Abstract
Sweat and sebaceous glands have important roles in maintaining homoeostasis. In addition to their well-established role in thermoregulation, eccrine sweat glands have immunomodulatory, antimicrobial and excretory functions. Apocrine sweat is an odorless viscous fluid that contains precursors of odoriferous substances. Sebaceous glands contribute to skin barrier function, produce androgens, and have antimicrobial as well as pro- and anti-inflammatory activities.
Keywords
antimicrobial peptides, apocrine gland, cytokines, eccrine gland, immunomodulation, sebaceous gland, sweat, thermoregulation
Eccrine, Apocrine, and Apoeccrine Sweat Glands
- ▪
Eccrine sweat glands are activated by emotional and thermal stimuli and are necessary for thermoregulation; they have a generalized distribution, with the highest density on the palms and soles
- ▪
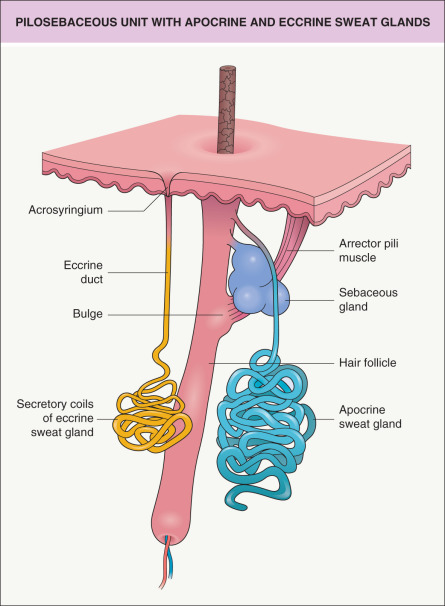
The eccrine secretory unit consists of a coiled secretory portion that drains into a long thin duct whose apical portion (acrosyringium) opens to the skin surface
- ▪
Innervation of eccrine glands consists of postganglionic sympathetic fibers that have acetylcholine as the principal neurotransmitter
- ▪
Apocrine sweat glands are androgen-dependent for their development and have an unclear function in humans; primary locations are the axillae, anogenital region, periumbilical region and nipples
- ▪
Apocrine glands, whose apical portion (acrosyringium) drains into terminal hair follicles, continuously secrete a sterile odorless viscous fluid that is rich in precursors of odoriferous substances
Introduction
The major sweat glands in humans are eccrine and apocrine glands ( Table 35.1 ). They vary in type and density, depending on anatomic location.
| CHARACTERISTICS OF SWEAT GLANDS | |||
|---|---|---|---|
| Eccrine | Apocrine | Apoeccrine * | |
| Localization | Entire body skin, highest density on palms and soles | Axillae, anogenital, periumbilical, nipples and areolae | Axillae |
| Morphology | Long, thin duct opens to skin surface | Short, thick duct opens into upper part of follicular canal | Duct comparable to eccrine gland duct |
| Secretory coil with narrow lumen | Secretory coil with wide lumen | Secretory coil with dilated and non-dilated tubular segments | |
| Cell types in secretory coil | Large secretory clear cells, dark cells, and myoepithelial cells | Epithelial (typically cuboidal) and myoepithelial cells | Eccrine and apocrine morphologic features |
| Main innervation/neurotransmitter | Sympathetic fibers/acetylcholine | Unclear/possible humoral effects of β-adrenergic receptor agonists | Sympathetic fibers/acetylcholine |
| Development | Present at birth | Present at birth | Probably not present before adolescence |
| No relationship to pilosebaceous follicle | Associated with terminal hair follicle | No relationship to pilosebaceous follicle | |
| Function/pathogenic role | Thermoregulation/role in hyperhidrosis and hypohidrosis | Unclear/some role in olfactory communication; role in follicular apocrine Fox–Fordyce disease | Unclear/possible roles in thermoregulation, axillary hyperhidrosis, and non-follicular apoeccrine Fox–Fordyce disease |
* Observed in some, but not all, studies of normal axillary skin.
Disorders of sweating are common and can be due to dysfunction of central sweat centers; sympathetic ganglia and their pre- and postganglionic fibers; or secretory glands and ducts. Clinical disorders include hyperhidrosis, hypohidrosis, and sweat retention. Hyperhidrosis, which can be emotional or secondary to systemic disease, often leads to social stigmatization. Hypohidrosis and anhidrosis may be markers for genodermatoses (see Ch. 63 ) and can result in life-threatening hyperthermia. Sweat retention disorders (i.e. miliaria) are typically transient and often occur after excessive sweating. Knowledge of the structure and function of sweat glands is important for proper diagnosis and treatment of sweating disorders (see Chs 39 & 159).
Structure, Function and Pathophysiology
In humans, there are two main types of sweat glands: eccrine and apocrine. They are distinct from one another structurally, developmentally and functionally ( Fig. 35.1 ). A third type of sweat gland, the apoeccrine gland, was described in 1987 by Sato et al., who found such glands consistently in the axillae of adults but not children. These apoeccrine sweat glands possessed certain morphologic and functional features of eccrine and apocrine glands. However, recent studies that examined serial histologic sections from control and hyperhidrosis patients, using routine and immunofluorescent staining, found no evidence of apoeccrine glands . Hence, their existence remains controversial.
Eccrine Sweat Glands
Structure
Approximately 1.5 to 4 million eccrine sweat glands are distributed over the entire cutaneous surface, with the exception of the external auditory canals, vermilion lips, clitoris, and labia minora. The highest density is found on the palms and soles. The eccrine secretory unit consists of a proximal coiled secretory portion in the lower dermis and subcutaneous tissue. This drains into a long thin duct with an apical portion (acrosyringium) that opens directly onto the skin surface (see Fig. 35.1 ) . The secretory coils contain two cell types interspersed within a single cell layer: (1) large clear cells responsible for the gland’s secretion of electrolytes and water; and (2) dark cells, of unknown function, with basophilic granules that are thought to produce sialomucin. Both cell types are surrounded by myoepithelial cells, which probably function to enhance the delivery of sweat to the skin surface. The ductal epithelium is composed of two or more layers of cuboidal cells without surrounding myoepithelium. The intraepidermal portion of the duct, the acrosyringium, is twisted like a corkscrew with similar coils in the stratum corneum . Stem cell markers (e.g. prominin-1/CD133) are expressed in the secretory and ductal portions of eccrine glands .
Innervation of the eccrine glands is provided by postganglionic sympathetic fibers that have acetylcholine (not norepinephrine) as their principal terminal neurotransmitter ( Table 35.2 ). These sympathetic fibers are controlled by the hypothalamic sweat center . The sweat center responds to its own temperature (as a reflection of core body temperature) as well as neural stimuli from the periphery.
| INNERVATION AND RECEPTOR PROFILES OF SWEAT GLANDS | ||
|---|---|---|
| Eccrine | Apocrine | |
| Nerve fibers near gland | + | ± |
| Cholinergic muscarinic (M 3 ) receptors | ++ | ± |
| α 1 -adrenergic receptors | + | − |
| β 2 – > β 3 -adrenergic receptors | + | + |
| Purinergic receptors | + | + |
Development
During embryogenesis, at 3 months’ gestation, sweat glands begin to develop as cords of epithelial cells that bud from the epidermal ridges on the palms and soles. By 5 months’ gestation, similar structures have appeared over the remainder of the body. Functional eccrine glands are present at birth and react to thermal and emotional stimuli. Unlike apocrine glands, they have no developmental relationship with the pilosebaceous follicle .
Function
Eccrine sweat is a sterile, dilute electrolyte solution that contains primarily sodium chloride (NaCl), potassium and bicarbonate. Other components include antimicrobial peptides (e.g. dermcidin) , proteolytic enzymes, glucose, pyruvate, lactate, urea, ammonia, calcium, amino acids, epidermal growth factor, cytokines and immunoglobulins. In addition, other organic compounds and heavy metals such as arsenic, cadmium, lead, and mercury are excreted in sweat . A recent study demonstrated that sweat activates NF-κB, ERK and JNK pathways in keratinocytes, resulting in the upregulation of interleukin (IL)-8 and IL-1β production . Keratinocyte outgrowths from eccrine sweat glands also have a role in re-epithelialization of human wounds .
The quantity and quality of eccrine sweat secretion varies greatly, depending on emotional and environmental stimuli. There are several methods for measuring sweat production ( Table 35.3 ). Under maximal stimulation, the body can produce 3 liters in 1 hour. Sweat is formed in two steps : (1) release of nearly isotonic primary sweat by the secretory coil; and (2) partial reabsorption of NaCl by duct cells, resulting in the delivery of a hypotonic fluid to the skin surface. The final concentration of NaCl may be higher when sweat is produced at a more rapid rate. The neuropeptide galanin and its receptors were recently shown to regulate transepithelial ion transport and fluid secretion from human eccrine sweat glands .
| METHODS FOR EVALUATING SWEAT PRODUCTION |
|
Continuous secretion of sweat provides a mechanism for thermoregulation via evaporative heat loss, maintenance of electrolyte balance, and keeping the stratum corneum moist to ensure fine tactile skills and pliability of the palms and soles. The excretory function of the sweat gland can be instrumental in the delivery of systemically administered drugs to the stratum corneum (e.g. ketoconazole, griseofulvin), and it provides an explanation for cutaneous side effects of certain chemotherapeutic drugs. Botulinum toxin can be used to treat eccrine hyperhidrosis. After dermal injection, botulinum toxin taken up by the nerve terminal interferes with proteins required to release acetylcholine at the neuroglandular junction (see Fig. 159.1 ).
Pathophysiology
Disorders affecting eccrine function involve modifications of sweat quantity or quality; these can be localized (e.g. distribution of the auriculotemporal nerve in Frey syndrome [see Ch. 39 ]) or generalized (e.g. hypohidrotic ectodermal dysplasia). The sweat glands are also affected in cystic fibrosis (CF). Individuals with CF lose excessive amounts of salt (NaCl) when they sweat, and the most common screening test for CF measures the amount of Cl − in the sweat.
Hyper- and hypohidrosis
Increased activity of eccrine glands is typically due to enhanced cutaneous sympathetic outflow in response to mental and thermal stimuli. The eccrine gland’s secretory clear cell is thought to represent the major source of sweat in hyperhidrosis . The emotional form of hyperhidrosis results primarily in excess sweating from the palms, soles, and axillae . This can be a major inconvenience, but it is not associated with morphologic or functional abnormalities of the glands, nor with internal diseases . Stimulation of eccrine sweat glands may also occur in association with hypoestrogenemia due to menopause and a variety of underlying disorders (see Ch. 39 ), including neurologic, infectious, neoplastic (e.g. lymphomas), and endocrine diseases.
Increased sweating can lead to overhydration and even maceration of the skin. This may be accompanied by secondary bacterial and fungal infections (see Chs 74 & 77). Proteolytic activity by resident and transient skin flora (microbiota) can lead to odor-producing substances (e.g. ammonia, short-chain fatty acids); this is referred to as eccrine bromhidrosis.
Hypohidrosis or anhidrosis may be congenital, e.g. due to a reduced number of eccrine glands in patients with hypohidrotic ectodermal dysplasia (see Ch. 63 ). It can also be acquired, e.g. due to poral occlusion in patients with scleroderma or administration of medications with anticholinergic effects. Acquired and inherited neuropathies represent additional causes of hypohidrosis.
Drug-related eccrine disorders
Concentration of drugs in the eccrine glands is well known. Drugs known to be secreted into eccrine sweat include chemotherapeutic agents (e.g. cyclophosphamide, cytarabine), antibiotics (e.g. ciprofloxacin, β-lactams) and antifungals. In patients receiving chemotherapy, excretion of toxic components into eccrine sweat is thought to play a role in the production of neutrophilic eccrine hidradenitis and other forms of toxic erythema of chemotherapy ( Fig. 35.2 ). Toxic insult to the eccrine ductal cells from chemotherapy leads to the histologic finding of eccrine squamous syringometaplasia (see Ch. 21 ).