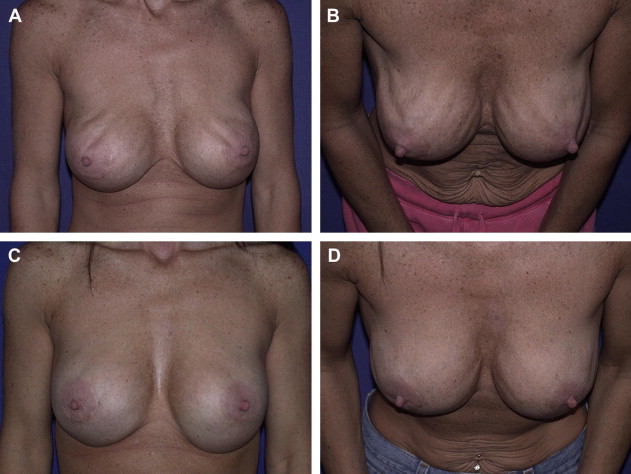
A 34-year-old gravida 3 woman presented 5 years after having subpectoral saline implants placed. Fill volume information was unknown but implants were reported to be 375 mL. The patient complained of rippling, a wide intermammary distance, animation deformity, lower fold malposition, and lateral malposition. She further requested an increase in implant size.
Examination confirmed these findings in a thin patient (BMI 20 kg/m 2 ), with an intermammary distance of 4 cm and base diameter of 13 cm ( CASE Fig. 4 A). With flexion of the pectoral muscles, significant displacement of the implants with distortion of the breast shape was observed (see CASE Fig. 4 B). Tissue coverage was estimated at 5 mm over the lower pole and lateral aspect.
At surgery, the pectoral muscle edge was mobilized from the anterior capsule (see CASE Fig. 4 C). Cautious medial capsulotomies were done, along with an inferolateral capsulorrhaphy to create a pocket to accommodate a 450-mL high-profile implant with a base diameter of 12.4 cm. An elliptical ADM graft (Strattice ™ Contour 2, Lifecell Corporation, Branchburg, NJ, USA) was used with 1 side affixed to the pectoral muscle edge and to the pocket along the inframammary and lateral folds to support the capsulorrhaphy. This concept of pectoral extension addresses the animation deformity by directing the force of muscle contraction away from the scar capsule while also improving implant coverage. Results were stable at 9 months (see CASE Fig. 4 D, E).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree