Abstract
Secondary microvascular surgeries can be subdivided into two categories: those that are part of a planned, multistage reconstruction and those that are the result of an unsuccessful primary reconstruction. The use of multistage reconstructive plans provides vastly improved long-term aesthetic and functional outcomes for patients with complex facial injuries. In conjunction with the principles of maintaining skeletal buttress integrity and carefully replacing lost tissues in kind, planned secondary revision is a powerful tool in the reconstructive surgeon’s armamentarium. By contrast, complications requiring unplanned secondary microsurgical procedures may damage patient trust and ultimately compromise outcomes. Unplanned secondary surgeries may be the result of overly ambitious primary efforts, a failure to identify the tissues involved in the defect at the time of presentation, or devastating complications. Addressing complex craniofacial trauma patients using a disciplined and systematic approach over several operative stages will result in optimal aesthetic and functional outcomes.
Keywords
secondary microvascular reconstruction, skeletal buttress integrity, aesthetic unit principle, planned surgical revision
Background
The preoperative planning and technical execution of craniofacial microsurgical reconstruction following traumatic injury is inherently complex. Patients may present with injury to any combination of tissue layers – including skin, vasculature, muscle, nerves, and bone – and reconstructive options span the entirety of the microsurgeon’s armamentarium. These challenges are compounded in secondary reconstruction as prior reconstructive efforts may limit the surgical options available. Primary reconstruction often reduces the number of available recipient vessels and donor sites, while local tissue damaged by scarring and multiple operative attempts is more difficult to manipulate and more prone to complications, including necrosis and flap loss. An overly ambitious primary reconstruction may require an unplanned return to the operating room for secondary repair, which limits surgical options and reduces patient trust. However, when part of a pre-planned, multistaged surgical solution, secondary reconstruction can yield better functional and aesthetic outcomes than a single-stage operation.
Rodriguez et al. have previously described their approach to complex craniofacial reconstruction, which aims to optimize both functional and aesthetic outcomes. In summary:
- 1.
Aesthetic unit: the integrity of the aesthetic unit depends not only on soft tissue volume but also on adequate structural support.
- 2.
Defect boundaries: when greater than 60% of an aesthetic unit is compromised, achieving an optimal result requires extending the defect boundaries to include the entire aesthetic unit so as to minimize scar burden and create unit homogeneity.
- 3.
Tissue requirements: donor site selection must consider specific tissue deficiencies, including skin, mucosa, fat, muscle, and bone, as well as color match and volume requirements.
- 4.
Bone and soft tissue support: deficient skeletal buttresses must be reconstructed with vascularized bone so as to provide adequate long-term support to the overlying soft tissues.
- 5.
Soft tissue volume: the volume of soft tissue included in the reconstruction should be in slight excess of the base volume of the defect and should be vascularized so as to minimize resorption with the understanding that subsequent soft tissue contouring is preferable to additional tissue transfers.
- 6.
Timing: early reconstruction minimizes morbidity by condensing the periods of healing from the initial injury and from the reconstruction into one event.
- 7.
Secondary revisions: planned multistage reconstructions utilizing distant flaps for support and coverage followed by local flaps for color and contour improvement are preferable to unplanned secondary operations necessitated by complications or inadequate primary reconstruction.
All of these principles can help guide surgical decision-making when approaching the traumatic facial injury. However, the last two points – timing and secondary revisions – are particularly relevant to secondary microvascular reconstruction and will be discussed more extensively, along with the principle of tissue requirements.
Surgical Anatomy
A wide variety of free flap options exist for the secondary reconstruction of traumatic defects of the face. The advantages and disadvantages of common donor sites are summarized briefly below.
Ulnar Artery Perforator Flap
First described by Lovie et al. in 1984, the ulnar artery perforator flap provides thin, pliable tissue with a long pedicle, the ascending branch of the dorsal cutaneous branch of the ulnar artery. The donor site is less morbid than the neighboring radial forearm flap, and the skin of the ulnar forearm flap is less hirsute than its radial counterpart, making the former better suited for coverage of non-hair-bearing areas. The thinness of the flap precludes it from use in covering defects requiring bulky soft tissue or involving hardware, which can be exposed as the denervated flap thins over time. Acceptance of the ulnar artery perforator flap by the microsurgical community has been sluggish owing to concerns over potential compromise to hand perfusion and innervation, despite long-term outcome analyses failing to demonstrate significant motor, sensory, or vascular impairment associated with ulnar forearm flap harvest.
Anterolateral Thigh Flap
Since its introduction by Song et al. in 1984, the anterolateral thigh flap (ALT) has become widely accepted as a workhorse flap for head and neck reconstruction. Its versatility in size and soft tissue components, reliable anatomy based off of the lateral circumflex femoral artery, and relative ease of harvest make the ALT a popular choice for the reconstruction of a wide range of head and neck defects. Disadvantages of the flap include its tendency – particularly in obese patient populations – to be especially bulky, its susceptibility to atrophy when muscle is included, and its potential to be hair-bearing.
Groin Flap
The groin flap, based off the superficial circumflex iliac artery, holds historical significance as the first successful free flap. Advantages of the flap include its ability to be designed with muscle or as a perforator flap of variable depth and its easily concealed donor site with minimal morbidity. However, the utility of the groin flap is limited by its relatively short pedicle length and somewhat variable anatomic course.
Iliac Flap
First described by Acland in 1979, the iliac flap provides substantial bone, muscle, soft tissue, and skin fed by the deep circumflex iliac artery (DCIA) and can be used for the reconstruction of extensive defects involving multiple tissue layers. The natural curvature of the iliac can approximate the angle of the mandible for jaw reconstruction, and the presence of vascularized bone provides the long-term bony support required for osseointegrated implants. Despite these advantages, its use as a vascularized bone flap has decreased with the advent of the free fibula flap owing to the iliac flap’s higher donor site morbidity and shorter pedicle length. In cases where fibula-based reconstruction is not an option the iliac flap is a viable reconstructive modality for osseous or myo-osseous defects.
Free Fibula Flap
The free fibula flap (FFF), first described by Taylor for use in lower extremity salvage and then by Hidalgo for mandibular reconstruction, is one of the most commonly used vascularized osseous and osseocutaneous flaps. Its advantages include a substantial quantity of high-quality bone stock, its ability to include a wide range of tissues, and its flexibility in maneuvering those tissues without compromising blood supply. Disadvantages include variability in the pedicle length depending on the length of fibula being harvested and the potential for donor site morbidity including compromised ankle function, leg weakness, and great toe contracture.
Clinical Presentation
History
A meticulous assessment of the patient’s injuries and surgical history is particularly critical when evaluating for secondary reconstruction, as prior reconstructive efforts are likely to limit the flap options available to the microsurgeon. The etiology of the injury must be ascertained and the patient’s psychological state must be assessed. The patient’s surgical history must be thoroughly reviewed and an inventory kept of available donor and recipient vessels. This practice is particularly critical when the patient is presenting from a different institution or when the secondary reconstruction is unplanned. If the secondary reconstruction is in response to an unforeseen complication of the primary reconstruction, the etiology of the primary failure must be ascertained so as to prevent a similar fate for the secondary reconstruction (as could be possible if the primary flap failed due to patient hypercoagulability).
Physical Examination
The goals for secondary craniofacial reconstruction depend on the patient’s presentation and the extent of previous reconstructive efforts. Thorough, accurate physical examination is therefore critical in determining the reconstructive need. In performing the physical exam, it is helpful to follow an algorithmic approach in order to ensure complete evaluation and to resist the temptation to focus on the most immediately apparent defects. Step-wise evaluation moving from the superficial to deep tissue layers, with emphasis on the integrity of the facial coverage, lining, and support structures, can provide a straightforward system with which to approach patients.
Beginning superficially, observe the extent and location of the injury, taking note of the aesthetic unit(s) involved. Evaluate the quality of the skin, including areas of insufficient wound coverage, scarring, the positioning of hair-bearing skin, and texture or color mismatches between the local and free flap skin. Identify any contour deformities. Assess the relevant muscular function, particularly the ability to smile and close the mouth for oral defects and the ability to close the eyes for periorbital defects. Evaluate specialized structures, including the oral and nasal mucosae, ocular conjunctivae, lacrimal apparatus, teeth, and tongue. Finally, examine the integrity of the underlying bone and cartilage, particularly the skeletal buttresses, which are critical for soft tissue support. Bony structure evaluation is often greatly enhanced through the use of radiography.
Radiologic Evaluation
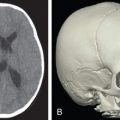
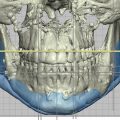
CT scans with axial, coronal, and sagittal sections using both bone and soft tissue windows are the definitive imaging modality for craniofacial defects. 3D CT scans can more precisely evaluate spatial displacement and asymmetry in volume of bony structures. Computer-assisted digital cephalometric analysis systems can be used in soft tissue volume evaluation to quantify areas of excess, deficient, or asymmetrical soft tissue distribution. Data from 3D imaging sources can be harnessed to create patient-specific implants and cutting guides, and virtual surgical planning using 3D scans from the patient can provide the surgeon with a more precise understanding of the patient’s unique anatomy and defect prior to surgery.
Surgical Indications
Firm indications for secondary microsurgical craniofacial reconstruction are difficult to state given that craniofacial injuries encompass a broad range of anatomical, functional, and aesthetic defects. Indications depend on whether the secondary surgery is part of a planned, multistage reconstruction or the result of an unplanned complication such as flap loss. In considering the need for secondary microvascular reconstruction, it is critical to take into account the overarching treatment plan as well as the tissue layers involved.
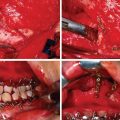
For defects involving bony tissues, reconstruction is indicated when the skeletal buttresses are compromised. Skeletal buttresses are thick areas of bone that protect delicate facial structures by absorbing impact to the facial skeleton. They are also responsible for transferring the forces of mastication to the cranial base. The skeletal buttresses can be grouped into the vertical and horizontal buttresses. The vertical skeletal buttresses are stronger and include three paired buttresses – the nasomaxillary, zygomaticomaxillary, and pterygomaxillary – as well as one unpaired midline buttress, the frontoethmoid–vomerine buttress. The weaker horizontal skeletal buttresses include the frontal bandeau, inferior orbital rim, and lower transverse maxillary. Skeletal buttress integrity is critical for maintaining facial height, width, and projection, as well as allowing the placement of osseointegrated dental implants in the maxilla or mandible, and preventing periorbital complications. In the lower two-thirds of the face, skeletal buttress integrity is critical for facilitating chewing, swallowing, oral competence, and speech. Microvascular mandibular reconstruction is indicated for mandibular defects greater than 5 cm, with flap selection dependent upon the location of the defect. Hard tissue support must be established before any soft tissue reconstruction occurs or the surgeon risks the development of severe complications, including functional incompetence and extreme contour deformities. In cases in which the primary reconstruction simply “filled the hole and closed the wound” with soft tissues, without addressing the underlying structural deformity, secondary reconstruction may involve removal of the primary soft tissue flap and replacement with a composite flap involving bone. The decision to use vascularized or nonvascularized bone is complicated by the lack of high-level scientific evidence decisively demonstrating the superiority of one technique relative to the other. Resorption rates of nonvascularized bone have been reported as high as 49.5% within 6 months, although these rates are highly variable. Several retrospective, single-institution studies have found that vascularized bone grafting yields higher bony union rates than its nonvascularized counterpart. The unpredictability of nonvascularized bone graft resorption and union rates has prompted many surgeons to use vascularized bone graft for the reconstruction of extensive defects requiring hard tissues.
For extensive soft tissue defects without hard tissue involvement, or in cases in which the primary reconstruction restored skeletal buttress integrity, secondary reconstructive efforts can focus on returning an adequate amount of soft tissue to the affected area. In these cases, the quantity of soft tissue transferred should be in slight excess of the amount lost in the defect in order to account for expected tissue resorption. Keeping in mind that muscle, particularly when denervated, atrophies almost completely and substantially more than fat and that nonvascularized tissue resorbs more extensively than vascularized tissue, vascularized fat is preferred for filling large soft tissue defects without a functional component.
Defects involving the oral or nasal mucosal linings must be repaired in order to guard against stenosis, contracture, and fistula. Muscle is preferred to skin for the reconstruction of lining surfaces due to its ability to mucosalize over time. Secondary reconstruction of mucosal surfaces is indicated in cases of insufficient or nonfunctional primary reconstruction, or as a second stage to replace skin from the primary reconstruction with mucosal tissue.
While many defects can be reconstructed with successful results, there are cases in which specialized structures, such as the eyelids or lips, prove particularly difficult to restore. These cases are often complicated by significant scarring and contracture from previous reconstructive efforts. Increasingly, vascularized composite allotransplantation of partial or full donor faces has been used to treat the most severe craniofacial defects. Traumatic injury currently accounts for the vast majority of defects warranting face transplantation, with mechanisms of injury including animal attacks, burns, and ballistic trauma. A massive undertaking on the part of both the patient and the surgical team, face transplantation is reserved for the most extensive defects that severely impact a patient’s function and quality of life despite conventional reconstructive efforts. When considering face transplantation, the benefits of recovering facial function and social reintegration must be weighed against the requirement for lifelong immunosuppression. Current face transplant efforts are limited by the difficulty of locating immunologically suitable donors. However, as immunosuppression modalities improve and donor pools are expanded, face transplantation may potentially grow into an accepted tool in the craniofacial surgeon’s armamentarium for addressing the most complex facial defects.
Surgical Technique
While specific surgical technique in secondary craniofacial reconstruction is largely a function of surgeon preference, there are several key principles that can help guide decision-making to optimize patient aesthetic and functional outcomes. Replacing lost tissues “in kind” as described by Gillies is an age-old reconstructive principle that holds true in the setting of secondary microsurgical reconstruction, and involves identifying discrepancies between the tissues incriminated in the original defect and those used in the primary reconstruction, with emphasis on coverage, lining, and support. For example, a patient presenting with a full-thickness forehead defect involving both hard and soft tissue may have been primarily treated with a flap including only soft tissue. This “filling the hole” approach to primary reconstruction does not adequately restore the underlying skeletal structure and thus requires secondary reconstruction with a flap involving hard tissue in order to restore normal contour and provide adequate soft tissue support. Replacing like with like is a guiding principle in secondary skin revision as well. Local advancement and transposition flaps are useful in covering de-epithelized free flaps with skin that more closely approximates the color and texture of the surrounding areas.
In deciding on the timing of a reconstruction, effort should always be made to begin the reconstructive process within the window of healing from the initial injury. This approach prevents the primary wound from healing extensively prior to reconstruction, thereby obviating the need to extend the borders of the defect by removing scar tissue when preparing the recipient site. Condensing the healing periods from the injury and reconstruction into one early phase significantly improves patient outcomes by reducing fibrosis and contracture. Capitalizing on this opportunity for better results is only possible when the reconstructive stages are carefully planned shortly after the time of injury, such that primary wound closure and stabilization is followed by secondary reconstruction of the bony support and restoration of soft tissue deficits within the initial window of injury. Delaying decisive reconstruction risks the patient being lost to follow-up or developing severe complications including soft tissue collapse, stenosis, and fistula formation while awaiting secondary reconstruction.
Early restoration of critical bony and soft tissue elements can then be followed by planned secondary or tertiary revisionary procedures to improve soft tissue contour and color match. Intentionally oversized flaps can be debulked once soft tissue atrophy has occurred and distant flaps can be resurfaced with local skin advancement to reduce scar burden and improve the aesthetic result. Approaching the reconstruction as a multistage process rather than a single procedure reduces complications from overly ambitious surgeries and allows for better long-term outcomes. Unplanned salvage operations necessitated by severe complications damage patient trust and impair long-term aesthetic and functional results. By contrast, a clearly communicated and expertly executed reconstructive plan improves both the patient’s long-term outcomes and psychological experience.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree