Pediatric Facial Reanimation Using a Functional Gracilis Muscle Transfer
Brad M. Gandolfi
Jeffrey R. Marcus
Michael R. Zenn
DEFINITION
Facial paralysis does not describe a disease but instead the sequelae of denervation resulting from a spectrum of diseases, congenital defects, and acquired defects with a similar effect on the facial musculature.
Facial nerve paralysis can involve the entire facial nerve distribution or only certain branches of the facial nerve. The facial nerve may be affected symmetrically or asymmetrically. The extent of the paralysis may be complete or incomplete relative to residual motor capacity.
The pathogenesis of facial paralysis is complex and dependent on the etiology of the disease.1,2 That discussion is beyond the scope of this chapter but briefly can be divided into congenital and acquired facial paralysis (Table 1). An accurate diagnosis is crucial to selecting the proper surgical (or nonsurgical) intervention.
Facial reanimation is a general term to describe restoration of facial movement. There are two main distinctions based on the status of native facial muscles:
Nerve transfers—When the denervating event is such that existing native facial musculature can be amenable to reinnervation with a functioning nerve, a nerve transfer is warranted. In this setting, the goal may be to restore multiple facial nerve targets—ie, multiple separate but coordinated movements.
Functional muscle transfer—In long-standing or congenital facial paralysis, native muscles are not present to perform the function. Therefore, a transfer of functioning muscle provides reanimation. In such cases, only one facial movement, rather than multiple, is the goal of the procedure. One muscle provides one movement.
This chapter focuses on free muscle transfer for restoration of smile in the pediatric population.
TABLE 1 Causes of Facial Paralysis | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||
ANATOMY
Facial Nerve
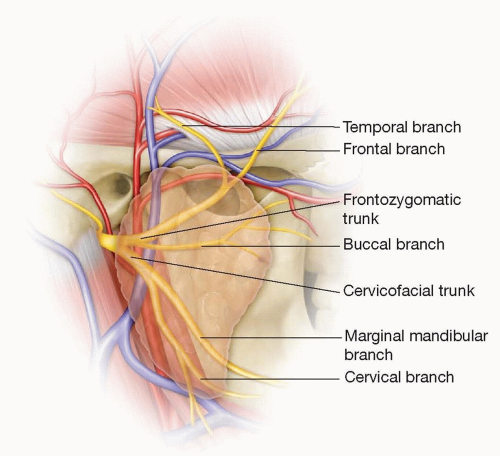
The facial nerve exits the stylomastoid foramen and enters the posterior parotid gland, dividing the gland into superficial and deep components.
The facial nerve divides into two main branches within the parotid gland: the frontozygomatic and the cervicofacial trunks. These branches further divide before exiting the parotid gland.
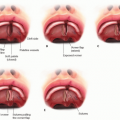
Five branches are classically described as exiting the parotid (frontal/temporal, zygomatic, buccal, marginal mandibular, and cervical). However, the branching pattern for each individual is unique, and many of the abovenamed nerves exist in multiples.3 The average number of branches exiting the parotid is 7 (FIG 1).
Buccal and zygomatic branches begin to arborize and cross within and beyond the parotid, resulting in redundancy in the functional territory of these buccal-zygomatic branches.3,4 It is difficult to separate the activity of these branches as purely buccal or zygomatic because stimulation often results in a mixed response. Due to mixed nerve function, sacrifice of one nerve for end-to-end coaptation does not result in functional loss.
The target for reanimation of smile is a buccal or zygomatic branch, selected based on targeted muscle stimulation. Stimulation should ideally identify a branch that produces vertical and horizontal movement of the commissure and upper lip consistent with the patient’s natural smile, with minimal extraneous movement.
Zuker point describes a relatively consistent buccal-zygomatic branch that meets the needs for facial reanimation.4 It is found at the center of a line drawn from the oral commissure to the helical root.
Nerve to the Masseter
Gracilis Muscle
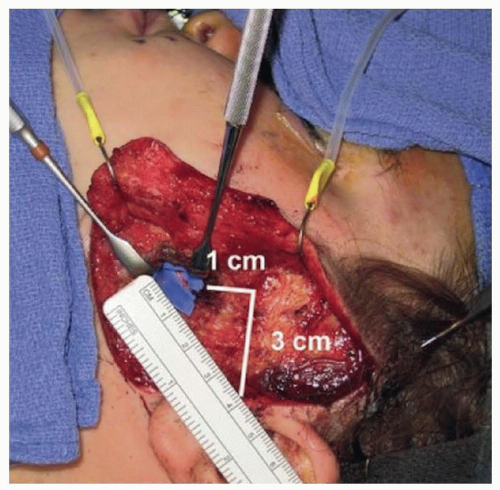
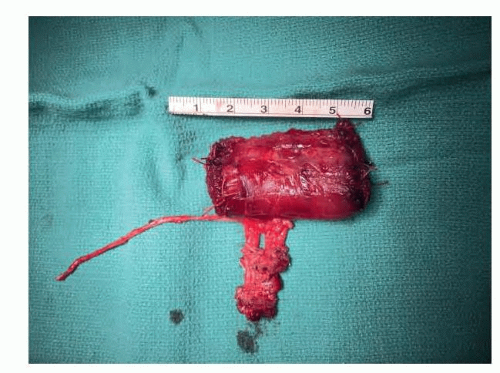
The gracilis muscle originates along the body and inferior ramus of the pubis, just below the pubic tubercle.7 It runs along the medial thigh and inserts into the medial tibial condyle, immediately deep to the sartorius muscle (FIG 3A).
The gracilis lies immediately posterior to the much larger adductor longus, which is easily palpated on most patients.
The muscle is supplied by the medial circumflex femoral artery, a branch of the deep femoral artery. The pedicle runs underneath the adductor longus en route to the gracilis.
The artery enters the muscle 5 to 12 cm inferior to the pubic tubercle. It is generally 1 to 2 mm in diameter and may have two or three branches at the level of the muscle (FIG 3B).
The artery is accompanied by two vena comitantes.
The artery and the accompanying vein are 5 to 6 cm when dissected to the deep femoral vessels.
The muscle has a minor pedicle at its distal aspect that is not appropriate for microvascular transfer.
A branch of the obturator nerve supplies the gracilis muscle. It splits from the anterior branch of the obturator behind the pectineus and runs with the branch to the adductor longus. The nerve joins the muscle from its lateral aspect, generally at a 45-degree angle to the vascular pedicle. The nerve pierces the muscle and arborizes within it.
The gracilis muscle has a tendinous origin from the pubis, which can be included with the flap. The muscle can be 3 to 4 cm wide at the level of the pedicle, requiring some muscle splitting to avoid bulky reconstructions.
PATIENT HISTORY AND PHYSICAL FINDINGS
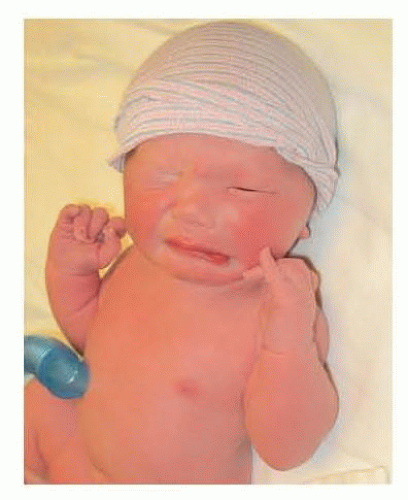
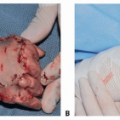
The most important initial distinction in the preoperative evaluation of pediatric patients with facial paralysis is whether the condition is congenital or acquired. For those with congenital paralysis, the condition may have been apparent at birth or recognized shortly thereafter (FIG 4). For those with acquired paralysis, the circumstances and details of the inciting event(s) should be carefully noted. The timing of functional loss is the most critical detail.
Functional impairment, including the following, should be noted:
Excessive tearing and/or dryness requiring frequent use of lubricating drops
Inability to close the eyes fully at night requiring the use of viscous eye lubricant
Oral incompetence (drooling) or speech articulation difficulties
Presence and extent of dental caries, which are predisposed on the affected side
Any prior treatments should be noted as well as any progression or improvement in the condition.
Prior medical, neurologic, or surgical history that would potentially be associated with the condition or the potential surgical treatments that may be considered should be noted.
A focused examination of the function of the facial nerve is performed, taking care to document both static and dynamic deficiencies. The motor territory associated with each branch should be graded relative to the completeness of the paralysis. We prefer the Toronto Facial Grading System to track our progress.2 If etiology of paralysis is unclear, level of injury can be elucidated using the Schirmer test, stapedius reflex test, salivary flow test, and taste examination.
Photographs and video of the following should be included in the initial evaluation:
Repose
Brow elevation
Eye closure (gentle)
Eye closure (strong)
Smile (lips together)
Smile (natural dental display)
Full dental display
Pucker
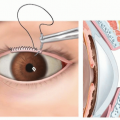
The ability to close the eyelids such that the cornea is protected must be assessed. Those who are unable to close fully and require viscous lubrication should be identified early and treated with priority ahead of smile reanimation.
Amblyopia may be seen in as many as 50% of patients with facial nerve palsies, and thus a thorough ophthalmologic examination is warranted.1 The eye examination should also assess for corneal pathology due to exposure.
The presence of the facial artery should be confirmed. It is palpable in most patients regardless of age.
The temporalis and masseter muscles should be palpated, particularly if considering the motor nerve to the masseter as a potential donor nerve. Normal strength and mass should be present.
Syndromic associations can be seen in congenital facial paralysis and can include hemifacial microsomia, Mobius syndrome, and Poland syndrome among others. Therefore, the examination should include an evaluation of the facial bones (orbits, zygoma, maxilla, and mandible), soft tissues, and ear. Mobius syndrome includes the presence of a cranial nerve VI deficit and can also be associated with a Poland deformity and congenital hand anomalies. Thus, cranial nerves VI, VII, IX, X, and XII should be evaluated for disability.
IMAGING
For candidate patients with long-standing or congenital paralysis, a radiographic workup is generally unnecessary. For those with associated congenital anomalies, such as craniofacial microsomia, craniofacial CT may be valuable for the comprehensive workup.
In cases of recently acquired facial paralysis, an EMG may be useful to establish a baseline, demonstrate improvement, or predict recovery. In congenital or long-standing cases, EMG does not provide information that contributes to treatment planning and therefore is not necessary.
NONOPERATIVE MANAGEMENT
Nonoperative management plays an important role in partial paralysis. A facial rehabilitation specialist may be able to obtain a more symmetric smile by strengthening weaker muscles and de-emphasizing stronger, more active muscles. Such treatment requires participation of the child.
In certain instances of incomplete paralysis, neurotoxins can be used on the unaffected side as an adjunct to obtain symmetry.
Long-standing, complete, and near-complete paralysis in the pediatric population generally requires surgical management.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree