Introduction
The surgical treatment of breast cancer has undergone a paradigm shift and evolved from the Halsted radical mastectomy to the simple mastectomy and now includes breast conservation therapy (BCT). BCT is defined as local excision of the primary tumor (i.e., lumpectomy, quadrantectomy) or breast-conserving surgery (BCS) followed by radiation therapy (RT). BCT was ushered into standard of care by six large randomized prospective trials demonstrating equivalent survival rates with BCT compared with mastectomy in early disease. RT has been shown to be a critical component of the treatment regimen by reducing the recurrence rate by 50% and reducing breast cancer deaths by 16% after BCS. The goals of therapy with BCT are tumor eradication, prolonging survival, and maximizing quality of life with oncoplastic strategies.
Biological Basis of Radiation in Treatment of Breast Cancer
RT is defined as the delivery of ionizing energy to control malignancy while limiting damage to surrounding normal tissues. Two major methods of radiation delivery are available: (1) External Beam Radiation Therapy (EBRT) and (2) Brachytherapy. EBRT delivers high-energy photon or electron x-ray beams to tissue from outside the body. Brachytherapy delivers lower energy radiation within the patient’s body to treat the volume of tissue in the immediate vicinity.
Ionizing radiation as a therapeutic modality underwent rapid growth in the early part of the 20th century when Regaud demonstrated that certain internal reproductive cells may be targeted without causing major burns to the skin. The overarching goal is to optimize the “therapeutic ratio,” a risk benefit analysis of healthy versus cancerous tissue, when planning a radiotherapy regimen. The effect of radiation is known as the continuum , which describes the sequence of events that occur when ionizing energy is directed at a cell. Strong circumstantial evidence suggests cellular damage is a result of direct action of charged particles or free radical formation leading to DNA damage. At the later end of the continuum, reproductive integrity is lost when DNA is unrepairable or mis-rejoined, leading to cell death over hours to years. Tumor control is achieved when clonogenic cells are destroyed or otherwise unable to maintain growth.
Based on the theoretical radiobiological modeling of the dose response of normal tissue compared with cancer cells, conventional fractionation divides the total radiation into several smaller doses over a period of several days to impart less toxic side effects on late responding healthy cells. Cellular damage is regulated by the 4Rs of radiotherapy: r epair of sublethal damage between dose fractions, r eassortment of cells into more sensitive stages of the cell cycle, r eoxygenation of tumor cells for increased sensitivity, and r epopulation of the surviving fraction due to cell division.
Radiation in Breast Conservation Therapy and Invasive Ductal Carcinoma
BCT has become an alternative to mastectomy for most patients with early stage invasive ductal carcinoma. This is possible with modern multimodal patient selection and treatment. Indications for BCT are as follows ( Table 20.1 ) :
| Indications | Contraindications |
|---|---|
| <5 cm tumor | Inability to achieve negative margins ∗ |
| Unifocal disease | Prior chest irradiation ∗ |
| Patient preference and compliance | Poor aesthetic outcome ∗ |
| Patients with comorbidities | Inflammatory breast cancer ∗ |
| Pregnancy | |
| Collagen vascular disease | |
| Prior breast augmentation |
Early Stage Tumor/Tumor Size
The majority of trials demonstrating equivalent outcomes to mastectomy had upper size limit of 2–4 cm, whereas others permitted an upper limit of 5 cm. In tumors >5 cm or stage III, neoadjuvant chemotherapy should be considered before BCS. Older trials indicate local failure rate was significantly higher than those initially candidates for BCT (14.5% vs 6.9%). With improved patient selection and coordination among specialties, the locoregional control has been shown to be equal in those who do and do not receive neoadjuvant chemotherapy.
Unifocal Disease
Historically, multifocal (two or more tumors in the same quadrant) and multicentric (two or more tumors in the separate quadrant or 4–5 cm apart) disease has been considered a contraindication to BCT, with historical ipsilateral breast tumor recurrence (IBTR) rates of 20–40%. However, studies with carefully selected patients demonstrate IBTR rates at 10 years are comparable to mastectomy. These tumors tended to be multifocal, smaller (≤1 cm), without extensive ductal carcinoma in-situ (DCIS), and in older women. Clear margins are paramount, as positive margins and low-grade tumors were the strongest predictors of IBTR (see “Inability to Achieve Clear Margins”).
Patient Preference/Compliance
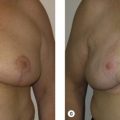
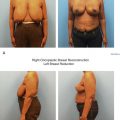
Patients presenting for BCT must be motivated to preserve the breast and be willing to adhere to the radiotherapy regimen to follow. Historically, no difference was seen in psychological adjustment between BCT and mastectomy, but improved body image and sexual function were seen in BCT cohorts. A recent study with the BreastQ questionnaire found breast conservation without reconstruction to be associated with lower physical well-being in the chest area, sexual well-being, and overall satisfaction compared with mastectomy and reconstruction, possibly due to resulting asymmetry and radiation effect ( Fig. 20.1 ). The addition of oncoplastic breast reconstruction results in high level of long-term satisfaction, improved quality of life, and self-esteem. Patients should be offered reconstruction when available and necessary (see “Inadequate Aesthetic Outcome”).
Certain situations make BCT untenable due to the consequences associated with radiation. The conditions are as follows (Table 1):
Inability to Achieve Clear Margins
Margins, based on the recommendations of consensus statements, are defined as invasive cancer on inked margins, the presence of which results in a twofold to threefold increase in IBTR ( Table 20.2 ). This increase is not nullified by a boost of radiation, systemic therapy, or favorable biology. So long as negative margins are achieved, consensus statements agree that routinely obtaining wider margins does not necessarily result in decreased risk of IBTR. However, when close margins present in younger patients with extensive intraductal component (EIC), re-excision may be beneficial. Tumor distribution, shape, or proximity to the chest wall may preclude the ability to achieve negative margins. The likelihood of this occurrence is increased with multicentric disease. Persistent positive margin after reasonable surgical attempts may be an indication to convert to a mastectomy.
| Society | Position |
|---|---|
| American College of Radiology (2015) |
|
| |
| American Society of Breast Surgeons (2013) |
|
| Society of Surgical Oncology/American Society for Radiation Oncology (2014) |
|
| National Comprehensive Cancer Network (2015) |
|
| American Society of Clinical Oncology (2014) |
|
Further differentiation can be made between extensively positive and focally positive margins, which is defined as tumor involvement in three or fewer low-power microscopic fields. The risk of 8-year IBTR among excised tumors with focally positive margins was only marginally increased and further mitigated by systemic therapy. These patients may still be candidates for BCT. Clinical consideration of re-excision and additional adjuvant systematic therapy is warranted if focal margin involvement is present.
Pregnancy
WBI should be avoided in pregnant women due to the risk of mutagenesis to the developing embryo. Application of accelerated partial breast irradiation (APBI) is still controversial and not routinely performed. Oftentimes, the pregnancy delays diagnosis of breast cancer, and patients present with larger tumors necessitating mastectomy. However, should the tumor characters allow BCT and the pregnancy allows for excision, radiation may be delayed until after delivery (see “Timing”).
Prior Chest Irradiation
Patients with a prior history of irradiation (breast cancer, Hodgkin’s lymphoma) are generally ineligible for BCT as total tolerable dose of the previously irradiated tissue will likely be exceeded. In such scenarios, the standard of care is mastectomy; however, case reports have been described with APBI in patients refusing mastectomy.
Collagen Vascular Disease
Many radiation oncologists will not treat patients with collagen vascular disease. There is an increased incidence in severe acute toxicity with RT delivered to the breast, presenting as severe desquamation. Coordination of radiation oncology and rheumatology is necessary should BCT be pursued.
Inadequate Aesthetic Outcome
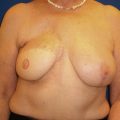
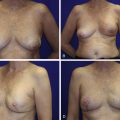
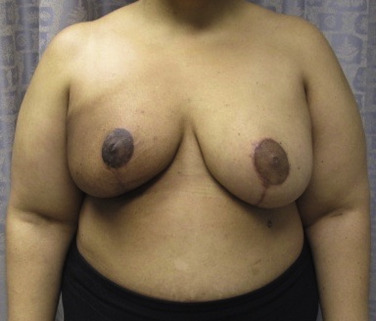
Patients with micromastia and a large tumor present a unique surgical challenge for BCT. Lack of reconstruction after BCT results in distortion, asymmetry, and lower physical and sexual well-being. Unfortunately, women with micromastia may be automatically triaged to mastectomy without full understanding of the options. Oncoplastic techniques have been described that combine volume displacement and replacement in a single procedure. Plastic surgery consultation should be considered when >10% breast volume loss is anticipated. Techniques for volume replacement include glandular tissue rearrangements, biplanar implant placement, and local and free flaps ( Fig. 20.2 ). These reconstructions mitigate volume loss and have been shown to result in high levels of patient satisfaction and improved quality of life. Consultation with plastic surgery should also be pursued with a subareolar position of the tumor or when the resulting scar is in a poor orientation.
Prior Breast Augmentation
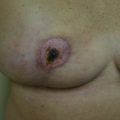
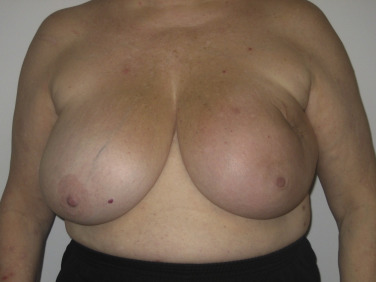
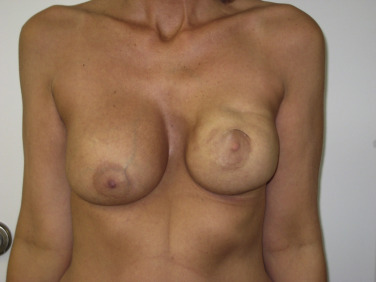
Prior breast augmentation has typically precluded BCT due to poor cosmetic outcome. Nahabedian et al in a retrospective review of 48 cases found patients with prior augmentation are more likely to undergo mastectomy (72.9%) compared with BCT (57%) with significantly lower rates of BCT (25.5% vs 43%), despite comparable disease stage. Tumors in augmented patients were more likely detected when smaller in diameter but less likely to be detected on screening mammography. Studies have demonstrated increased complications following BCT in the setting of prior breast augmentation, including erosion, pain, and capsular contracture that range from 30–65%, despite optimized radiation dosimetry ( Fig. 20.3 ). Up to half of the patients required implant removal demonstrating that, even with modern radiotherapeutic techniques, the incidence of capsular contracture and poor cosmesis remains high.
The following conditions require special consideration:
Age
Although age is not a contraindication in and of itself, a meta-analysis of trials indicates that, of women who underwent BCT, those who were <40 years old had a 5.9% per year chance of IBTR, a 2.7% per year for 40–49 years, a 1.9% per year for 50–59 years, a 1.6% for 60–69 years, and 1.0% for ≥70 years. Locoregional recurrence and mortality are similarly high in mastectomy among younger women. This may be in part due to the higher incidence of adverse tumor biology. However, large cohort studies indicate improved survival may be seen in women >50 years and those with comorbidity not treated with chemotherapy when comparing BCT to mastectomy.
Nodes
Patients with positive axillary nodes do not have an increased incidence of IBTR compared with patients with positive nodes. In fact, a large cohort study found benefit of BCT for T2N1 versus T2N0, perhaps due to the use of systemic therapy for node-positive patients. Patients with extracapsular node involvement do not have an increased risk of IBTR.
Extensive Intraductal Component
Patients with EIC warrant careful consideration. EIC is defined as intraductal carcinoma (DCIS) prominently present within the borders of the primary tumor (when DCIS comprises 25% or more of the tumor or present within all visualized ducts) and when intraductal carcinoma is present in adjacent tissue, either as an extension beyond the infiltrating margin or as separate foci in grossly normal adjacent tissue. Negative margins are difficult to achieve with diffuse microcalcifications, despite en bloc incisions. So long as positive margins are achieved, IBTR are mitigated in tumors with EIC. Postoperative mammography can be helpful in identifying residual calcifications for re-excision.
Large Breasts
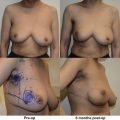
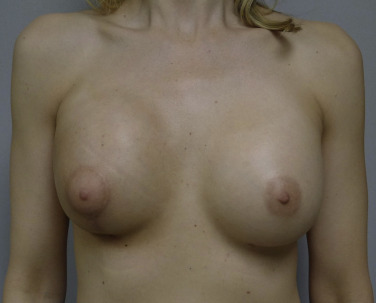
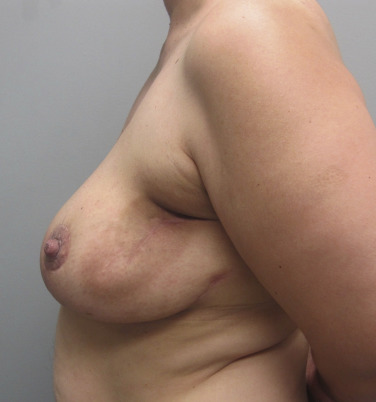
BCT in large breasts has been demonstrated to have significantly inferior cosmetic result compared with small breasts due to the larger skin folds resulting in dose inhomogeneity and increased skin toxicity. Large-breasted women were found to have late radiation changes in 39% of cases, compared with 6% in women with small breasts. However, this does not preclude large-breasted women from receiving BCT. An oncoplastic therapeutic mammaplasty may be performed in conjunction with adjuvant radiation to improve cosmetic outcome ( Fig. 20.4 ).
Whole Breast Irradiation
External Beam Radiation Therapy (Whole Breast Irradiation)
The diversity of clinical indications for breast radiotherapy contributes to the diversity in the characteristics of EBRT. Electron EBRT travels a finite depth within the soft tissue, and surface dose increases with increasing electron energy. As such, electron EBRT is useful for superficial treatment of chest wall and internal mammary nodes or when a limited area surrounding a defect requires a boost dose after WBI. Photon beams penetrate greater depths, with theoretical less total radiation dose to the skin. The majority of patients receiving EBRT are treated with tangential photon fields. Cobalt machines delivering photons were historically the main modality in which many of the initial trials were based on. Disadvantages include less precisely defined beam edge irradiating surrounding tissue (penumbra), poor delivery to deeper tissues, and staff exposure. Over the past 25 years, linear accelerators (LINACS) have replaced cobalt machines due to the rates of secondary malignancies and heart disease. LINACS, with modern modifications, are designed to deliver precise doses of radiation in a wide range of beam energies with limited personnel exposure.
Timing after Surgery
The optimal timing of RT after surgery is unknown, although 6–8 weeks is the generally agreed upon upper limit. Huang et al in a systematic review found 1.62 higher odds of IBTR when radiotherapy was administered >8 weeks after surgery. The absolute increase in the risk of IBTR is 1.0% per month delay in starting radiotherapy. Delays >3 months are associated with increased mortality. When chemotherapy is required, no difference in IBTR or survival has been found if WBI occurs before or after treatment, so long as RT is begun within 7 months from surgery. Staggering treatment is preferred over concurrent therapies to reduce toxic effect.
Radiation Dosage: Standard Fractionation versus Hypofractionation
The schedule for standard fractionated WBI, daily doses of 2 Gy in 5 weeks for a total dose of 45–50 Gy, have been established by previous trials ( Table 20.3 ). Radiation assists in locoregional control of the disease, eliminating potential microdisease, reducing the IBTR rates such that it is equivalent to mastectomy. The rationale for delivery of such dosages is based on dogma that has suggested normal tissue was more sensitive to larger fraction size. Therefore, smaller daily fractions induce irreparable damage to malignant tumor DNA (which typically lack repair mechanisms) with less collateral injury to surrounding tissues.
| Whole Breast | |
| Irradiation | 45–50 Gy over 5 weeks (5 per week, 1.8–2.0 Gy each session) |
| Boost | 60–65 Gy over 7 weeks (5 per week, 1.8–2.0 Gy each session) |
| Hypofractionation | 40–42 Gy over 3 weeks (5 per week, 2.6–2.8 Gy each session) |
| 3D Conformal | 35–38.5 Gy over 10 fractions, twice daily |
| IMRT | 40 Gy over 15 fractions |
Studies with >10 year follow-up have suggested that breast cancer cells have similar dose response curve rates to radiation when compared with native tissue. Delivery of fewer, larger fractions, also known as hypofractionated-WBI (hWBI), is as effective in tumor control with similar normal tissue damage and cosmetic outcome (see Table 20.3 ). The updated American Society for Radiation Oncology (ASTRO) 2018 guidelines now specify hWBI to be appropriate in any age, stage, and with chemotherapy.
Radiation Boost
Additional radiation treatment or a “boost” delivered to the lumpectomy excision site after WBI has been shown in EORTC randomized control trials to decrease the risk of IBTR, but not mortality, through 20 year follow-up compared with WBI alone. These effects were most pronounced in women ≤40 years, who are at greater risk for IBTR. Boost did not modify mortality risk in these patients. However, radiation boost increased the incidence of severe breast fibrosis by fivefold compared with patients who did not receive a boost. This trial did not assess margin status. Although patients with close margins may benefit from increased doses, the literature and consensus statements are mixed in this regard.
Partial Breast Irradiation
Broadly speaking, partial breast irradiation may be delivered as APBI or intraoperative radiation therapy (IORT).
Accelerated Partial Breast Irradiation
APBI focuses radiation to 1–2 cm of tissue surrounding tumor beds considered to be high risk and is directed to where IBTR usually occurs ( Fig. 20.5 ). APBI may be delivered with brachytherapy or external beam radiation. Brachytherapy requires specific equipment and infrastructure. Interstitial therapy is a complex technique where multiple rows of catheters are left protruding out of the skin for 1 week. Delivery of radiation doses requires dedicated suites to protect personnel. Applicator-based brachytherapy relies in commercially available device to deliver radiation. This device may be placed intraoperatively or as a second procedure. Multilane applicators have permitted varying doses to reduce radiation exposure to normal tissue. Studies, although promising, are limited in size and design.
3D Conformal Therapy
3D conformal radiation therapy (3D-CRT) represents the first of conforming techniques, referring to radiotherapy that is able to “conform” to the target shapes and potentially delivering complex dosages within targets while minimizing inadvertent damage to normal tissue ( Table 20.4 ). In this forward-planning technique, treatment parameters (number of beams, intensity, shape, angle) are decided, and the resulting dose distribution is calculated and evaluated. This technique allows for improved spatial distribution of dose, but normal tissues may not be completely excluded. Treatment dose is delivered over the course of 5–7 days in 10 fractions that are delivered twice a day with at least 6 hours between each course for a total dosage of 35–38.5 Gy. Although smaller studies have shown positive results, the RAPID trial, randomizing 2135 women with ≤3 cm breast cancer or DCIS to 3D-CRT or WBI, demonstrated poor aesthetic outcome at 3 years based on interim examination of results. The delayed development of fibrosis, fat necrosis, and poor aesthetic outcome were corroborated by smaller studies. This likely indicates the subtle variations in planning techniques and dose constraints result in substantial differences in outcome.