Abstract
The venous system of the lower extremities is a complex, interconnected network consisting of large- and small-caliber vessels. Incompetence within this system manifests clinically as visible telangiectasias, reticular veins, and varicosities. As the severity increases, other cutaneous findings such as edema, dermatitis, lipodermatosclerosis, and ulceration may become evident. Diagnostic evaluation of patients with venous insufficiency includes assessment of the saphenofemoral junction (SFJ) by duplex ultrasonography. In the setting of telangiectasias, reticular veins, and varicosities without SFJ incompetence, routine sclerotherapy +/− ambulatory phlebectomy is indicated. When SFJ incompetence is detected, minimally invasive procedures such as endovenous radiofrequency or laser ablation are the criterion standard therapeutic modalities and can be performed utilizing local tumescent anesthesia. Newer techniques such as polidocanol microfoam injection and cyanoacrylate adhesive closure (CAC) can be done without the requirement for tumescent anesthesia. The dermatologic surgeon can now successfully treat incompetent leg veins of any size, providing both medical and cosmetic benefits to patients.
Keywords
venous anatomy, venous disease of the leg, phlebology, telangiectasias, reticular veins, varicose veins, superficial and deep venous systems, sclerotherapy, ambulatory phlebectomy, endovenous laser and radiofrequency ablation
- ▪
Superficial telangiectasias, reticular veins, and varicose veins of the lower extremities are interconnected and develop after impairment of venous return
- ▪
Poor venous return results from venous valvular incompetence or primary muscle pump failure
- ▪
A pretreatment physical examination to assess the extent and cause of the venous abnormalities should be performed with the patient in a standing position
- ▪
The physical examination should be supplemented by duplex ultrasonography when saphenous vein reflux is suspected
- ▪
Both preoperative and postoperative compression are important in the management of venous insufficiency
- ▪
Sclerosing agents should destroy the entire vascular wall, producing permanent fibrosis of the vessel
- ▪
The minimal concentration and volume of sclerosing agent should be used to achieve the desired results
- ▪
All of the affected portions of the superficial venous system should be treated, starting with the most proximal
- ▪
Large veins should be treated before smaller veins, and veins should be treated from proximal to distal
- ▪
FDA-approved sclerosing agents include sodium tetradecyl sulfate and polidocanol, both liquid and microfoam formulations
- ▪
Hypertonic saline (11.7–23.4%) and glycerin (72% mixed 2 : 1 with 1% lidocaine, with or without epinephrine) are not FDA-approved for vein sclerosis, making their usage off-label
- ▪
The most common local side effects following injection of sclerosing agents are hyperpigmentation and telangiectatic matting and less often cutaneous ulcers
- ▪
Patients generally require one to three sclerosant injection treatment sessions, spaced at 6- to 8-week intervals
- ▪
Utilizing tumescent anesthesia, incompetent saphenous veins and large tributaries over 4 mm in diameter can be effectively treated by: ambulatory phlebectomy; radiofrequency or laser thermal ablation; or cyanoacrylate adhesive closure
Introduction
Varicose and telangiectatic leg veins increase in extent and severity with increasing age. The evolution of safe and effective treatment options has made sclerotherapy, ambulatory phlebectomy, and endovenous laser ablation (LA) and radiofrequency (RF) ablation expanding areas within dermatologic surgery. The utilization of improved diagnostic modalities, more efficient sclerosing agents, and simplified surgical techniques has rendered the treatment of venous disease safe, efficient, and highly satisfying for both physician and patient. This chapter presents the most recent advances in sclerotherapy, ambulatory phlebectomy, endovenous LA and RF ablation, and cyanoacrylate adhesive closure (CAC) as they relate to the management of telangiectasias, reticular veins and varicose veins, both non-saphenous and saphenous.
Venous Anatomy, Physiology and Pathophysiology
The peripheral venous system functions both as a reservoir to store extra blood (e.g. during pregnancy) and as a conduit to return blood from the periphery to the heart and lungs. The correct function of the venous system depends on a complex series of pumps and valves that are individually frail, yet the system as a whole performs well under extremely adverse conditions ( Fig. 155.1 ). Superficial veins carry less than 5% of venous blood, while the deep venous system carries 95% of the venous blood.
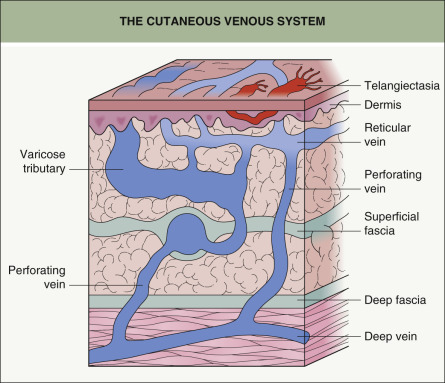
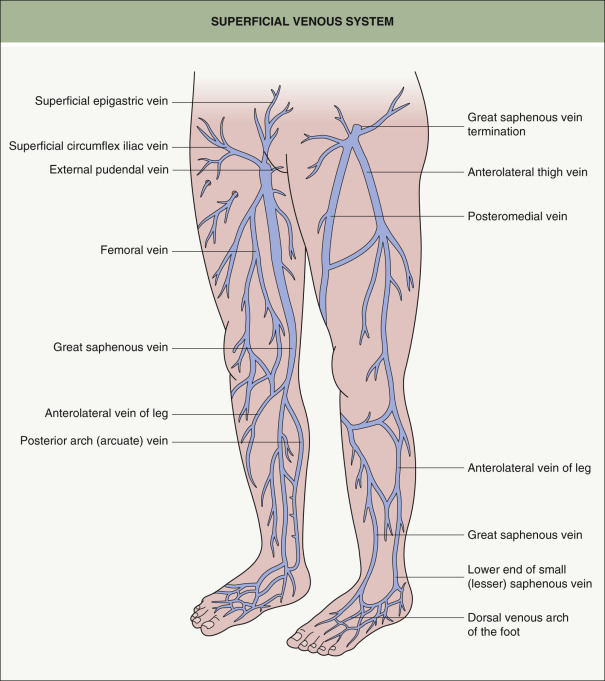
The superficial venous system is a complicated, variable network of interconnecting veins . A few of the larger truncal superficial veins are fairly constant in location. These truncal superficial veins serve as a conduit to pass blood centrally into the deep venous system. The two principal named superficial veins are the small saphenous vein (SSV), which runs from the ankle to the knee, and the great saphenous vein (GSV), which runs from the ankle to the groin ( Fig. 155.2 ).
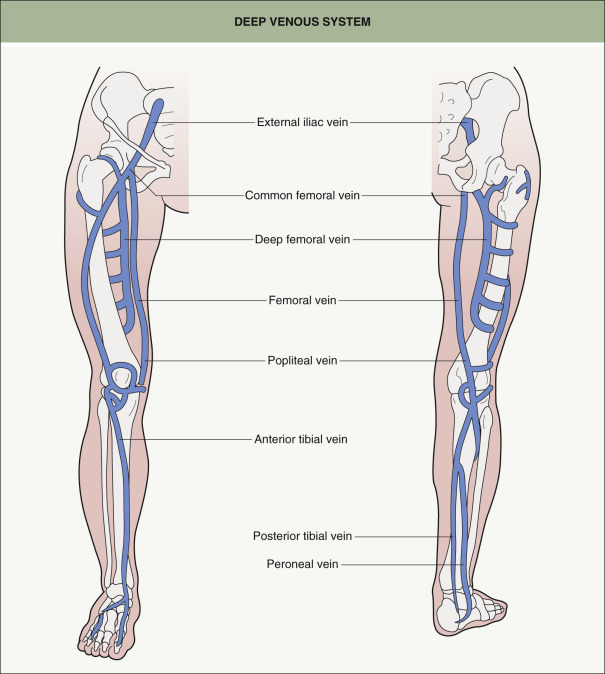
An interconnecting system of perforating veins that pass through the deep fascia connects the superficial veins to the deep veins of the calf or thigh ( Figs 155.1 & 155.3 ). All venous blood eventually is received by the deep venous system on its way back to the right atrium.
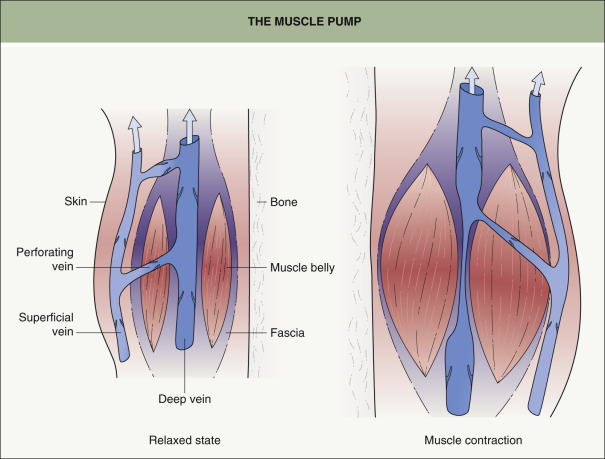
Blood is propelled back to the heart via the pumping action of the foot and calf muscles, which squeeze blood out of a venous segment through one-way valves when muscle contractions increase the pressure within the fascial muscle compartment ( Fig. 155.4 ). Venous pathology can result from primary muscle pump failure due to venous obstruction (thrombotic or non-thrombotic) or from venous valvular incompetence .
A genetic predisposition for the development of varicose veins has been observed, with both autosomal dominant and recessive patterns of inheritance described. Approximately 70% of patients can identify superficial venous disease as a familial trait. Hormonal influences from pregnancy or estrogen and progesterone supplementation are believed to be additional predisposing factors .
In summary, telangiectasias, reticular veins, and varicose veins are a reflection of hereditary and hormonal factors as well as static gravitational pressures and dynamic muscular forces transmitted through failed venous valves. While genetic traits cannot be altered, the influence of hemodynamic forces and hydrostatic pressure can be modified, both by sclerotherapy and surgical techniques.
Physical Examination of the Phlebology Patient
The purpose of the clinical examination is to survey the main venous trunks in order to determine where the primary or highest points of reflux are located. During the examination, the patient should be standing, preferably on a platform. It is important to determine the grade of venous insufficiency . The recommended CEAP classification system is based on clinical manifestations (C), etiologic factors (E), anatomic distribution of disease (A), and the underlying pathophysiologic findings (P) ( Table 155.1 ) . The venous severity scoring (VSS) system is based on three elements: the number of anatomic segments affected, grading of symptoms and signs, and disability. The consensus statement on this approach to clinical assessment was published worldwide in 25 journals and books in eight languages.
| CEAP CLASSIFICATION OF CHRONIC VENOUS DISORDERS |
| Clinical classification (C) |
|
| Etiologic classification (E) |
|
| Anatomic classification (A) |
|
| Pathophysiologic classification (P) |
|
For the practicing physician, CEAP is an instrument for establishing the correct diagnosis, guiding treatment and assessing prognosis. It is important to stress that CEAP is a descriptive classification, while VSS and quality-of-life (QoL) scores represent instruments for longitudinal clinical research in order to assess outcomes. In modern phlebologic practice, the vast majority of patients will have a duplex scan of the venous system of the leg, which will provide data on E, A and P. In basic CEAP, the single highest descriptor should be used for clinical class, e.g. a patient with varicose veins, swelling and lipodermatosclerosis will be C4b (see Table 155.1 ). However, use of just the C-classification provides no advantage over previous classification schemes that were based solely on clinical appearance. Even in basic CEAP, where a duplex scan is performed, classification of E, A and P with multiple descriptors is recommended. For A (anatomic classification), the simple s, p and d descriptors should be used, e.g. a symptomatic patient with stasis dermatitis, primary disease of the superficial and deep veins, and reflux would be classified as C4aS; Ep; As,d; Pr. While venous disease is complex, it can be described using this system, and clinicians are encouraged to use all four components of CEAP.
A disposable sclerotherapy garment is an excellent means for preserving patient modesty while gaining full access to the entire lower extremity. A two-step examination stool also aids in the clinical assessment. The medial plantar and ankle regions are examined for the presence of specific skin changes, including clusters of telangiectatic veins (referred to as “corona phlebectasia”), hemosiderin deposition, livedoid vasculopathy (atrophie blanche), lipodermatosclerosis, and/or active or healed ulceration (see Ch. 105 ). These findings suggest a chronic state of venous insufficiency, located in either the saphenous veins or the deep venous systems. The lateral posterior malleolar region is similarly examined. Skin changes in this area most often indicate small saphenous vein insufficiency.
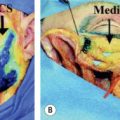
There are three branch veins that are important for evaluating the saphenofemoral junction (SFJ) . Attention is first directed toward the anterior abdominal wall. The presence of varices in this area (“caput medusae”) indicates insufficiency of the superficial epigastric vein (see Fig. 155.2 ); this is always abnormal and should alert the phlebologist to underlying deep venous obstruction, usually in the iliofemoral segment (see Fig. 155.3 ). The posteromedial thigh vein and the anterolateral thigh vein are often also insufficient in patients with GSV insufficiency and this usually presents as dilated reticular veins and telangiectasias within the involved anatomic zones .
Laboratory Evaluation of the Superficial Venous System
Vascular testing is indicated for symptomatic patients in whom the source of reflux is unclear by physical examination as well as several other clinical situations ( Table 155.2 ). In the past, a key instrument was a handheld Doppler device, which detects increased or decreased frequency shifts from ultrasound waves that are reflected from blood cells coming toward or going away from the Doppler ultrasound probe, respectively . Handheld Doppler devices have 4 MHz probes that penetrate more deeply (up to 8 cm) and 8 MHz probes that penetrate more superficially (0.5–2 cm).
| INDICATIONS FOR VASCULAR TESTING |
|
When utilizing a Doppler device, the examiner places the probe over the vein in question, and then compresses distal to the probe to create a flux sound ( Fig. 155.5A ) . Because the veins have one-way valves, if a vein is normal there will be no reflux or reversal of flow upon release of the distal compression. If a reflux sound is heard after release of the distal compression, this is abnormal and diagnostic of venous valvular insufficiency ( Fig. 155.5B ). It is best to hold the probe at a 30° to 45° angle . Reversal of blood flow, as the patient increases abdominal pressure with the Valsalva maneuver, indicates incompetent valves from the SFJ to the distal-most site of the probe. When placed on the medial superior thigh over the GSV a few centimeters distal to the SFJ, reversal of blood flow for >0.5 seconds indicates an incompetent SFJ. These patients require endovenous RF or laser closure of the GSV before any treatment of the distal varicose, reticular and/or telangiectatic leg veins.
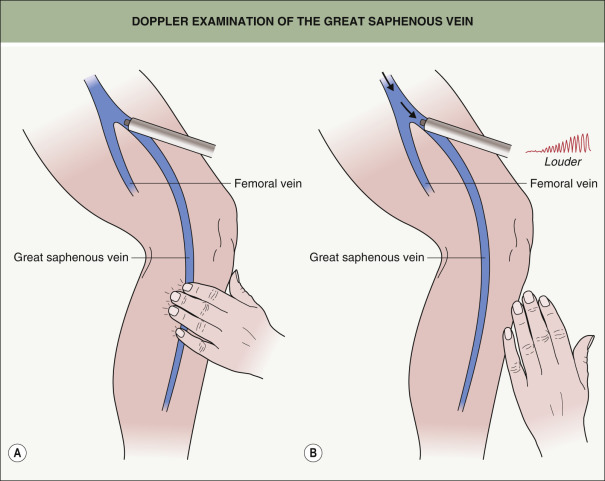
Nowadays, most vein experts utilize duplex ultrasonography (also referred to as duplex scanning) which is a faster, highly accurate, non-invasive technique that can provide both anatomic and physiologic information regarding the venous system ( Table 155.3 ). It is the modality of choice, not only for pretreatment assessment of varicose veins, but also for guidance during endovenous RF and laser procedures and for performing follow-up studies. Duplex examination of normal veins involves the use of a 7.5–12 MHz high-resolution B-mode scanner. In B-mode (brightness mode) ultrasonography, a linear array of transducers simultaneously scans a plane through the body and creates a two-dimensional image on a screen ( Fig. 155.6 ). This type of scanning is more commonly known as 2D mode. Velocity information is presented as a color-coded overlay superimposed on a B-mode image and is called color Doppler.
| COMPARISON OF DOPPLER ULTRASOUND DEVICE AND DUPLEX ULTRASOUND SCANNER FOR PRESCLEROTHERAPY EVALUATION | ||
|---|---|---|
| Doppler | Duplex | |
| Portability | Yes | Yes |
| Easy to use | Yes | Yes |
| Cost (2017) (approximate in US dollars) | Unidirectional: $150–$750 | Color: $20 000–$50 000 |
| Information obtained |
|
|
| Reliability | Less reliable because of blind, non-timed, pulsed sound beam | More reliable because of actual visualization of venous anatomy |
Compression in Sclerotherapy and Venous Disease
Graduated compression has many benefits in patients with venous disease including providing symptomatic relief, hastening venous ulcer healing, and minimizing the risk of venous ulcer recurrence . By reducing the diameter of veins, compression therapy increases flow velocity and decreases the chance of thrombus formation. Following sclerotherapy (which is essentially a controlled thrombophlebitic reaction), compression reduces the diameter of the resulting thrombosis of the treated vein, which minimizes the inflammatory reaction, thereby decreasing the extent and incidence of post-treatment hyperpigmentation, recanalization of the vein, and telangiectatic matting. Compression also increases the contact between the sclerosant and the endothelial lining of the vessel wall, potentiating pan-vessel obliteration. There are three main types of graduated compression: graduated elastic compression stockings or bandages, inelastic compression garments or bandages, and pneumatic compression pumps . Compression bandages are not recommended, as they are difficult to apply in a graduated manner and hold their compression for only a few hours after application.
Graduated elastic compression stockings are most commonly employed following sclerotherapy. They come in four classes, based on the pressure generated by the stocking at the level of the ankle, ranging from 20–30 mmHg (class I) to 50–60 mmHg (class IV) (see Ch. 105 ) . Most sclerotherapy patients wear thigh-high or panty-hose stockings. In addition, most manufacturers now offer so-called “fashion hose” which approximate 18 mmHg pressure and are sheer, leading to greater patient compliance. In a multicenter study performed by Weiss et al. , 20–30 mmHg graduated compression stockings were shown to increase sclerotherapeutic efficacy and diminish post-treatment hyperpigmentation. At least 3 days of compression were necessary to produce this positive effect and 3 weeks of daytime wear was found to be the optimal time required for the maximal effect .
We recommend that all patients wear graduated compression stockings for 1 week (“24/7”) after the procedure. It is important for the patient to wear the stockings 24 hours a day, even while in the shower and when sleeping. If one removes the stockings in the shower, the veins dilate. If one removes the stockings at night, the superficial system also dilates. For this reason, it is recommended that all patients wear the thigh-high stockings, as these are more hygienic and easier to use than panty-hose, which extend up to the waist.
Sclerosing Solutions
The optimal sclerosing agent is one that induces pan-endothelial destruction or removal . Thrombosis alone will not obliterate vessels because intact endothelium contains tissue plasminogen activator that can dissolve thrombi, leading to recanalization. The ideal sclerosing agent should possess no systemic toxicity and produce local endothelial destruction that fully extends to the adventitia with minimal thrombus formation . The resultant vascular destruction induces an inflammatory reaction which leads to fibrosis and eventual obliteration of the vessel.
There are several important points to remember when choosing a particular sclerosing solution and determining the appropriate sclerosant concentration ( Table 155.4 ) :
- 1.
If the sclerosant is too weak, insufficient endothelial damage will occur, leading to thrombosis secondary to varicose vessel wall damage. However, there will be no fibrosis and recanalization of the vessel occurs.
- 2.
Too strong a solution may lead to uncontrolled destruction of vascular endothelium and other vessel wall layers, which may eventuate in hyperpigmentation, neoangiogenesis (telangiectatic matting), and ulceration secondary to sclerosant extravasation.
- 3.
Choose the minimal sclerosant concentration (MSC) that will cause irreversible damage to the cellular wall of the abnormal vessel.
- 1.
Hyperosmotic agents – the osmotic agents include hypertonic saline, hypertonic saline–dextrose, and non-chromated glycerin. Their primary mechanism of action involves endothelial cell damage via dehydration ( Fig. 155.7A ).
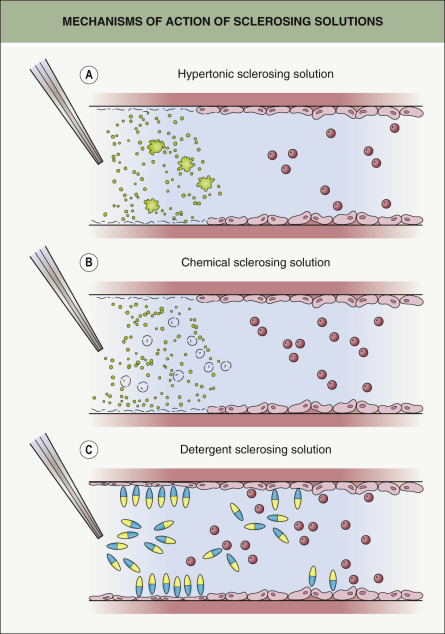
Fig. 155.7
Mechanisms of action of sclerosing solutions.
A Hypertonic, which induces endothelial damage via a gradient dehydration effect. B Chemical, which injures cells by acting as a corrosive. C Detergent, where the hydrophobic portion of the detergent molecule (blue) attaches to the endothelial cell wall while the hydrophilic portion (yellow) draws water into the cell, leading to an overhydrated maceration effect.
Adapted from Goldman MP, et al. Sclerotherapy, 5th edn. London: Mosby, 2011.
- 2.
Chemical irritants – the chemical irritants include chromated glycerin (glycerin plus potassium chromate) and polyiodide iodide. They injure cells by acting as corrosives; their cauterizing effect is thought to be due to the associated heavy metal ( Fig. 155.7B ).
- 3.
Detergent sclerosants – the detergent sclerosants include sodium tetradecyl sulfate, polidocanol, and sodium morrhuate. This group of agents causes vascular injury by altering the surface tension around endothelial cells ( Fig. 155.7C ).
| A GUIDE FOR THE SELECTION OF SCLEROSING SOLUTION CONCENTRATION AND VOLUME BY VESSEL TYPE | ||
|---|---|---|
| Vessel | Solution concentration | Volume (per region) |
| Telangiectatic matting (after previous treatment) | Glycerin, 72%, diluted 2 : 1 with 1% lidocaine with epinephrine | 0.1–0.2 ml |
| Telangiectasias (up to 1 mm) | Glycerin, 72%, diluted 2 : 1 with 1% lidocaine with epinephrine | 0.1–0.3 ml |
| Sodium tetradecyl sulfate, 0.1–0.25% Polidocanol, 0.25–0.5% | ||
| Hypertonic saline, 11.7% | ||
| Hypertonic saline, 10% and dextrose, 25% | ||
| Venulectasias (1–2 mm) | Sodium tetradecyl sulfate, 0.25–0.5% Glycerin, 72% | 0.2–0.5 ml |
| Polidocanol, 0.5–0.75% | ||
| Hypertonic saline, 11.7–23.4% Hypertonic saline, 10% and dextrose, 25% | ||
| Reticular veins (2–4 mm subcutaneous blue veins) | Sodium tetradecyl sulfate, 0.1–0.25%, foam | 0.5 ml (may increase to 1–3 ml if filling of reticular vein by foam is observed) |
| Polidocanol, 0.25–0.5%, foam | ||
| Non-saphenous varicose veins (3–8 mm) | Sodium tetradecyl sulfate, 0.5–1%, foam | 0.5 ml for liquids, 3 ml of foam per injection site in a large-capacity vein |
| Polidocanol, 1–3% * | ||
| Saphenous varicose trunks (usually >5 mm) | Polidocanol foam (Varithena ® ), 1% | 0.5 ml for liquids (low-volume injection critical at high concentrations), 3 ml with foam |
| Sodium tetradecyl sulfate, 1–3%, foam | ||
| Polidocanol, 3–5% ** | ||
* 1% is the highest concentration available in the US.
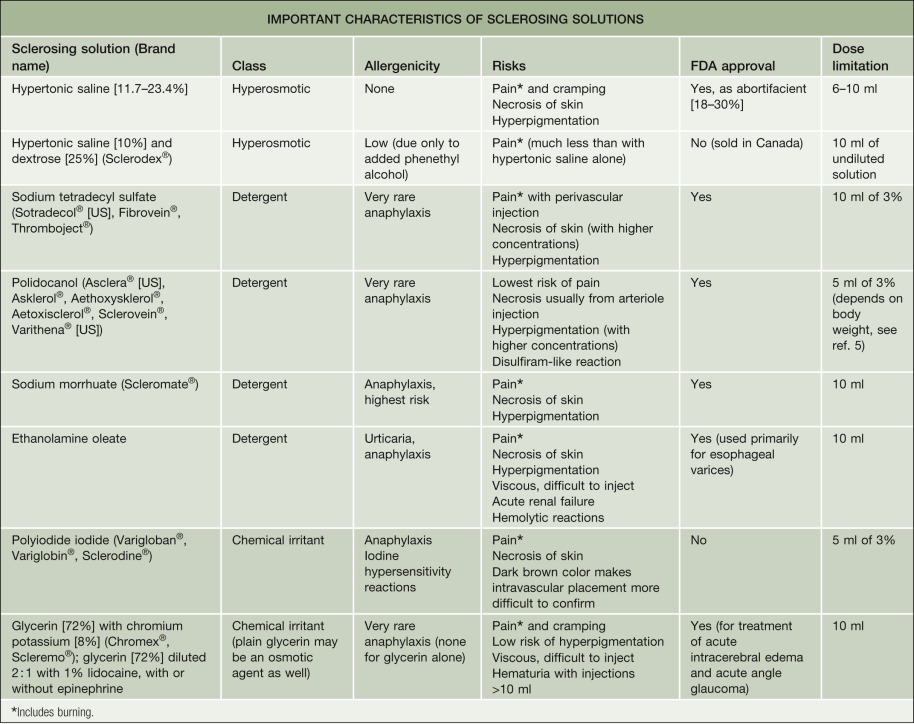
The only agents that are Food and Drug Administration (FDA)-approved for sclerotherapy in the US are sodium tetradecyl sulfate, sodium morrhuate (fatty acids in cod liver oil), and polidocanol , including the 1% foam formulation (Varithena ® ). Hypertonic saline is approved as an abortifacient rather than for the treatment of varicose veins. Glycerin is approved as a hyperosmotic agent for the treatment of acute intracerebral edema and acute angle glaucoma. Polyiodide iodide, a chemical irritant, lacks FDA approval for any indication. Glycerin, hypertonic saline (11.7–23.4%), polidocanol (0.25–5%), and sodium tetradecyl sulfate (0.1–3%) are the current sclerosants of choice in the US.
Determining the minimal volume and concentration of the most appropriate sclerosing agent is a key factor in producing effective sclerotherapy results. This concept, termed the minimal sclerosant concentration (MSC), will enable the sclerotherapist to achieve maximal results while minimizing the complication profile . A guide to suggested sclerosing agents and initial choices of sclerosant concentrations and volumes for instillation as related to vessel type and diameter is presented in Table 155.4 .
It is also important to have a working knowledge of the complication profile of each sclerosing solution (see Table 155.5 ). The osmotic sclerosants such as hypertonic saline are associated with burning and cramping on injection and an increased incidence of ulcerative necrosis secondary to extravasation.
Sodium tetradecyl sulfate and polidocanol have been associated with a low incidence of allergic reactions, ranging from urticaria to anaphylaxis (<0.01%) . However, with increasing usage, it is now thought that the risk of type I (i.e. immediate hypersensitivity) reactions is extremely low. In fact, after over 30 years of use in over 60 000 patients, the authors have never seen an allergic reaction except for one case of urticaria. Hyperpigmentation has been reported in 5–30% of patients treated with all sclerosing solutions except glycerin, which has a <1% incidence of pigmentation. In the authors’ experience, the incidence of hyperpigmentation is related more to the concentration of the sclerosing agent as well as patient skin type, vessel type, and other vascular fragility factors .
Creating foaming detergent solutions by mixing them with air or CO 2 (typically in a mixture of 1 ml of solution with 4 ml of air or CO 2 ) has been found to increase the potency of the sclerosing solution twofold while decreasing its adverse effect profile fourfold. Foam displaces blood and remains for an extended time in the target vessel. This makes the sclerosing solution more efficient, so that it may be utilized at lower concentrations. The use of CO 2 rather than room air allows the gas–sclerosant bubbles to breakdown more quickly, minimizing the possibility of gas embolization into the arterial circulation via a patent foramen ovale. Of note, the detergent moiety in the foam is broken down within seconds and/or within centimeters of its injection, so that the foam bubbles that reach the heart are purely air with no toxicity.
Techniques for Treating Telangiectasias and Reticular Veins
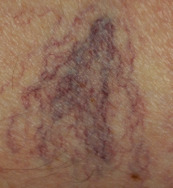
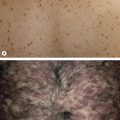
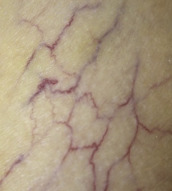
The treatment of telangiectasias and reticular veins is most commonly performed for cosmetic improvement, but up to 50% of these patients also report a variety of symptoms, from dull aching legs to throbbing pain over the telangiectasia . Telangiectasias are best defined as nearly flat, red to blue vessels 0.1–1 mm in diameter ( Table 155.6 ). Venulectasias are blue- to violet-colored vessels, sometimes distended above the skin surface and most often 1–2 mm in diameter. Reticular veins have a cyanotic blue to blue–green hue and are visually 2–4 mm in diameter. When a complex of reticular veins is located on the lateral thigh, it is felt to be a vestige of embryonal development known as the lateral subdermal plexus .
| CLASSIFICATION OF VEINS AND CLINICAL APPROACH TO TREATMENT OF VENOUS PATHOLOGY | ||||||
|---|---|---|---|---|---|---|
 | Type | Description | Diameter (mm) | Color | Treatment | 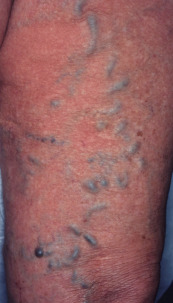 |
| I | Telangiectasias (spider veins) Telangiectatic matting | 0.1–1 | Usually red network |
| ||
| II | Venulectasias | 1–2 | Violaceous |
| ||
Get Clinical Tree app for offline access 
| ||||||