Abstract
Secondary enophthalmos and diplopia following suboptimal primary orbital reconstruction lead to aesthetic and functional deficits, respectively. An increase in the orbital volume as small as 2.1–2.3 mL has been shown to result in clinically significant globe malposition. Large combined medial wall and orbital floor fractures, particularly ones with compromised inferonasal bony strut support, carry the highest risk of requiring secondary reconstruction. Symptoms may present in a delayed fashion, as a poorly fixed implant becomes displaced or changes in orbital anatomy after trauma are not considered properly.
Persistent diplopia following orbital fracture repair may be broadly categorized into restrictive or paralytic etiologies. A thorough patient history and physical examination are critical in differentiating the two. In particular, a strabismus evaluation by an extraocular muscle specialist is imperative. Recent thin-cut computed tomography imaging of the face may offer further insight into the etiology of the patient findings. Anatomical causes of symptoms result in restrictive strabismus and most often improve with revision surgery, whereas paralytic diplopia secondary to neuromuscular injury will not benefit from reoperation.
Restrictive diplopia or enophthalmos greater than 2 mm are generally an indication for revision surgery. Most surgeons perform secondary orbital reconstruction approximately 3–6 months following initial repair. The surgical considerations and approaches relevant in primary orbital reconstruction apply to secondary surgery. Given tissue changes following orbital trauma, secondary orbital reconstruction has traditionally been seen as extremely challenging. New technologies, such as intraoperative navigation and computer-assisted surgery, have proven invaluable in optimizing patient outcomes following revision orbital surgery.
Keywords
computer assisted surgery (CAS), intraoperative navigation, orbital reconstruction
Background
Persistent enophthalmos and diplopia following primary orbital reconstruction lead to unsatisfactory aesthetic and functional outcomes, respectively. Diplopia after surgical repair of orbital fractures has been reported in 8%–52% of patients, while clinically significant enophthalmos has been reported in 27%. Large combined medial wall and orbital floor fractures tend to carry a higher risk of enophthalmos, particularly if the fracture compromised the inferonasal bony strut support. Secondary orbital reconstruction has traditionally been seen as extremely challenging, and keys to success are careful preoperative planning, appropriate imaging and identifying which patients are most likely to benefit from surgical treatment.
Changes in orbital volume as small as 2.1–2.3 mL have been shown to result in clinically significant globe malposition. Suboptimal reduction of orbital fractures in the acute setting results in increased orbital volume and in both secondary enophthalmos and diplopia. Symptoms may present in a delayed fashion, as a poorly fixed implant becomes displaced or changes in orbital anatomy after trauma are not considered properly. For instance, if the posterior bony orbit remodels following trauma, the retrobulbar volume may increase; the orbital fat is frequently repositioned posteriorly, further compromising support of the globe. Such anatomic reasons for globe malposition tend not to improve, and frequently worsen, over time following inadequate primary reconstruction.
Persistent diplopia following orbital fracture repair may be broadly categorized into either restrictive or paralytic etiologies. Poor primary reduction may lead to increased orbital volume and muscle impingement. Developing adhesions surrounding the implant and implant displacement may also restrict muscle movement. Such anatomic causes of symptoms result in restrictive strabismus and most often improve with revision surgery. Conversely, paralytic diplopia secondary to neuromuscular injury will not benefit from reoperation, although symptoms tend to improve over time with conservative management which depends upon nerve regeneration.
Clinical Presentation
Patient history and physical examination should include all details relevant to any orbital trauma patient. Specific details pertinent in secondary reconstruction include a thorough understanding of prior reconstructive procedures, and a timeline of presentation of symptoms. Operative reports should be obtained if possible, detailing surgical approaches, incision choice, and implant type. An understanding of how symptoms have been changing over time may lend clues to the etiology. Diplopia that has been improving very slowly suggests neurogenic causes; new onset symptoms that have worsened since primary repair are more suggestive of a restrictive process.
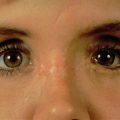
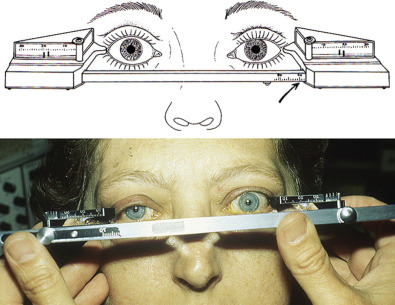
Compared to patients with acute orbital trauma, those who have undergone prior repair warrant a more thorough eye examination. Patients should be referred for strabismus evaluation by an extraocular muscle specialist. A detailed extraocular motion exam may help differentiate between restrictive and paralytic symptoms, as the latter is unlikely to improve with repeat surgery. Additionally, accurate measurements of the relative position of orbital rims, lateral, and medial canthi, and pupils should be taken. An exophthalmometer may be used to measure globe position relative to the lateral orbital rim, or the ear canal if necessary ( Fig. 3.7.1 ).
Radiological Evaluation
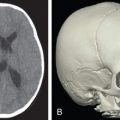
Recent computed tomography imaging should be available as part of any secondary reconstruction evaluation. As in acute trauma, thin-cut computed tomography (CT) imaging of the face is the gold standard for evaluating the bony orbit. In patients with contraindications to CT imaging, MRI has been used to assess orbital volume and anatomy. Repeat imaging should be compared to prior studies and focus on identifying abnormalities amenable to surgical revision. Orbital volume should be assessed and compared to the contralateral uninjured side if possible. Any implant displacement or impingement, herniation of orbital contents, or muscle entrapment should be noted.
Surgical Indications
Persistent symptoms, including diplopia, enophthalmos, or disfigurement, attributable to an anatomical surgically correctable defect generally warrant repair. Most surgeons consider enophthalmos greater than 2 mm as clinically significant and an indication for surgery. An observer will generally not notice globe asymmetry of 1–2 mm. Restrictive diplopia caused by muscle impingement, or enophthalmos in the setting of orbital content herniation or implant displacement are likewise reasons for reoperation. Care must be taken to rule out paralytic diplopia unlikely to improve with surgery.
Another consideration in planning revision orbital surgery is that of best timing. Postponing nonemergent reoperation for several months affords tissue time to heal and symptoms to stabilize. On the other hand, waiting more than one year after initial repair may result in significant bone remodeling and scarring that could complicate reoperation. Many surgeons choose to wait 3–6 months before undertaking any elective orbital revision. Acute symptoms suggestive of nerve or muscle impingement, implant displacement, or orbital compartment syndrome warrant immediate intervention, as they would following primary injury.
Surgical Techniques
Surgical approaches used in primary orbital reconstruction apply in secondary procedures. Transconjunctival, subciliary, or transcutaneous incisions may all be used; scarring and aberrant anatomy may make reoperation more challenging, but the surgical approach may be chosen independently of that used in primary repair. Some surgeons have even employed endoscopic approaches, although success is highly dependent on experience in endoscopic surgery. Most importantly, the techniques employed in revision orbital reconstruction should be tailored to the specific preoperative diagnosis and mechanism behind symptoms. All prior orbital implants should be removed, and replaced with pre-planned devices. Care must be taken as radiolucent implants may be difficult to visualize on preoperative imaging and intraoperatively. Retained implant material should be suspected if a prefabricated implant or virtual planning results do not correspond to intraoperative findings. Some surgeons avoid placement of radiolucent orbital implants partly because of challenges during any revision procedures.
Computer-assisted surgical (CAS) planning is essential for precise secondary orbital reconstruction. Recent axial orbital CT scan data should be available for virtual planning. Software mirrors the intricate orbital wall anatomy of the uninjured side onto the symptomatic orbit. Any deficits and asymmetries may readily be seen by superimposing the contralateral template. Although preformed implants are generally not recommended in secondary reconstruction, virtual planning software may aid in planning cuts and position of fixation screws if necessary. Use of CAS has been shown to improve accuracy of bony reconstruction and reduce the need for bony revision surgery.
Patient-specific implant fabrication has advanced greatly in recent years and is imperative in secondary orbital reconstruction ( Fig. 3.7.2 ). Accurate restoration of the intricate anatomy of the orbital walls is critical to success, but is greatly complicated by prior attempts at repair and delayed timeframe. Further, the non-emergent nature of the surgery affords time for careful implant design ( Fig. 3.7.3 ). Many surgeons exclusively employ custom implants in cases of secondary reconstruction. The technology has been reported to achieve orbital volumes comparable to the contralateral side with favorable long-term outcomes. Notably, intraoperative overcorrection of enophthalmos by an average 2.7 mm is necessary for satisfactory results.