(1)
Universidade Federal Fluminense, Niterói, Rio de Janeiro, Brazil
When cases of AIDS began to be reported in the early 1980s, in the most of the time cutaneous mucous manifestations presented profuse clinical forms, occurring typically in the immunocompetent individual. In fact, these more aggressive forms are most visible in immunosuppressed patients not only by HIV, but also by other causes (neoplasia, transplanted, frequent users of steroids, antineoplastic…).
On the other hand, we should not ignore that infectious diseases are usually dependent on the following factors, among others: number of infectious germs, virulence of these germs and host resistance. Thus, the clinical manifestations can be very aggressive in an immunosuppressed individual if the infecting load is too large and/or the infecting virulence of the etiological agent is also very high.
Nowadays, especially in Brazil, where almost all HIV people registered in STD/AIDS programs receive antiretroviral last generation drugs, some manifestations, once common, have become much less frequent. There is a same concerning about the vertical transmission, as well as the deaths caused by HIV/AIDS. It usually happens due to the decrease of the blood viral load and the increase in the CD4 cells number which considerably reduce the manifestation of opportunistic infections. However, the use of antiretroviral drugs, here and around the world, may cause several side effects.
Our message is addressed to all professionals working with STD, mainly the health team actuating in regions where there is a prevalence of STD/HIV. As with STD, we should not postpone the opportunity of an early approach on sexuality topics, STD/HIV and reproductive health, including counseling on a possible syphilis, HIV and hepatitis B testing. Oncotic colpocitology for women should be included.
10.1 Synonymy
AIDS, Acquired Immune Deficiency Syndrome.
10.2 Concept
Represents an advanced immunosuppression state caused by HIV virus. The main viral action is to induce the decrease of T CD4+ (CD4) lymphocyte count to levels below 20% of the normal value (i.e., <200 cells/mm3).
10.3 Incubation Period
For aids: 5–10 years without antiretroviral treatment.
10.4 Etiological Agent
HIV vírus belongs to the Retroviridae family, which is composed of four virus: HTLV I, II, III and IV. HTLV III corresponds to HIV. Later on, two types of HIV (1 and 2) were discovered with distinct geographical predominance, and each one of them with subtypes A, B, C, D and E. They consist of RNA genome with protein capsid and lipoprotein cover inside the capsid. Enzymes such as the reverse transcriptase, protease, and others responsible for the viral multiplication are found in viral RNA, over which the current antiretroviral therapy is based. The envelope contains two glycoproteins, Gp120 and Gp41, which guarantees the binding of the virus to the host cell. HIV has tropism to T helper lymphocytes, macrophages and dendritic cells.
10.5 Clinical Manifestations (Figs. 10.1–10.115)
Acute Infection: acute infection is defined as the first few weeks of HIV infection until the emergence of anti-HIV antibodies (seroconversion), which usually occurs around the fourth week after the contagion. Currently it is known that 50–90% of individuals infected have a set of clinical manifestations, called Acute Retroviral Syndrome (ARS), formerly called Seroconversion Syndrome, which usually occurs between the first and third week after infection, limited to 3–4 weeks. It has a very varied range of clinical pictures, from oligosymptomatic forms to lagging manifestations with rarer complications, such as aseptic meningitis and Guillain-Barré syndrome. In a third of cases ARS can manifest itself as a Mononucleosis-like syndrome (symptoms of an acute viral syndrome: fever, adenopathy, axillary and cervical occipital, erythematous, cutaneous mucous rash pharyngitis, myalgia, arthralgia, diarrhea, headache, nausea, vomiting and hepatosplenomegaly). Cutaneous mucous lesions are small aphthous ulcers that can be observed in the mouth, anus and genitals. Along with the viremia there is an abrupt fall of CD4 lymphocyte. A transient leukopenia with lymphopenia and later CD4/CD8 inversion can occur. Then, due to the biological response of the organism, CD4 rises up and the viral multiplication falls, remaining at stable levels. The level of viral multiplication will dictate the progression (fast or slow) for what we call aids.
Asymptomatic or Latent Phase: 3–12 weeks from the beginning of the infection, most patients seroconverts (serology). Anti-HIV humoral immunity partially stops viral replication, causing the decrease of viremia and the increase of CD4 levels, however, never to previous normal levels. This phase can last from 2 to 20 years (an average of 10 years) and generally the CD4 count is higher than 350 cells/mm3.
Symptomatic Phase: emerges with the continuity of the viral replication increasing the viremia and making the CD4 drop to values below 350 cells/mm3. It is characterized by mild to moderate immune compromised with increased frequency of manifestations of some diseases such as bacterial, fungal and common protozoan infections in the community and even neoplasia. With no treatment, a progressive CD4 drop to less than 200 cells/mm3 happens, determining a strong immunosuppression and emergence of severe opportunistic diseases, defining the acquired immunodeficiency syndrome (AIDS), which can lead to death of patient. This phase has a varied duration, but an average of 2–3 years.
The list of diseases defining AIDS, main opportunistic diseases, therapeutic consensus and pregnancy procedures can be found in www.aids.gov.br/aids.
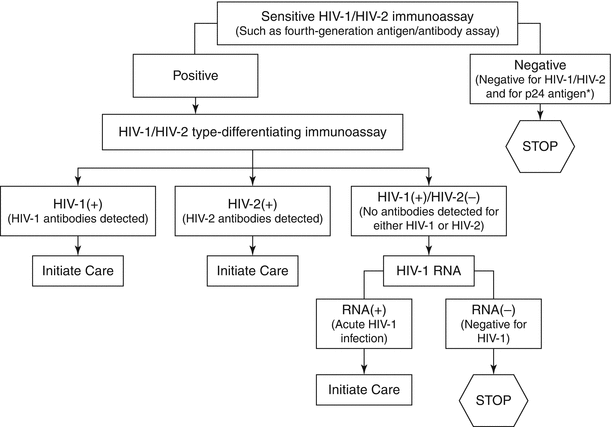
10.6 Laboratory Diagnosis [1, 2]
Diagnosis can be made of two ways, combined or not, as follows: directly, the detection of the virus or its components (proteins and other antigens, genetic material, culture isolation, etc.), or indirectly, by detection of the immune response to the virus. The antibodies through serum tests are the methods traditionally carried out. The serologic positivity usually occurs between 3 and 12 weeks from contamination. Almost all infected patients present positive serology up to 6 months of contagion. In the immunological window period, ranging from HIV infection until the laboratory detection, patient can transmit the virus, but the serology is still negative. The technology advance has allowed a diagnosis with a smaller immunological window, currently 4–6 weeks.
Tests types are as follows:
- (a)
Serologic classification tests:
- 1.
Immunoassays (IE), as the immunoenzymatic type tests ELISA (Enzyme-Linked Immunosorbent Assay). The latest, of fourth-generation, combine detection of antigens, besides the traditional antibodies.
- 2.
Rapid tests (RT): simple immunoassays that can take 30 min. There are several formats of RT, and the most frequently used are: devices (or strips) of immunochromatography of lateral flow, dual migration immunochromatography (DPP), immunoconcentration devices and the solid phase.
- 1.
- (b)
Complementary tests: false-positive results may occur in classification tests. For this reason, additional tests are necessary for the diagnosis definition. Additional tests use different formats and principles. Are included in this category:
- 1.
Serological: Western Blot (WB), Immunoblot (IB) or line immunoassays (LIA—Line Immuno Assay), including Rapid Immunoblot (RIB) and Indirect Immunofluorescence (IIF), HIV-1/HIV-2 antibody differentiation assay [Multispot™], the latter a rapid test of ultimate generation.
- 2.
Molecular tests: detect pro-viral DNA or RNA, is the second most used, called the “viral load”, and commonly made by the quantitative polymerase chain reaction method.
- 1.
10.7 WHO Case Definition for HIV Infection
- 1.
Adults and children aged 18 months or older. HIV infection diagnosis based on:
positive HIV antibody test (rapid or laboratory-based enzyme immunoassay). It is usually confirmed by a second HIV antibody test (rapid or laboratory-based enzymeimmunoassay) relying on different antigens or of different operating characteristics.
and/or;
positive virological test for HIV or its components (HIV-RNA or HIV-DNA or ultrasensitive HIV p24 antigen) confirmed by a second virological test obtained from a separate determination.
- 2.
Children younger than 18 months. HIV infection diagnosis based on:
a positive virological test for HIV or its components (HIV-RNA or HIV-DNA or ultrasensitive HIV p24 antigen) confirmed by a second virological test obtained from a separate determination taken more than 4 weeks after birth. Positive antibody testing is not recommended for definitive or confirmatory diagnosis of HIV infection in children until 18 months of age.
10.8 Evaluation of Diagnosis Methods
Since the beginning of HIV epidemic, the infection serologic diagnosis is made with at least two tests, one for the classification and the second, more specific and different from the first one, to confirm. The most widely utilized combination, usually called the gold standard, was accomplished through a selection immunoassay, such as ELISA, followed by Western Blot (WB) as a confirmation test. The World Health Organization (WHO) accepts, for example, that two different rapid tests in series are sufficient to conclude the diagnosis in locations with few resources and high prevalence. Modern tests allow the using of flowcharts for the diagnosis, according to the local prevalence of the disease and the availability of resources in each scenario, so it should be observed the protocols defined for each country to confirm the disease. It is important to point out that all newly diagnosed individuals should test their viral load, which in fact constitutes a third test to ratify the presence of infection. If there is any doubt about the diagnosis it is recommended to repeat the tests in intervals of at least 30 days.
10.9 Treatment and Cure Control
An incurable, but tractable disease.
The antiretroviral treatment is mandatory as soon as possible of all individuals presenting aids defining illnesses, as well as of pregnant women and of those with less than 500 CD4 cells/mm3. There is an increasingly world tendency towards an early start, even in asymptomatic individuals with good immunity. This tendency was adopted in Brazil since 2014 to reduce HIV sexual transmission. The precocity of the treatment also postpones the breakdown of the immune system, and reduces the inflammatory process induced by HIV, the supporter element of thromboembolic phenomena, thus preventing cerebrovascular and cardiovascular diseases, and premature aging of the organism. To treat HIV it is necessary at least three antiretroviral combination drugs, two of them belonging to different classes. Treatment is complex and needs medical supervision to assess adaptations of the organism to the treatment, their side effects and possible difficulties in following correctly the medical recommendations, that is, to adhere to treatment. A good multidisciplinary monitoring can keep the disease under control for many years. So, with no viral resistance (a growing problem) or uncontrollable intercurrences. It is obvious that the patient, their family members and sexual partners must actively participate in this monitoring.
According to its capabilities and delivery model for medications, each country has therapeutic protocols concerning the initial scheme, drugs used with co-infections such as tuberculosis and viral hepatitis, rescue schemes for patients with resistant virus. The treatment usually begins with two nucleoside reverse transcriptase inhibitors and non-nucleoside inhibitor or a protease inhibitor potentiated with ritonavir. In Brazil, for example, there is the universal recommendation to start the treatment by combining tenofovir, lamivudine and efavirenz. As there is a large number of researches on AIDS, data arise every year often modifying the procedures. We recommend the regular consultation of the some guidelines [3–5].
10.10 Antiretroviral Drugs Classes [3–5]
Reverse Transcriptase Nucleoside Inhibitor: act on the enzyme reverse transcriptase, incorporating to the DNA strand created by the virus. Make this chain imperfect, preventing the virus reproduction. They are the following: Abacavir, Didanosine, Stavudine, Lamivudine and Zidovudine.
Reverse Transcriptase Non-nucleoside Inhibitors: block directly the enzyme action and the virus multiplication. They are the following: Efavirenz, Nevirapine, Rilpivirine and Etravirine.
Nucleotides Inhibitors: Tenofovir.
Protease inhibitors: act on the protease enzyme, blocking its action and preventing the production of new copies of cells infected with HIV. They are the following: Atazanavir, Darunavir, Fosamprenavir, Indinavir, Lopinavir, Nelfinavir, Ritonavir, Saquinavir and Tipranavir.
Fusion inhibitors: prevent that virus enter into the cell, thus it cannot reproduce itself. Example: Enfuvirtide.
CCR5 receptor antagonist: Maraviroc.
Integrase inhibitors: block the activity of the enzyme integrase, which is responsible for the integration of HIV DNA into the human DNA (genetic code of the cell). Thus, inhibits the replication of the virus and its ability to infect new cells. Examples: Raltegravir, Dolutegravir, Elvitegravir/Cobicistat.
10.11 Complications
Result from the immunosuppression caused by viral infection (opportunistic infections, neoplasm, consumptive syndrome) and by adverse reactions to therapy with antiretroviral drugs (hepatotoxicity, neuropathies, pancreatitis…).
10.12 Differential Diagnosis
Differential diagnosis should be made with all diseases capable of manifesting as a mononucleosis syndrome: cytomegalovirus, rubella, toxoplasmosis, hepatitis, syphilis (especially in the roseola’s fase).
10.12.1 Notes
Transmission can occur through blood transfusion, accident with perforating and cutting material, sharing of syringes and needles, sexual and perinatal contact (transplacental, during delivery and during breastfeeding).
The mother-to-child transmission can be drastically reduced if antiretroviral therapy is carried out during pregnancy, especially at time of birth. Today the elective caesarean is a great choice of delivery route, reducing the possibility of HIV transmission to the fetus. However, patients with a viral load of less than 1000 copies/mL may not benefit from the c-section. In such cases, the indication of c-section can be by obstetric criteria. However, we should be aware that the emergency caesarean section increases the possibility of vertical transmission. Clamping the cord as soon as possible is imperative, as well as avoiding invasive measures in the fetus.
Breastfeeding is formally contraindicated.
Recurrent vulvovaginal candidiasis may be the first clinical manifestation of HIV infection.
HIV infected patients with PID manifestation or invasive cervical cancer have disease classification changed.
10.12.2 Vaccines in People Living with HIV
Vaccination recommendations for HIV carriers, adults or children, usually follow the same principles of the rest of the population, with some exceptions/special care.
First, the immunogenicity in this group of people is smaller, mainly those with CD4 counts less than 200–350, and more doses or higher doses are needed.Special care must be taken with vaccines made with live attenuated agents. None can be used if the CD4 cell count is less than 200 cells/microL (or percentage less than 15% in children up to 5 years of age) or clinical signs of immunosuppression.
All the attenuated vaccines (rotavirus, varicella zoster, yellow fever, MMR) should be administered individually with an interval of at least 1 month. Exceptions: do not use attenuated vaccines against polio or influenza, even in those with good immunity. Choose the vaccines with inactive antigens.
The BCG vaccine is controversial: contraindicated by the American CDC, independent of the immune status. The Ministry of Health of Brazil recommends vaccination of children with HIV with good immunity in a single dose, without reinforcement.
10.12.3 Zoster/Varicella
The zoster and varicella vaccine, despite using the varicella-zoster attenuated virus, has different presentations, indications, and dosages.
Immunization against varicella: recommended for children and adults with CD4 count equal or greater than 200 cells/microL (or percentage greater than or equal to 15% in children up to 5 years of age), without clinical signs of immunosuppression, and do not have vaccine history (2 doses) or varicella. The anti-varicella IgG serum dosage can solve doubts.
Dosage: Example: Varivax™, two 0.5 mL SC doses with at least 4–8 weeks interval.
Immunization against zoster: promising evidence recommend vaccination of HIV carriers with more than 60 years of age with CD4 count equal or greater than 200 cells/microL, without clinical signs of immunodsuppression, and preferably in use of antiretroviral drugs with suppressed viral load. Check the recommendations according to indication of local health authorities.
Example: Zostavax™ 19,400 UI/0.65 mL dose. Dose: single SC dose in deltoid. Discontinue use of anti-herpetic medications 24 h before the dose and do not use them for at least 14 days.
10.13 HPV
People living with HIV should receive HPV vaccination scheme. In countries where the Gardasil 9 Valent™ vaccine (6, 11, 16, 18, 31, 33, 45, 52, 58) is already available, this should be the first indication. Otherwise, our specification is vaccination with Gardasil 4 Valent™ (6, 11, 16, 18). The vaccination schedule should be three doses: 0 day, 60 days and 180 days in boys/girls more than 9 years old, men/women.
Other vaccines are in research.
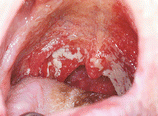
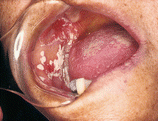
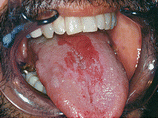
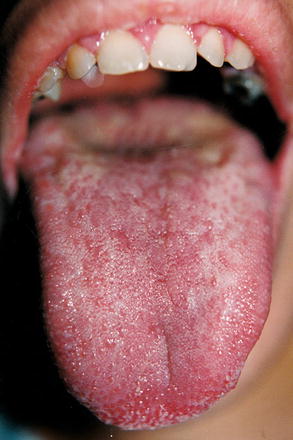

Fig. 10.1
Pseudomembranous candidiasis. White detachable plates by scraping, located in oropharynx, characterizing pseudomembranous Candidiasis in a patient with AIDS. It is possible to observe a brown pigmented area in the back of the tongue due to smoking. In this region, the tongue papillaes are hypertrophied due to Candida infection, giving the clinical aspect of the so-called black hairy tongue

Fig. 10.2
This image shows white plates easily deployable and intensely erythematous erosions in the jugal mucous and dorsum of the tongue due to pseudomembranous Candidiasis, which constitutes one of the most common clinical findings in HIV positive patients

Fig. 10.3
Candidiasis represented by an eroded irregular erythematous area well demarcated with disappearance of the filiform papillae on the dorsal surface of the tongue of a patient with AIDS

Fig. 10.4
Candidiasis in patient with AIDS. Clinical manifestation less intense than those presented previously. It is classic in medical practice the involvement of oral, pharyngeal and esophageal mucosa by Candida sp. infection in immunodeficiencies, and it should be pointed out that it involves not only HIV
10.14 Hairy Leukoplakia
A particular form of leukoplakia described in 1981 for the first time as a white lesion of epithelial hyperplasia and hyperkeratosis, which is not removed during the scraping with wooden spatula, for example. Its presentation varies from white vertical strokes to leukoplastic thick and wrinkled areas. Represents an opportunistic infection related to the presence of the Epstein-Barr virus (EBV), almost exclusively in patients infected with human immunodeficiency virus (HIV). Although rare, there are cases reported in patients receiving organ transplants and even in immunocompetent individuals.
It is a well-defined white lesion, which varies from flat to a plaque with vertical roughened striations. Frequently it occurs along the side edges of the tongue, with occasional extension to the dorsal or ventral surface, being common the bilateral presentation, however, it occasionally occurs unilaterally and can rarely get in the jugal mucosa.
Clinical differential diagnosis of hairy leukoplakia includes idiopathic frictional hyperkeratosis, leukoplakia (biting tongue), tobacco and candidiasis associated leukoplakia.
The treatment of hairy leukoplakia is not usually specific or necessary.
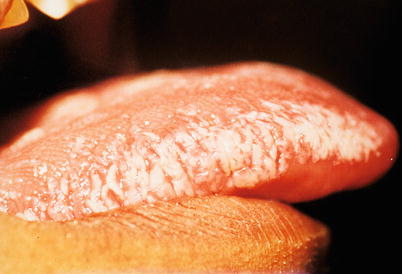
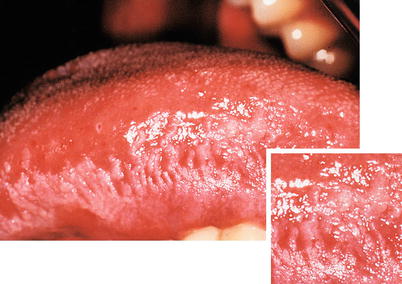
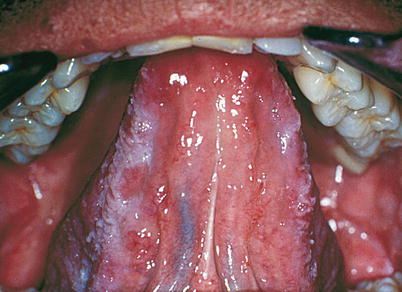
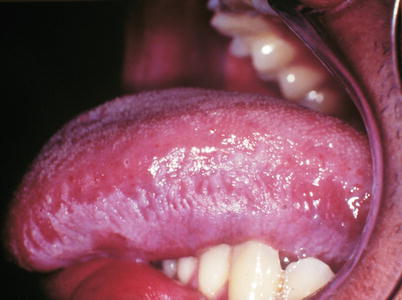
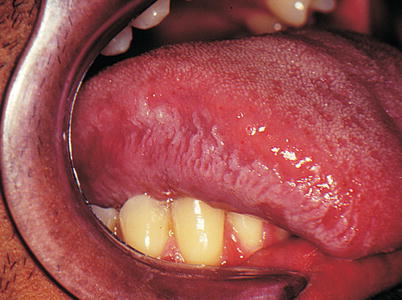
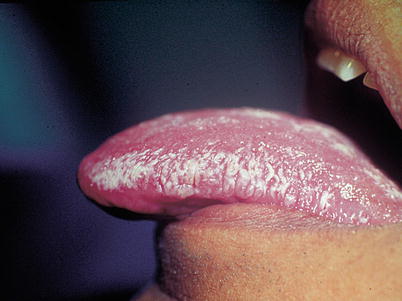
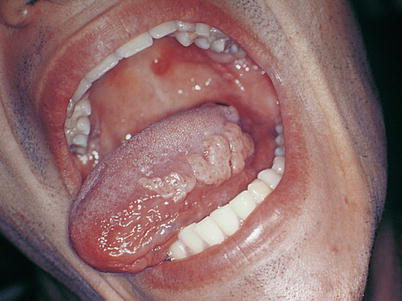
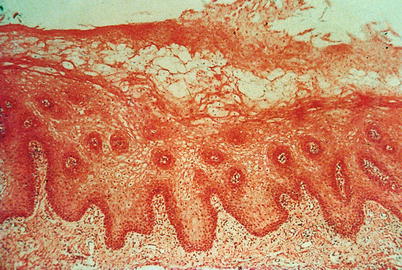
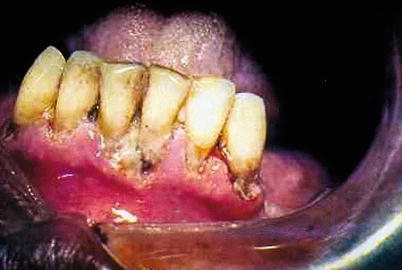

Fig. 10.5
Hairy leukoplakia in patients with AIDS. White plaque extending around the lateral border of the tongue, roughened in its surface and showing vertical striations

Fig. 10.6
Hairy leukoplakia in patients with AIDS. In the zoomed image its striated and wrinkled appearance can be observed

Fig. 10.7
Oral hairy leukoplakia in patients with AIDS. The Figure shows the presence of bilateral leukoplakia, as well as lesions extending into the ventral surface of the tongue, which are common aspects of the condition. It is important to point out that this lesion is suggestive of aids and its causal agent is the Epstein-Barr virus (EBV-HHV-4)

Fig. 10.8
Same patient of previous Figure, in which it is observed in detail the wrinkled and ridged appearance of whitish plaque characterizing oral hairy leukoplakia on the left side of the lateral border and lingual belly

Fig. 10.9
The image of the lateral border of the tongue on the right side of the same patient of previous Figures reveals the presence of a corrugated and grooved white plaque, characterizing bilateral oral hairy leukoplakia

Fig. 10.10
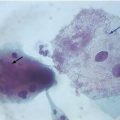
White plaque located on the lateral edge of the tongue of irregular surface and rough aspect in HIV-positive patient. The lesion involves the entire length of the lateral border of the tongue. The biopsy of material revealed hyperkeratosis and balloon cells in the upper layers of the stratum spinosum. In addition, nucleus of epithelial cells showing an aspect of “pearl necklace” was observed. Such microscopic findings are compatible with the diagnosis of oral hairy leukoplakia

Fig. 10.11
Patient in the secondary stage of syphilis, being possible to observe the presence of high whitish color plaques with rough surfaces and irregular forms located in the ventral lateral edge and the left side of the tongue. This condition is known as plane condyloma or papular syphilide. It should be noted that this is a necrotic lesion and that it can be removed by scraping, as opposed to hairy leukoplakia. An eroded irregular erythematous area on the palate can be observed

Fig. 10.12
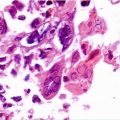
Histological section showing irregular and thick parakeratosis and keratinization on the surface of the epithelium, which is hyperplastic and shows a stained band of balloon cells, little rosy on the upper portion of the spinous layer. This epithelial picture is not exclusive of hairy leukoplakia, however, when combined with practice, it is strongly suggestive of the diagnosis. Viral DNA detection of Epstein-Barr virus should complement the recognition of the lesion

Fig. 10.13
Necrotizing periodontitis showing ulceration of the gingival tissue associated with gingival papillary necrosis in patients with AIDS
10.15 Noma
Condition also known as Cancrum oris, noma stomatitis or necrotizing stomatitis. It is an opportunistic infection with rapid progression caused by normal components of the oral microbiota that becomes pathogenic during periods of commitment of the immunity state. The most common predisposing factors are the following: malnutrition, dehydration, poor oral hygiene, recent diseases, malignant neoplasm, immunodeficiency (including aids).
The process often begins as a painful ulceration in the gingiva extending quickly and causing necrosis as in acute necrotizing ulcerative gingivitis. The areas of necrosis may develop in the soft tissue without continuity with the gingiva or extend deeply into tissues and develop pale zones originating areas of yellow necrosis. The process can spread to the underlying bone, with large areas of possible osteomyelitis. The typical characteristics are: fetid odor, significant pain, fever, general malaise and regional lymphadenopathy.
Treatment consists of controlling the predisposing factors, as well as the infection itself. Nutrition must be restored, as well as hydration and electrolyte balance, along with the administration of antibiotics. Conservative debridement should be performed in large areas of necrosis.
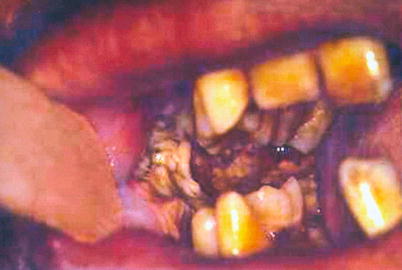

Fig. 10.14
Noma in patient with AIDS. Extensive yellowish necrosis area with rapid progression, which began in gingiva and extended to the jugal mucosa, promoting severe tissue destruction resulting from anaerobe microorganisms, fusiform bacilli (Fusobacterium fusiform) and a spirochete (Borrelia vicent)

Fig. 10.15
Noma in patient with AIDS. Exteriorization of cutaneous lesion as a result of intrabuccal tissue destruction observed in Fig. 10.14
10.16 Kaposi’s Sarcoma
Kaposi’s sarcoma, first described in 1872 by the Hungarian physician living in Austria Moritz Kaposi, is an uncommon vascular neoplasm originated in the proliferation of endothelial cells, although the dermal dendrocytes and submucosa, macrophages, lymphocytes and mastocytes may play a role in the genesis of the lesions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree