A more complete understanding of the relationship between clinical scar appearance and the corresponding histology over time may help guide management and the evaluation of the treatment response.
Additional noninvasive technologies, such as reflectance confocal microscopy, may aid in the microscopic evaluation of scars.
The morphology of a scar is classified into its physical characteristics, appearance, and symptoms.
Scar assessment scales have been developed and implemented for the objective analysis of scars. Each scale differs from the next and may only be applicable to a unique set of scars.
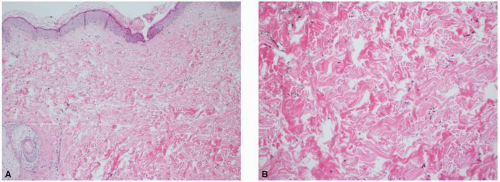
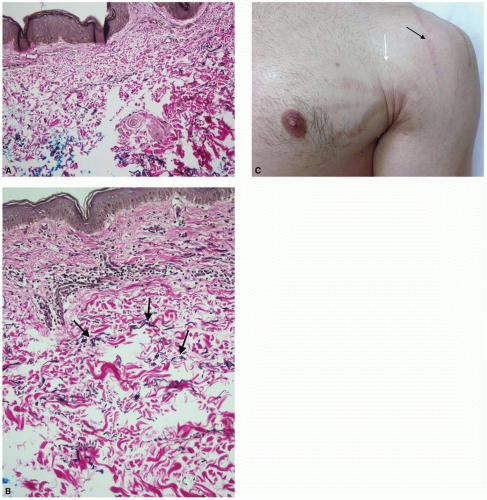
microvessels in hypertrophic and keloidal scars in comparison to normal scars and normal dermis because of endothelial cell swelling and increased endothelial cell density. Additionally the degree of vascular occlusion was higher in keloids with fewer and more flattened vessels, especially centrally within the lesion, suggesting less vascular supply in a keloid as compared with a hypertrophic scar. The growth of fibroblasts and increased collagen deposition may result in this occlusion.7,8 On the contrary, other studies have demonstrated increased vasculature of keloids and hypertrophic scars in comparison with normal scars and normal skin.1 Amadeu et al.4 reported that the volume occupied by vessels in the papillary dermis was 79.7% higher in hypertrophic scars (P < 0.01) and 62.5% higher in keloids (P < 0.05) as compared with normal skin. Similar findings were noted in the reticular dermis, with hypertrophic scars having 62.9% higher vessel volume (P < 0.025) and keloids having 68.5% higher vessel volume than normal skin (P < 0.001). The benefit of pressure dressings and vascular lasers for the treatment of these pathologic scars contradicts our understating of the role of hypoxia in their pathogenesis. At this point, further research needs to be conducted to fully understand this interplay.
Table 5-1 Flowchart Describing the Histologic Maturation of a Scar Over Time | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||
S100B, HMB45, and α-melanocyte-stimulating hormone (α-MSH) for comparison. They discovered no statistically significant difference in melanocyte number between hyperpigmented and hypopigmented scars and uninjured skin samples. There was, however, a statistically significant difference in the amount of melanin and α-MSH, and immunohistochemical evidence of stimulated melanocytes in hyperpigmented versus hypopigmented scars.
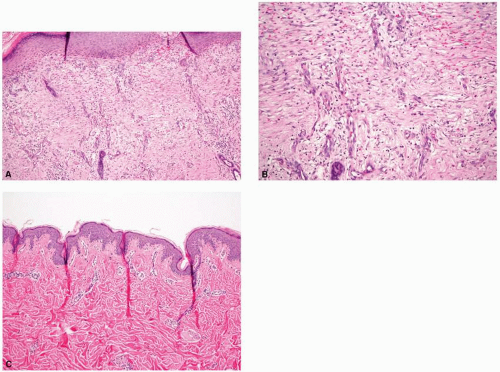
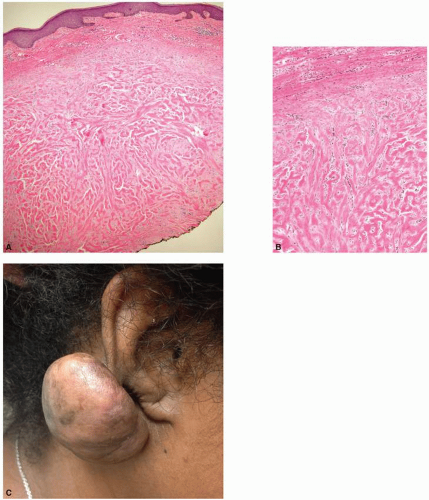
in collagen typing between laser-treated and -untreated burn scars. It is well described that normal skin contains a combination of type I and type III collagen; fetal skin contains a higher proportion of type III collagen, and burn scars contain a higher proportion of type I to type III collagen. A course of three ablative fractionated CO2 laser treatments to burn scars eventuated in a collagen profile approaching that of normal skin, with a posttreatment increase in type III collagen as demonstrated by the Herovici stain (Fig. 5-6). Additionally, Taudorf et al.15 found statistically significant clinical improvement in various scar types (normal, hypertrophic, and atrophic) after three monthly nonablative fractional laser treatments (P < 0.0001 vs. untreated), with corresponding histology indicative of collagen remodeling. There is a predominance of thickened collagen within a scar as compared with the normal surrounding dermis. The healing process that follows fractionated photothermal injury ultimately leads to the remodeling and reorganization of collagen that begins to approach that of normal skin. A later study completed by Connolly et al.16 discovered that, counterintuitively, treatment of erythematous burn scars with a fractionated CO2 laser led to a statistically significant increase in vascular density as determined by anti-CD31 immunostaining, despite a decrease in clinically apparent erythema during the treatment course (Fig. 5-7). These histologic findings as illustrated above have both supported and challenged our understanding of the mechanisms leading to clinical improvement in scars. Perhaps we may use these predicted and unforeseen findings to further propel scientific discovery and improved management.
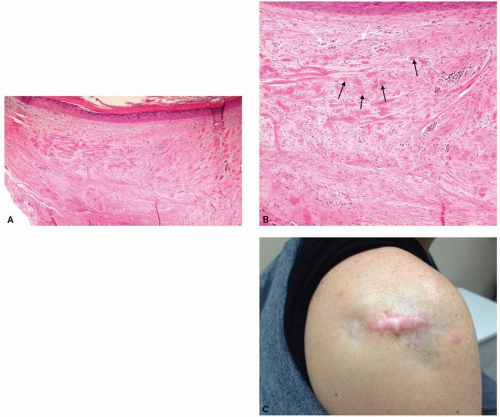
resolution to classical histology, without tissue damage (Fig. 5-8). Additionally, it may be used to detect the dynamic microscopic changes of a particular skin lesion over time. RCM may provide promising in vivo evaluation and comparison of many scar types, and may also be used for the pre- and posttreatment analyses of scars, among other dermatologic conditions.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree