Abstract
In spite of the craniofacial surgeon’s best efforts to implement preventative measures, he/she will, on occasion, be called upon to correct secondary soft tissue deformities that result from surgical exposures or the traumatic injuries themselves. In these cases, careful planning and judicious use of the techniques presented are the key to optimizing patient outcomes. The chapter outlines the proper evaluation and treatment of secondary soft tissue deformities across the several subunits of the head and neck.
Keywords
soft tissue, trauma, secondary reconstruction
Despite the craniofacial surgeon’s best efforts to minimize soft tissue disruption, secondary scarring and deformity are not infrequent following the treatment of facial injuries. These deformities may arise as a consequence of the trauma itself or may be an iatrogenic consequence of the exposure and soft tissue disruption required for facial fracture fixation. Such scarring and deformity are both functionally and psychologically disturbing to the patient and often need to be addressed surgically. This is especially true in the head and neck region, where anatomical distortions measured in millimeters can be visible at conversation distance. The purpose of this chapter is to describe these deformities, how to avoid them through modified surgical approaches, and the techniques used for correcting them secondarily.
Background and Etiology
Prevention of secondary deformity is the primary long-term goal of treating acute facial injuries. However, as outlined in the previous chapter on primary repair of soft tissue injuries, the need for secondary revision is often unavoidable. When the zone of injury is extensive, healing and soft tissue viability become unpredictable, limiting the reconstructive options available in the acute period. In addition, patients may be too unstable to receive an optimal primary reconstruction. In these cases in which the patient requires stabilization first and foremost, the immediate surgical goal is confined to soft tissue closure and coverage of vital structures and bone.
Soft tissue disruption is often associated with underlying skeletal injury in need of open reduction and internal fixation. The surgical approaches to the craniofacial skeleton required for fixation carry with them a predictable risk of iatrogenic secondary deformity. Ptosis of soft tissues, transection or disinsertion of muscles, atrophy of fat pads, alopecia, and nerve damage are just some of the postoperative complications attributed to craniofacial exposures. These complications arise from the required soft tissue dissection, subperiosteal degloving, and subsequent scarring.
With the advent of plate and screw osteosynthetic systems for the craniofacial skeleton, internal fixation of facial fractures followed the principles of extensive fracture exposure in order to maximize rigidity and minimize interfragmentary movement. This principle was followed even in cases of low energy trauma with simple fracture patterns and in spite of a lack of understanding of the physiological loading of the craniofacial skeleton. Over time it has become clear that the complications arising from the soft tissue disruption and biological compromise inherent in this aggressive approach offset the benefits of rigid fixation. This has lead to the paradigm for facial fracture stabilization changing over the last few decades, with a shift from stabilization by maximal rigidity to stabilization using the minimum stiffness required to achieve bone healing, consequently better preserving biology, which in turn has led to a reduction in complication rates.
This approach is also less likely to disrupt soft tissue support structures and hence result in less secondary soft tissue deformity. As an example, lower eyelid incisions, a significant source of secondary soft tissue deformity, can often be avoided in the fixation of low-energy zygoma fractures not requiring orbital floor exploration. Mechanical stabilization of these fractures can effectively be achieved with fixation at the zygomatic–frontal fracture and/or at the zygomatic–maxillary buttress. Furthermore, the inferior orbital rim fracture site can be readily visualized from the upper buccal sulcus incision ( Fig. 3.3.1 ), precluding the need for the lower lid exposure for the purposes of ensuring an adequate reduction in cases where orbital floor exploration is not necessary.
Complications can also arise from the fixation hardware itself, with visibility and prominence, loosening, pain and infection being common reasons for secondary removal. Complete mitigation of hardware-related complications is impossible, which again emphasizes the need to achieve stable bony fixation with the least amount of hardware possible.
Initial Assessment
History and Physical Examination
Comprehensive assessment should begin with a history documenting details of the initial injury and primary treatment. The timeline of secondary interventions, both surgical and nonsurgical, should also be documented. The patient’s chief concerns need to be thoroughly explored and placed in order of priority. Risk factors for poor soft tissue healing such as diabetes, smoking, use of glucocorticoid steroids, and personal or family history of keloid scarring need to be identified.
Examination should assess for facial asymmetries, tissue deficiencies, and distortion of key structures and landmarks. Both static and dynamic asymmetries should be assessed along with function of all divisions of the facial nerve. If bilateral injuries are present, the use of preinjury photographs will assist in this process. The quality of the soft tissues should then be assessed noting all scars and their characteristics including pigmentation, vascularity, pliability, and height. For each soft tissue deformity an attempt should be made to determine the underlying cause. This can be as simple as the incorrect alignment of key landmarks during the primary repair, or in complex cases, a combination of malalignment, abnormal scarring, tissue deficiency, and bony malunion. The contribution of each of these factors to the deformity must be carefully considered to appropriately plan for stepwise correction. Assessment of the underlying facial skeleton should determine if accurate reconstruction has been achieved, and if not which areas are malunited, deficient or unstable. Temporal and cheek contour deformities are then examined in relation to the facial skeleton. The mobility of displaced soft tissues should be assessed to determine if they are fixed or passively correctable.
A cranial nerve examination should be performed with particular attention to the facial and trigeminal nerves. The power of each muscle group and the presence of synkinesis should be noted along with any functional issues such as corneal exposure, nasal airway obstruction, and oral incompetence. Dysaesthesias and painful neuromas can result from damage to any of the trigeminal nerve branches. The most common cause of posttraumatic trigeminal nerve injury is compression followed by partial nerve laceration and, infrequently, complete laceration. Operative decompression and neurolysis at the foramen or fracture site is effective in most compressive injuries. Complete lacerations require microsurgical repair, and may require autologous nerve grafting if the repair is delayed.
Finally, a set of standard facial photographs should be obtained for documentation and operative planning. Three-dimensional (3D) facial scanning technology is also becoming more accessible for use in preoperative planning and documentation. These techniques have the advantage of allowing computer-based simulation of corrective procedures and development of 3D templates to aid in soft tissue reconstruction.
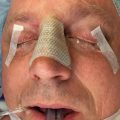
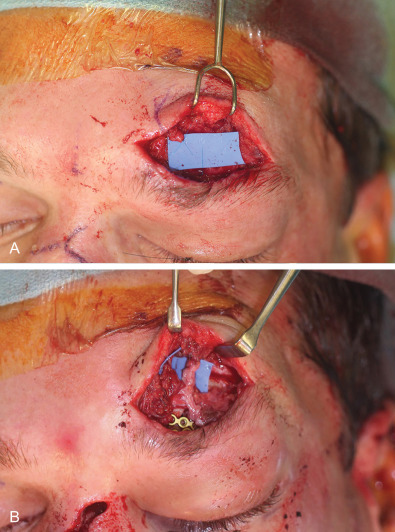
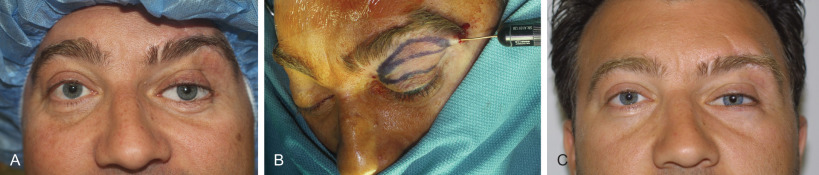
Fig. 3.3.2 demonstrates a case of posttraumatic dysaesthesia of the supraorbital nerve following laceration and fracturing of the supraorbital rim. Laceration of the nerve went unrecognized during primary soft tissue repair and open reduction internal fixation. Secondary exploration was carried out revealing the nerves’ discontinuity and direct microsurgical repair was performed with eventual resolution of symptoms.
Radiographic Examination
In evaluating significant secondary soft tissue deformities, computed tomography (CT) examination should be routinely performed to assess the adequacy of skeletal reconstruction, including symmetry and position of key segments. High resolution images are useful for preoperative planning of skeletal corrective procedures, including osteotomies, bone grafting, and customized implants. Soft tissues can also be assessed through CT providing information on their quantity, quality, and location relative to bony structures. Axial images best demonstrate the soft tissues of the midface and their relation to the infraorbital rim. Coronal slices are best for evaluation of the temporalis muscle and overlying fat pad.
Principles of Secondary Soft Tissue Correction
Sequence of Revision
A specific sequence of reconstruction provides optimal correction of posttraumatic facial soft tissue deformities. Skeletal deformities are corrected first, as an accurately reconstructed facial skeleton is required for the correct repositioning or reconstruction of soft tissues. Defects and contour irregularities of the facial skeleton should be corrected with bone graft, free tissue transfer or alloplastic implants depending on the clinical situation. Grossly malpositioned bony segments should be osteotomized and repositioned.
With skeletal correction complete, the deformity and deficiency of the soft tissues can be appropriately addressed. Soft tissues should be fully freed from any points of adhesion to allow for tension-free resuspension. Bone suture anchors are useful for fixation of soft tissues. Key soft tissue landmarks such as the canthi and oral commissures are carefully repositioned with anchoring of ligamentous attachments to bone, and resuspension of muscles.
Finally, residual soft tissue deficiencies are corrected with submuscular or subcutaneous volume augmentation.
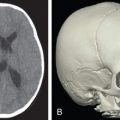
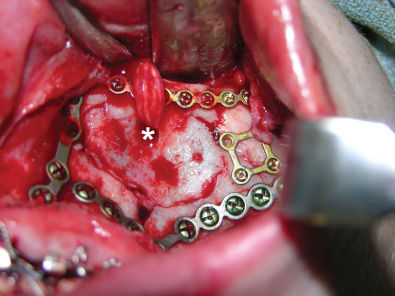
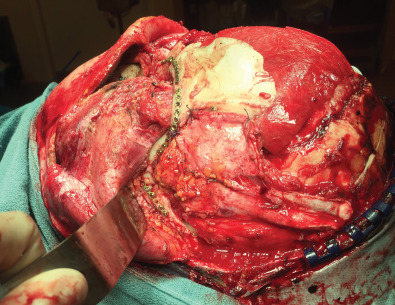
Fig. 3.3.3 demonstrates a patient following a motor vehicle collision resulting in a compound skull fracture and loss of the right zygomatic arch. Initial treatment of the patient’s skeletal defects was limited due to their critical condition and severe neurotrauma. Following the acute phase, staged secondary reconstruction was planned. First, a custom made PEEK implant to address the cranial and zygomatic defects was designed based on CT imaging. Following skeletal correction, the true soft tissue deficit in the temporal fossa became apparent and could be appropriately addressed. Serial fat grafting was then employed to complete the soft tissue reconstruction.
Soft Tissue Contour Augmentation
Corrective techniques for soft tissue contour deformities include autologous fat grafting, synthetic fillers, alloplastic implants, and occasionally free tissue transfer. Selecting the appropriate technique requires consideration of amount of volume loss, quality of the overlying skin and soft tissues as well as the defect location. Small volume deficiencies are readily corrected using autologous fat grafting, while moderate-sized defects may require serial grafting. Hyaluronic acid-based synthetic fillers can be useful for small volume soft tissue augmentation, however the temporary nature of these products makes them less ideal in this patient population.
Fat Grafting
Although initially described over 100 years ago it was not until the 1990s that the modern technique for fat grafting became well accepted. Fat grafting has now become a common procedure within the armamentarium of most reconstructive surgeons. Although there is much debate over the technical aspects of graft harvest, preparation, and injection, evidence is largely lacking to endorse one protocol over another. Our current practice follows the principles of structural fat grafting described by Coleman.
- 1.
Gentle handling of fat to prevent damage to its delicate structure of parcels.
- 2.
Limited centrifugation to refine and concentrate the fat, removing non-viable components.
- 3.
Small volume injection of fat aliquots.
As seen in Fig. 3.3.4, our usual donor sites are the distal thigh/medial knee or the abdomen accessed through a stab incision made within the umbilicus, leaving an inconspicuous scar. The location of harvest has not been proven to affect overall graft take and so can be adjusted based on availability of adipose tissue and patient preference. Harvesting of fat using both syringe suction and pump aspiration have been shown to yield equivalent graft take, and so the choice is based on surgeon preference and availability of equipment. Our preference is to use syringe suction because of the small volumes required for facial augmentation, its simplicity, and low cost. Harvest sites are infiltrated with 1% lidocaine with 1 : 200,000 epinephrine diluted in normal saline (generally 1 : 5). Once harvested, the fat is centrifuged for 3 minutes at 3000 rotations per minute. The aqueous layer is then drained from the syringe and the oil is decanted. Further oil can be wicked from the fat using gauze. The prepared fat is then transferred into one milliliter (mL) syringes for injection.
If performed with the patient awake, such as in the case of small defects, injection sites are sparingly infiltrated with local anesthetic containing epinephrine, and stab incisions are made in inconspicuous locations such as within the hairline, natural creases or existing scars. Each pass of the cannula should deposit a small parcel of fat into a tunnel surrounded by vascular tissue. Multiple tunnels are created with successive passes and no more than 0.1 mL of fat deposited per pass as the cannula is withdrawn. Larger deposits per pass are thought to inhibit graft take and lead to fat necrosis and oil cyst formation. Depth of injection can be varied depending on the desired effect. Augmentation of depressed scars is accomplished through superficial injection into a subdermal plane. An intermediate subcutaneous plane is injected for augmentation of soft tissue volume. A deep plane of injection adjacent to the periosteum will affect the draping of soft tissues over the bony skeleton.
With proper technique, long-term graft take is reported up to 60%. Optimal correction in one sitting is difficult because of the variability in rates of graft take, and patients should be informed about the possibility of multiple grafting sessions. The most common complication following fat grafting is visible irregularities, which can occur due to superficial placement of graft or fat necrosis and oil cyst formation. Fat embolism is a known but very rare complication following vascular cannulation. Use of blunt injection cannulas and epinephrine solution at the injection site help to mitigate this risk.
Fig. 3.3.4 demonstrates a patient with significant facial deformity following repair of extensive midfacial fracturing. Staged secondary reconstruction began with osteotomy and repositioning of the malunited zygoma and enucleation of the left globe with eventual prosthetic placement. A lateral canthoplasty was then performed to correct the canthal dystopia and ectropion. With the skeletal and periorbital structures accurately repositioned, fat grafting to the left upper lid hollowing was undertaken. Superficial injection of graft was avoided to prevent contour irregularities in the thin eyelid skin. A slight overcorrection was obtained immediately following grafting, leading to a satisfactory postoperative result.
Temporal Contour Deformity
The temporal fossa is filled by the temporalis muscle and superficial temporal fat pad creating a smooth convex contour from the insertion of the temporalis fascia on the superior temporal line to the zygomatic arch. Concavity in the temporal region presents with prominence of the lateral orbital rim and zygomatic arch. This can result from either soft tissue volume loss or expansion of the skeletal boundaries of the temporal fossa. Lateral displacement of the zygomatic arch or rotation of the zygoma leads to increased depth of the temporal fossa with resulting contour deformity. Soft tissue volume loss in the temporal fossa can result from atrophy or displacement of the temporalis muscle or temporal fat pads. The deformity is most commonly associated with coronal flap exposure of the lateral orbital rim and zygomatic arch. MRI and cadaver studies have demonstrated that atrophy or prolapse of the superficial temporal fat pad is the primary cause of the contour deformity following this approach and failure to close the periosteum over the zygomaticofrontal suture (orbital periosteum and temporalis fascia). Dissection within the superficial fat pad may result in ischemic injury to the fat with subsequent atrophy, or dehiscence of supporting ligaments causing inferior displacement of the fat pad.
Direct injury to the temporalis muscle from trauma or surgical access can also result in tissue loss, atrophy or ptosis. In traumatic injuries repair and resuspension of the muscle should be performed as best as possible with priority placed on filling the anterior portion of the temporal fossa adjacent to the lateral orbital rim and zygomatic arch ( Fig. 3.3.5 ). Resuspension is facilitated through use of drill holes, suture anchors or fixation to fracture plates. If elevation of the temporalis muscle is required for surgical access to the lateral skull base, a cuff of muscle and fascia at the superior temporal line should be left to facilitate accurate resuspension.
The patient’s fracture type and overall injury severity can also influence postoperative rates of temporal hollowing. Higher energy injuries are more difficult to accurately reconstruct, and have more direct soft tissue injury. Polytrauma patients are also at risk of significant decreases in body mass index, which increases their risk of temporal hollowing.
Prevention of temporal contour deformity focuses on avoiding disruption of the superficial temporal fat pad during coronal flap elevation. Direct damage to the fat pad, its vascular supply and dehiscence of its suspensory fibers are all proposed mechanisms for the development of hollowing. Our preferred method and the most common variation of the coronal approach passes through the anterior layer of the deep temporalis muscle fascia 2–3 cm above the zygomatic arch. Dissection then continues on the undersurface of the superficial fascia leaving the underlying fat pad undisturbed. This theoretically allows for simultaneous protection of the fat pad and temporal branch of the facial nerve which is closely associated with the periosteum of the zygomatic arch. Use of marking sutures on both sides of the cut temporal fascia allows for accurate reapproximation during closure. Some authors bypass dissection of the superficial temporal fat pad entirely by dissecting above the temporalis fascia until the superior posterior edge or the zygomatic arch can then be directly accessed. The reported rates of both facial nerve injury and temporal hollowing are low with this approach. However, in the setting of severe trauma it may be difficult to perform this approach safely given the displacement of bony landmarks and the disruption of soft tissue planes. Another approach transects the temporalis fascia above the superficial temporal fat pad and continues the dissecting on the undersurface of the deep layer of the temporalis fascia. This supratemporalis approach bypasses the fat pad entirely and also preserves the blood supply from the middle temporal artery. There are currently no large randomized trials showing definitive benefit to one approach over another.
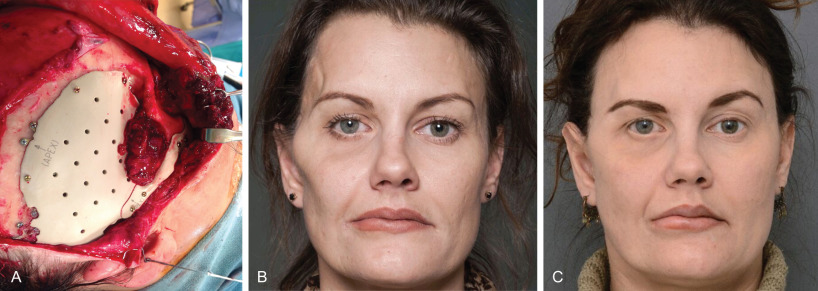
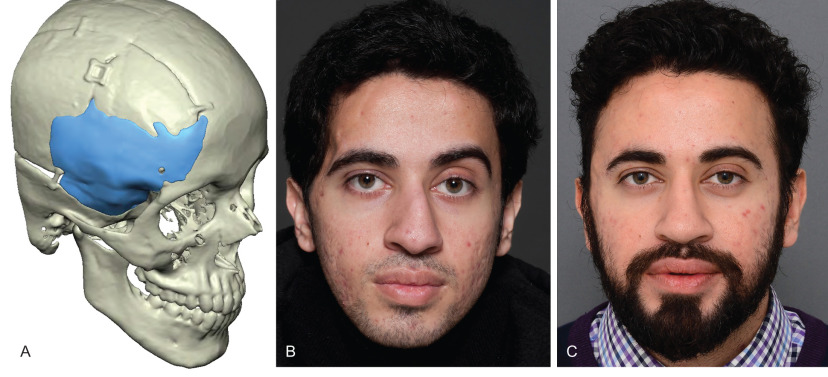
Several treatments have been described for correction of established temporal hollowing, with the goal of treatment being to address any underlying bony abnormality and deficiencies of the soft tissues. Prior to the popularization of structural fat grafting, soft tissue augmentation was achieved with many different alloplastic materials including hydroxyapatite, porous high-density polyethylene, and methylmethacrylate. These firm, adynamic materials were most often placed in a submuscular plane to mask their borders and prevent palpability. Unfortunately dissection of the temporalis muscle could itself lead to further soft tissue atrophy. Subcutaneous placement was performed in patients where the temporalis muscle was missing or deficient, and although this filled the soft tissue defect, it resulted in an unnatural appearance. Custom temporal implants theoretically offer a technologically superior means of mirroring the unaffected side following traumatic injuries of the temporal region, however the limitations of using alloplastic materials still apply. The thin traumatized soft tissues present in these cases do little to hide visible irregularities and shadow lines that occur even with the best implant-based reconstruction. When an implant is required, such as for cranial reconstruction, secondary use of structural fat grafting offers an excellent means of “fine tuning” the reconstruction. Fig. 3.3.6 demonstrates a case of posttraumatic temporal hollowing with temporal bone resorption and soft tissue atrophy following a decompressive craniectomy. A custom PEEK implant was first placed to reconstruct the defect in the temporal bone and correct the bony deficiency. Serial fat grafting was then used to complete the reconstruction, providing excellent symmetry and masking of the implant.
For patients without skeletal defects our current practice for correction of temporal hollowing is primarily through structural fat grafting. Depending on the degree of deformity one or more sessions may be required to achieve full correction. The success of this technique in head and neck reconstruction has been well documented. Dermal fat grafting has also been successfully described for temporal soft tissue augmentation, with the authors claiming lasting correction in a one-stage procedure. Unlike implant-based reconstructions, fat grafting can provide a softer, less artificial appearance and feel. It also mitigates the risk of implant-related complications such as infection and extrusion.
Cheek Ptosis
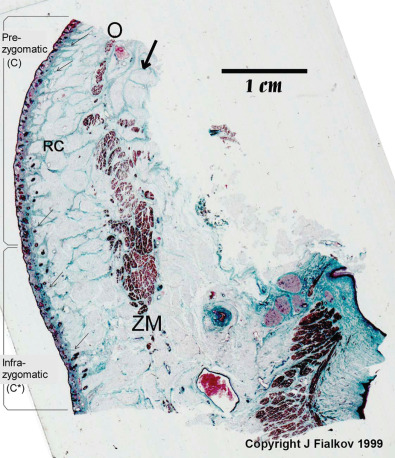
Ptosis of cheek tissues is a well-known complication following subperiosteal degloving of the midfacial skeleton. The anterior surface of the maxilla and zygoma are the origin of attachment for the muscles of the upper lip, as well as the suspensory ligaments supporting the midface soft tissue. Release of these periosteal attachments therefore results in descent of the midface and cheek soft tissues. This retinacular support system starts at the periosteum and forms an arborized pattern as it connects to the superficial musculoaponeurotic system (SMAS) before finally inserting into the dermis. A histological depiction of this is shown in Fig. 3.3.7 .
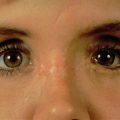
Disruption of this retinacular support network results in the appearance of premature facial aging. This may occur as a consequence of a direct traumatic injury, or as a result of operative midfacial soft tissue degloving. The characteristics of this deformity are malar volume loss, skeletonization of the lid–cheek junction with ptosis of the malar fat pad, and fullness of the nasolabial fold due to descent of the soft tissues. Midface ptosis also increases the downward pull on the eyelid increasing the risk of ectropion and scleral show following lower lid approaches. This malar volume loss occurs despite adequate bony reconstruction and was initially thought to be due to soft tissue atrophy secondary to the trauma. Without correction, the descended periosteum heals to the anterior maxillary wall in its new descended position.
Prevention of the deformity is always preferred to secondary correction. Phillips et al. described a technique for prevention of cheek ptosis following a subciliary approach in which the descended periosteum is identified and resuspended to the inferior orbital rim prior to closure. This can be done either through drill holes in the orbital rim, suture anchors or around existing hardware applied for fixation of the rim. A heavy absorbable or permanent suture is used for resuspension of the tissues. Correcting the position of the ptotic cheek tissues prevents secondary pull on the lower lid which can lead to ectropion and scleral show. The suture purchase on the cheek tissues should include enough of the deep periosteal layer to ensure transmission of the suspensory force through the retinacular network to the skin.
Established ptosis of cheek tissues is a reconstructive challenge due to retraction and scarring of the descended periosteum and shortened mimetic muscles. Success in correction is dependent on the degree to which the scarred soft tissues can be mobilized and repositioned. The surgical approach to the infraorbital rim used during the initial procedure should be determined and again implemented so as to not introduce any additional scarring in the lid. Once the infraorbital rim has been exposed, the maxilla and zygoma are degloved over their anterior surface. Full mobility of the cheek tissues is only achieved when the scarred contracted periosteum is sharply released. This release should be performed well inferior to the infraorbital nerve. The superior edge of the periosteum is grasped and placed on vertical traction ensuring there is full mobility of the cheek tissues. The mobilized periosteum is then secured with a heavy permanent suture to the infraorbital rim through drill holes, with suture anchors or around existing hardware.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree