Patients are well informed and seek autogenous breast reconstruction. The motivating factors include a preference for autologous tissue reconstruction and the complementary improvement in body contour, safety concerns surrounding implants, and implant-related complications in the setting of previous radiation therapy. In this article a variety of perforator flaps from donor sites that include the trunk (thoracodorsal artery perforator and intercostal artery perforator), abdomen (deep inferior epigastric artery perforator and superficial inferior epigastric artery), and buttock (superior gluteal artery perforator and inferior gluteal artery perforator) are described. Flaps from the trunk can be pedicled for partial breast reconstruction, and free flaps from the other donor sites can completely restore a natural-looking breast. The information obtained from preoperative CT and MRI can direct the surgeon toward the most successful operative plan. However, the decision as to which flap may be most appropriate for an individual patient is complex. This article reviews pertinent surgical anatomy, preoperative planning, intraoperative decision making in flap elevation, and reported outcomes.
A variety of flap designs can be used to create a breast mound that appears and feels similar to the natural breast. The trend in breast cancer treatment is toward less deforming methods to excise the tumor while sparing normal surrounding tissue. The development of perforator flaps parallels this trend in that current techniques have evolved toward the harvest of autologous tissue with minimal disruption of the muscle through which the blood vessels pass. The evolution of workhorse flaps such as the free transverse rectus abdominis myocutaneous (free-TRAM) flap into the muscle sparing-TRAM (MS-TRAM) and deep inferior epigastric artery perforator (DIEP) flap variations highlights this trend toward lower donor site morbidity.
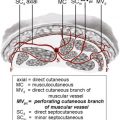
A variety of methods have been used to name and describe perforator flaps, including anatomic location and arterial supply. In the absence of a standardized naming scheme for these flaps, multiple names for the same flap proliferated in the literature, resulting in confusion and misunderstanding. With the Gent consensus, however, it is now standard to name a perforator flap according to its artery of origin. Anatomic studies show that the parent artery to a perforator flap commonly arises on the deep surface of the muscle and gives off musculocutaneous perforating vessels that penetrate through the muscle to supply the overlying fat and skin. In other instances, a vessel may branch off the parent artery and travel through a septocutaneous membrane to the skin paddle. These vessels are referred to as septocutaneous perforator vessels.
Careful, meticulous dissection of the perforating vessels down through the muscles to the parent artery yields the purest form of a perforator flap and thereby preserves the entire muscle along with its motor nerves. Occasionally, anatomic variation or concerns about tissue perfusion encountered intraoperatively may guide the decision to include some muscle in the flap, thereby changing the design of the flap to one of its muscle-sparing variants.
The psychological power of immediate breast reconstruction combined with the bonus of an abdominoplasty or buttock lift appeals to many patients. However, not all patients are candidates for immediate reconstruction, and not all women opt for mastectomy. Some women may require delayed reconstruction or partial reconstruction of breast-conserving deformities. In addition, the paucity of lower abdominal tissue or previous abdominal surgery, such as cosmetic abdominoplasty, liposuction, laparotomy, or previous pedicled TRAM reconstruction may preclude using the lower abdominal tissues for reconstruction. These contraindications coupled with the need for alternative options for autologous breast reconstruction drive the search for new donor sites and the development of new flap designs.
Lower abdominal flaps
Flap designs for the lower abdominal donor site highlight the evolution and sophistication of microsurgical technique. Hartrampf’s introduction of the pedicled TRAM flap in 1982 ushered in the modern era of breast reconstruction. Further investigation into the vascular anatomy of this flap design revealed that the dominant arterial supply to the rectus abdominis muscle is the deep inferior epigastric artery (DIEA). Understanding that the rectus abdominis muscle can carry a larger subcutaneous fat and skin paddle when based on the deep system, many microsurgeons adopted the free-TRAM flap for breast reconstruction.
For many reconstructive surgeons, it became clear that donor site morbidity could be lowered by reducing the amount of muscle taken with the flap and injury to the muscle left behind. The concepts of donor site muscle sparing were introduced with the segmental latissimus dorsi myocutaneous and free-TRAM flaps. Koshima refined this concept even further when he used the skin paddle overlying the rectus abdominis muscle for reconstruction of the mouth and groin. Koshima’s flap designs were based on a single paraumbilical perforating vessel off the DIEA and composed of subcutaneous fat and skin only. Soon after, Allen performed the first deep inferior epigastric perforator (DIEP) flap for breast reconstruction by transferring the lower abdominal fat and skin of a TRAM flap while sparing the underlying rectus abdominis muscle.
Whereas pedicled TRAM and free-TRAM flaps remain workhorses, the quest to minimize donor site morbidity has given rise to the development of the MS-TRAM and DIEP flap designs, each of which spare more muscle. Nahabedian and colleagues developed a classification system to describe the degree of sparing of the rectus abdominis muscle: MS-0 describes the sacrifice of the full-width (partial length) of the muscle; MS-1 describes the preservation of the lateral segment; MS-2 describes the preservation of the lateral and medial segments; and MS-3 refers to the preservation of all the rectus muscle (the equivalent of a DIEP flap).
This classification system shows the continuum that exists among these flaps, which require the disruption of the anterior rectus sheath and variable amounts of rectus abdominis muscle. Hence, the primary advantage of a perforator flap is that it spares the underlying muscle and motor innervation, thereby reducing morbidity and preserving functionality.
Distinct from these flaps is the superficial inferior epigastric artery (SIEA) flap, which was first described by Grotting in 1991 and termed the free abdominoplasty flap. This design precludes dissection below the rectus sheath because the superficial epigastric artery and vein arise from the femoral vessels and head directly into the subcutaneous fat of the lower abdomen.
DIEP Flaps
Most women with breast cancer who have or have had mastectomy are potential candidates for DIEP flap breast reconstruction and will be of an age where they have excess fat and skin overlying the lower abdomen. The fat in this region is typically soft and easy to shape and therefore most closely approximates the feel of a normal breast. In most cases, and even for most thin patients, the tissue found in the lower abdominal region is sufficient to reconstruct a breast mound. In an 8-year review of 172 patients who underwent breast reconstruction with tissue from the lower abdomen, Granzow and colleagues found that the weight of the harvested flaps averaged approximately 120% of the weight of the mastectomy specimens in immediate reconstructions. Thus, the harvested flap is often trimmed of excess tissue and thin patients (who tend to have smaller breast volumes to replace) often have enough lower abdominal tissue to closely match the contralateral breast.
An absolute contraindication specific to the use of the lower abdominal tissues for breast reconstruction is previous abdominoplasty. Previous abdominal liposuction does not preclude DIEP flap elevation, but caution should be exercised to ensure proper preoperative and intraoperative perforator evaluation before committing to elevating the abdominal tissue as a DIEP flap in this situation. Active smoking within 4 to 6 weeks of planned surgery contributes to fat necrosis/partial flap loss, delayed wound healing, and random flap loss at both the mastectomy and abdominal donor sites.
Previous abdominal surgery through incisions that traverse the desired skin paddle can also affect the design of the flap reconstruction. For example, the subcutaneous fat and skin lateral to an appendectomy scar may not adequately perfuse after flap elevation. Similarly, the skin paddle contralateral to a midline abdominal scar may not perfuse well enough after flap elevation to survive transfer to the breast. However, each hemiflap could be viable if elevated on its own DIEA or SIEA pedicle.
Patients are also encouraged to complete any radiation therapy to the chest before surgical breast reconstruction. Although the perforator flaps usually tolerate radiation well, a superior long-term result is typically obtained in reconstructions performed after rather than before chest wall radiation. This strategy spares the flap from the damaging effects of radiation and allows the removal of thick, stiff irradiated chest wall skin. It is then replaced with soft, nonradiated abdominal skin and soft tissue.
Excellent state-of-the-art descriptions of DIEP flap elevation have been published; the reader is directed to these articles for detailed description of technique.
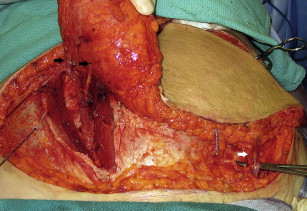
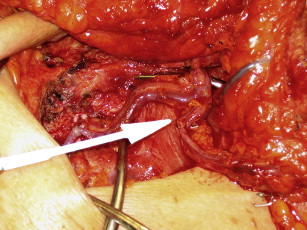
For all flaps, a fusiform ellipse is outlined on the lower abdomen that extends from the suprapubic crease inferiorly to just above the umbilicus superiorly and laterally to the anterior superior iliac spine. However, the amount of tissue that can be safely taken and still allow for closure of the donor site must be estimated. The surgeon must also estimate how much of the lower abdominal flap is required to provide the volume necessary for appropriate reconstruction of the breast. For unilateral DIEP flap reconstruction, Zones 1 to 3 can be reliably transferred on a single adequately sized perforator from the medial row and zone 4 is typically removed. An adequate perforator should have a vein greater than 1 mm in diameter where it enters the flap and a palpable or visibly pulsating artery. The portion of the flap that is used for breast reconstruction should be centered on the dominant perforator to provide the best flap perfusion and to avoid fat necrosis ( Fig. 1 ). In the situation in which the entire lower abdominal flap is desired or needed to match the large contralateral breast, the contralateral deep inferior epigastric (DIE) pedicle, a perforator from the contralateral hemiflap, or the contralateral superficial system pedicle can be anastomosed to the side branch of the main ipsilateral pedicle, thereby creating a supercharged or turbocharged construct ( Fig. 2 ).