This is a brief review of the history of the role of acellular dermal matrices in breast reconstruction surgery, with a summary of several currently available products, including a table of comparisons. Key features, including biologic tissue source, surgical preparation, sterility, polarity, contraindications, shelf life, and cost, are examined. A paucity of data exists to directly compare AlloDerm ® , DermaMatrix ® , Strattice ™ , Permacol ™ , DermACELL, FlexHD ® , SurgiMend ® , and ALLOMAX™ for breast reconstruction; most studies relate to hernia repair. An ideal acellular dermal matrix product is still unavailable but the information provided in this review should facilitate a breast surgeons decision-making process.
- •
Alloderm is the most well supported ADM, with abundant evidence and historical experience among plastic reconstructive surgeons.
- •
Surgeons should be aware if a selected ADM requires orientation and be able to distinguish the different sides.
- •
Human-based products likely result in a less inflammatory reaction.
- •
Several ADMs are actually aseptic and NOT sterile, which may be a factor contributing to the increased risk of infection with ADM use in expander-based reconstruction.
- •
AlloDerm ® and DermACELL™ are exposed to antibiotics during processing and, therefore, patient allergies and sensitivities should be considered before implantation.
- •
Newer ADMs require less rehydration/rinsing in preparation for use.
- •
The use of ADMs in breast reconstruction is gaining popularity but may not be covered by insurance. Submitting for preapproval is critical to avoid significant costs to patients.
ADM options
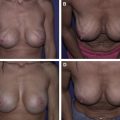
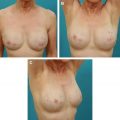
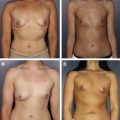
Breast reconstruction continues to evolve and, for many surgeons, now frequently incorporates use of a variety of acellular dermal matrices (ADMs), especially for lower-lateral coverage of implants, nipple reconstruction, secondary correction of breast deformities, and reinforcement of the abdominal donor defects in autologous tissue reconstruction. The introduction of these ADMs has facilitated immediate implant reconstruction. Advantages versus complete submuscular coverage include improved implant positioning via defining the inframammary and lateral mammary fold, preventing window-shading of the pectoralis muscle, serving as an internal support, shorter expansion times, and even protective effects against radiation changes. ADMs have also advanced correction of secondary breast deformities, including minimizing bottoming out, masking implant rippling, treating capsular contracture, addressing malposition, and compensating for overlying soft tissue deficiency. Concerns about using ADMs have included potential increases in infection, inflammatory reaction, seroma, masking or mimicking tumor recurrence, and obviously cost.
The diversity of available tissue products poses a significant challenge to reconstructive breast surgeons trying to select an optimal ADM and few data exist to compare these products directly. Many of the earliest studies were performed using animal models by each manufacturer and not only are the data unpublished but also they may not correlate with in vivo performance. The existing published data are mostly based on reconstruction of abdominal wall or hernia repair, which cannot necessarily be extrapolated to breast reconstruction. These products vary in many aspects, including the source of tissue, processing, storage, surgical preparation, available sizes, and cost. Several ADMs claim added benefit of cross-linking, a process that may improve the long-term tensile strength but also potentially results in an increase in foreign body reaction and encapsulation. A final consideration involves the limitations of individual insurance coverage, which may vary for each of these products. Although AlloDerm ® is gaining acceptance, many of these other ADMs are considered experimental and may result in substantial costs passed onto patients, which should be taken into consideration when submitting for insurance preapproval.
AlloDerm ® (LifeCell)
Probably the most familiar product to most plastic surgeons, AlloDerm ® was the first human dermis product available in 1994. Despite the initial indication for burn coverage, it has been subsequently expanded into a variety of applications, including abdominal wall reconstruction, head and neck reconstruction, and breast reconstruction. Early success has translated into widespread popularity among plastic and reconstructive surgeons, with the first reported case of its use in breast reconstruction in 2005 as an inferolateral sling. This was followed shortly thereafter with a report on the use of acellular cadaveric dermis with expander-based reconstruction. Today, AlloDerm has even been used as an adjunct for nipple reconstruction.
AlloDerm ® Storage and Preparation
AlloDerm ® Regenerative Tissue Matrix is a cadaveric split-thickness skin graft that is aseptically processed with sodium chloride and sodium deoxycolate and freeze-dried. It is supplied in an inner (Tyvek) pouch, which is not sterile. The package labeling, originally indicating refrigeration at 1°C to 25°C (34°F–77°F), was required to ensure the labeled shelf life, a significant distinguishing factor from other products. In a recent statement released by the manufacturer on July 14, 2010, however, the manufacturer found no deleterious effects on graft performance in animal models after exposure of −30°C (−22°F) for 10 days and 60°C (140°F) for 45 days. The product should not be used if bent, broken, or cracked before rehydration, which may require 10 to 40 minutes, another clinically noteworthy inconvenience. The AlloDerm ® itself should not be further sterilized. The manufacturer recommends submerging the tissue completely for a minimum of 5 minutes or until the backing separates in warm (up to 37°C) normal saline or lactated Ringer solution with gentle agitation, then transferring to a second bath, which can include antibiotics until fully rehydrated and pliable. The matrix must then be used within 4 hours of rehydration.
The newest available product is AlloDerm ® Regenerative Tissue Matrix (RTM) Ready to use which does not require refrigeration or rehydration. The inner pouch is actually sterile and the product is terminally sterilized to a Sterility Assurance Level of 10 −3 .
AlloDerm ® Use in Surgical Implantation
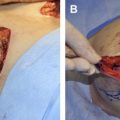
AlloDerm ® has 2 distinct surfaces and, therefore, requires attention to orientation during surgical implantation. The dermal side of the product is easily recognized by the dull, rough texture and bloodstained appearance when in contact with blood. This is generally placed against the more vascularized wound bed or tissue, such as mastectomy skin flaps, to improve revascularization. Premeshed grafts actually contain a letter “L” in the mesh pattern, which is visible in the proper orientation when the basement membrane side is up. When used for implant-based reconstruction, the manufacturer recommends 2 drains, one between the implant and AlloDerm ® and another underneath the skin flap, but commonly only a single drain is placed above the AlloDerm ® and beneath the mastectomy flap. The matrix is available in a variety of sizes and thicknesses ranging from 0.23 mm to 3.30 mm. Large sheets are available in a range from 4 cm × 12 cm to 16 cm × 20 cm for the implantable thick and X-thick products (1.04–2.28 mm and 2.30–3.30 mm, respectively).
Contraindications for AlloDerm ® Use
An under-recognized issue with AlloDerm ® pertains to the contraindications. During the processing of tissue, a variety of antibiotics are used and, theoretically, exclude any patient sensitive to any of the antibiotics listed on the package or to Polysorbate 20, used in the buffer solution.
AlloDerm ® Literature Reviews
AlloDerm ® has also been studied extensively, with 515 references in the PubMed database, including 57 related to breast reconstruction. The technique of breast reconstruction using AlloDerm ® is well described by Spear and colleagues in Surgery of the Breast: Principles and Art . LifeCell has spent 4 years developing a novel primate model 92% homologous to humans to further evaluate the immune response of ADMs beyond the 4 weeks after which other animal models form cross-species reactions. AlloDerm ® has also been reported to reconstruct the abdominal wall fascial defects after abdominal-based flap reconstruction. Unlike other newer products, AlloDerm ® has reported long-term follow-up addressing complications and evidence that it does not interfere with postoperative radiographic evaluation. A recent review series by Jansen and Macadam found 93% of the studies only level IV evidence but an overall lower rate of capsular contracture when compared to cases without AlloDerm. A subsequent cost analysis justified the use of AlloDerm ® for direct-to-implant breast reconstruction. AlloDerm ® has served as a pioneer in the application of ADMs for breast reconstruction and newer ADMs lag behind in supportive evidence at this time.
AlloDerm ® (LifeCell)
Probably the most familiar product to most plastic surgeons, AlloDerm ® was the first human dermis product available in 1994. Despite the initial indication for burn coverage, it has been subsequently expanded into a variety of applications, including abdominal wall reconstruction, head and neck reconstruction, and breast reconstruction. Early success has translated into widespread popularity among plastic and reconstructive surgeons, with the first reported case of its use in breast reconstruction in 2005 as an inferolateral sling. This was followed shortly thereafter with a report on the use of acellular cadaveric dermis with expander-based reconstruction. Today, AlloDerm has even been used as an adjunct for nipple reconstruction.
AlloDerm ® Storage and Preparation
AlloDerm ® Regenerative Tissue Matrix is a cadaveric split-thickness skin graft that is aseptically processed with sodium chloride and sodium deoxycolate and freeze-dried. It is supplied in an inner (Tyvek) pouch, which is not sterile. The package labeling, originally indicating refrigeration at 1°C to 25°C (34°F–77°F), was required to ensure the labeled shelf life, a significant distinguishing factor from other products. In a recent statement released by the manufacturer on July 14, 2010, however, the manufacturer found no deleterious effects on graft performance in animal models after exposure of −30°C (−22°F) for 10 days and 60°C (140°F) for 45 days. The product should not be used if bent, broken, or cracked before rehydration, which may require 10 to 40 minutes, another clinically noteworthy inconvenience. The AlloDerm ® itself should not be further sterilized. The manufacturer recommends submerging the tissue completely for a minimum of 5 minutes or until the backing separates in warm (up to 37°C) normal saline or lactated Ringer solution with gentle agitation, then transferring to a second bath, which can include antibiotics until fully rehydrated and pliable. The matrix must then be used within 4 hours of rehydration.
The newest available product is AlloDerm ® Regenerative Tissue Matrix (RTM) Ready to use which does not require refrigeration or rehydration. The inner pouch is actually sterile and the product is terminally sterilized to a Sterility Assurance Level of 10 −3 .
AlloDerm ® Use in Surgical Implantation
AlloDerm ® has 2 distinct surfaces and, therefore, requires attention to orientation during surgical implantation. The dermal side of the product is easily recognized by the dull, rough texture and bloodstained appearance when in contact with blood. This is generally placed against the more vascularized wound bed or tissue, such as mastectomy skin flaps, to improve revascularization. Premeshed grafts actually contain a letter “L” in the mesh pattern, which is visible in the proper orientation when the basement membrane side is up. When used for implant-based reconstruction, the manufacturer recommends 2 drains, one between the implant and AlloDerm ® and another underneath the skin flap, but commonly only a single drain is placed above the AlloDerm ® and beneath the mastectomy flap. The matrix is available in a variety of sizes and thicknesses ranging from 0.23 mm to 3.30 mm. Large sheets are available in a range from 4 cm × 12 cm to 16 cm × 20 cm for the implantable thick and X-thick products (1.04–2.28 mm and 2.30–3.30 mm, respectively).
Contraindications for AlloDerm ® Use
An under-recognized issue with AlloDerm ® pertains to the contraindications. During the processing of tissue, a variety of antibiotics are used and, theoretically, exclude any patient sensitive to any of the antibiotics listed on the package or to Polysorbate 20, used in the buffer solution.
AlloDerm ® Literature Reviews
AlloDerm ® has also been studied extensively, with 515 references in the PubMed database, including 57 related to breast reconstruction. The technique of breast reconstruction using AlloDerm ® is well described by Spear and colleagues in Surgery of the Breast: Principles and Art . LifeCell has spent 4 years developing a novel primate model 92% homologous to humans to further evaluate the immune response of ADMs beyond the 4 weeks after which other animal models form cross-species reactions. AlloDerm ® has also been reported to reconstruct the abdominal wall fascial defects after abdominal-based flap reconstruction. Unlike other newer products, AlloDerm ® has reported long-term follow-up addressing complications and evidence that it does not interfere with postoperative radiographic evaluation. A recent review series by Jansen and Macadam found 93% of the studies only level IV evidence but an overall lower rate of capsular contracture when compared to cases without AlloDerm. A subsequent cost analysis justified the use of AlloDerm ® for direct-to-implant breast reconstruction. AlloDerm ® has served as a pioneer in the application of ADMs for breast reconstruction and newer ADMs lag behind in supportive evidence at this time.
Strattice™ (LifeCell)
A more recently introduced product of LifeCell, Strattice™ Reconstructive Tissue Matrix, is a terminally sterilized, processed porcine dermis. The matrix is preserved in a phosphate-buffered aqueous solution with stabilizers designed to minimize tissue attachment, which is more advantageous in hernia repair to minimize adhesions, but effects on capsule formation are unclear.
Strattice™ Storage and Preparation
This product is stored at room temperature but the surgical preparation requires only 2 minutes of soaking in room temperature saline or lactated Ringer solution because it is fully hydrated in the original packaging and should be discarded if it is dry when opened. Similar to AlloDerm ® , the inner package is not sterile and the tissue must be removed with sterile forceps. Strattice™ is supplied in 2 versions (pliable and firm) and only a single thickness, which ranges from 1.5 to 2.0 mm. The smallest available size is 6 cm × 8 cm with the largest available up to 25 cm × 40 cm, which is a huge advantage for large abdominal wall defects. The 6 cm × 8 cm, 5 cm × 16 cm, and 8 cm × 16 cm pliable sheets are recommended for breast reconstruction.
Contraindications for Strattice™ Use
Strattice™ is contraindicated in patients with sensitivities to porcine material or Polysorbate 20.
Strattice™ Literature Reviews
Only 11 citations in PubMed are available regarding Strattice™. In a direct comparison of a human AlloDerm versus Strattice™ for hernia repair, Campbell and colleagues found less cellular and vascular infiltration in Strattice™ but improved tensile strength at 4 weeks. Although this is beneficial for hernia repair, vascularized tissue is critical to preventing infection whenever a breast prosthesis is placed. A single case report on histologic analysis of Strattice™ in breast reconstruction with an expander describes intraoperative adherence to the pectoralis muscle and overlying soft tissues. Biopsy demonstrated mild fibroblastic reaction with focal tissue integration at 6 weeks but there were areas within the Strattice™ in which angiogenesis could not be demonstrated.
DermaMatrix ® (Synthes/Musculoskeletal Transplant Foundation)
DermaMatrix ® is an acellular human dermis product processed by the Musculoskeletal Transplant Foundation (MTF) and available through Synthes. The tissue is processed with sodium chloride and detergent to remove the epidermis and viable dermal cells, then disinfected with acidic and antiseptic reagents so that it passes the United States Pharmacopeia Standard 71 for sterility. The final process involves freeze-drying.
DermaMatrix ® Storage and Preparation
Because it is freeze-dried, the product must be rehydrated in room temperature saline or lactated Ringer solution, which can take up to 3 minutes. The inner package can be placed directly onto the sterile field. To facilitate proper orientation, a notch is located in the upper left corner when the epidermal or basement membrane side is facing up. There are 4 available thicknesses (0.2–4 mm, 0.4–0.8 mm, 0.8–1.7 mm, and 1.7+ mm), which vary in sizes from 1 cm × 2 cm to 6 cm × 16 cm. DermaMatrix ® is not recommended in patients with autoimmune connective tissue disease.
DermaMatrix ® Literature Reviews
A review of published studies via a PubMed search for DermaMatrix ® yielded only 8 results. In a 2008 study comparing AlloDerm ® with DermaMatrix ® in immediate expander-based breast reconstruction, 30 patients (50 breasts) were divided into 2 cohorts. The only statistically significant difference was in the average number of days the drains were in place, 11 for AlloDerm ® versus 13 for DermaMatrix ® . The investigators reported a shorter time to final expansion, larger incremental volumes of expansion, and larger final expanded volumes with DermaMatrix ® but no difference between the final expanded volume-to-implant ratio. No histologic difference in vascularity was appreciated. Anecdotally, the investigators reported AlloDerm ® more elastic and pliable. No differences in complication rates, such as wound infection or seroma, were noted. Another study comparing 4 soft tissue substrates implanted in a submuscular murine model found AlloDerm ® became spherical and softer whereas DermaMatrix ® retained its shape and consistency.
FlexHD ® (Ethicon/Musculoskeletal Transplant Foundation)
FlexHD ® is another human allograft matrix that is minimally processed to remove the epidermal and dermal cells and available through an alliance with MTF and Ethicon. The tissue is soaked in a hypertonic solution to remove the cellular elements and rinsed with a detergent and then a proprietary disinfectant before packaging in a 70% ethanol solution. There may be trace amounts of residual ethanol on the product but no antibiotic contamination, which minimizes the potential risks of any sensitivities to antibiotic exposure. FlexHD ® cannot be subject to additional sterilization procedures.
FlexHD ® Storage and Preparation
The product is prehydrated and does not require refrigeration. A sterilized foil pouch is contained within the Tyvek package. If not used immediately within 30 minutes, the product should be maintained in a sterile saline bath and implanted or discarded within 24 hours. Similar to AlloDerm ® , the tissue must be oriented and there should be an indicating notch in the upper left side. The product is available in 3 different thicknesses: thin (0.4–0.8 mm), thick (0.8–1.7 mm), and ultrathick (1.8 mm), with variable sizes from 1 cm × 2 cm for implantation to a maximal size of 20 cm × 25 cm. A specific breast package is available for purchase in bilateral reconstruction, which offers a slight financial discount. FlexHD ® is not recommended in patients with autoimmune connective tissue disease.
FlexHD ® Literature Review
A brief search on PubMed for FlexHD ® generated only 3 results. One in vivo study using FlexHD ® and AlloDerm ® for hernia repair in a rabbit model demonstrated similar tensile strength. In a different in vitro study, FlexHD ® induced significantly more interleukin-1β, an inflammatory cytokine produced by activated macrophages, than AlloMax™ and AlloDerm ® .
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree