Abstract
Orbital fractures are among the most common facial fractures and carry a significant cost to the healthcare system. Incidence and etiology vary greatly with geography, demographics, and socioeconomic factors. Assessment on initial presentation includes detailed history and physical examination, coupled with thin-cut maxillofacial computed tomography imaging. Patients with evidence of optic nerve compression or muscle entrapment require prompt surgical intervention within 24–48 hours. In the absence of an indication for acute surgery, reconstruction should be performed within 1–2 weeks. Several surgical approaches and implant options are available depending on fracture characteristics and location. New technologies, including intraoperative navigation and computer-assisted surgery, are emerging to assist with reconstruction of complex defects. A thorough understanding of periorbital anatomy and long-term changes in the orbit following trauma are critical to successful orbital fracture repair. Management of orbital trauma at specialized eye centers has been associated with lower cost and improved outcomes compared to treatment at other hospitals. Patients should be monitored postoperatively for acute complications, such as vision loss or retrobulbar hematoma. Suboptimal primary reconstruction frequently leads to long-term complications, such as persistent enophthalmos and diplopia, requiring reoperation.
Keywords
orbital trauma, orbital implants, orbital reconstruction
Background
Orbital fractures are among the most common facial fractures, and their associated cost to the healthcare system has been on the rise. Surgical repair of orbital fractures has been shown to add a day of hospitalization and an average of $22,000 in hospital charges for the average trauma patient. Treatment of orbital trauma at high-volume specialized eye centers has been shown to reduce cost compared to management at other hospitals. The incidence and etiology of orbital fractures vary greatly with geography, demographics, and socioeconomic factors, such as rates of violence and access to automobile safety technology. Among the civilian population within the United States, 38% of orbital fractures result from motor vehicle collisions, followed closely by 34% from assault; falls and sport injuries account for another 15% and 7%, respectively. Sixty-eight percent of orbital fracture patients are male and over half are aged between 18 and 44. Approximately 1 in 4 orbital fractures in the United States are managed surgically.
Surgical Anatomy
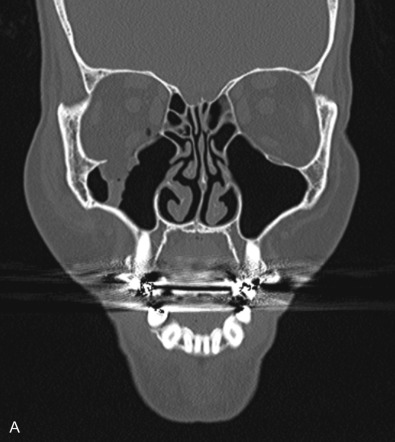
A thorough understanding of orbital anatomy is key to achieving optimal clinical outcomes and minimizing postoperative complications. The bony orbit is approximately 30 mL in volume and 35–40 mm in length. The orbital floor is composed of the maxillary, zygomatic, and palatine bones. The thin medial wall includes the orbital plate of the ethmoid bone, lacrimal bone, frontal process of the maxillary bone, and the lesser wing of the sphenoid ( Fig. 1.9.1 ). The posterior maxillary wall is frequently used as a bony landmark for the orbital apex, beyond which dissection should be avoided to minimize the risk of injury to the optic nerve.
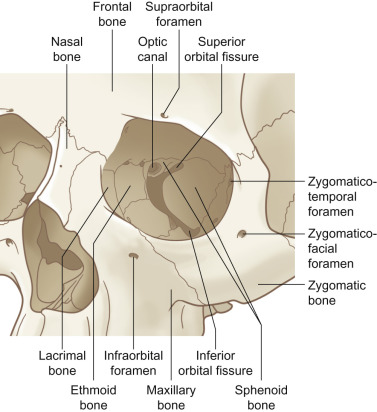
The orbit includes six muscles responsible for extraocular movements. The four rectus muscles originate at the posterior orbit at a fibrous ring called the annulus of Zinn. The inferior rectus runs along the orbital floor and attaches approximately 6.5 mm inferior to the limbus. It must be evaluated for entrapment or rounding on computed tomography (CT) following orbital trauma. The inferior oblique muscle has its origin at the maxillary bone, just lateral to the opening of the nasolacrimal duct, and inserts on the lateral aspect of the globe. Its origin may come in the way of surgical access, particularly during combined floor and medial wall fracture repair. The anterior and posterior ethmoidal arteries run along the frontoethmoidal suture line at the superior border of the medial orbital wall, and must likewise be protected during dissection. The anterior ethmoidal artery lies approximately 24 mm from the lacrimal crest; the posterior ethmoidal artery may be found 36 mm from the lacrimal crest and approximately 6 mm from the orbital apex.
Clinical Presentation
As with any injury, a detailed patient history should be taken upon presentation following orbital trauma. Evidence of head or neck injury, loss of consciousness, nausea, and vomiting are all particularly relevant in facial trauma patients. Muscle entrapment or increased orbital pressure may trigger the oculocardiac reflex, leading to syncope, nausea, vomiting, or even life-threatening arrhythmias. The clinical history should include perception of a clear understanding of the mechanism of injury; a direct blow to the orbit or high-energy trauma should raise the suspicion for globe injury or other associated injuries. In fact, the mechanism of injury has been found to be a better predictor of visual prognosis than the specific pattern of facial fractures. The age, baseline vision acuity and occupational history will likewise factor into the surgical management of an orbital fracture. Although focus is generally placed on preserving primary and downward gaze, patients should be asked about any activities that require prolonged upward gaze or extensive extraocular movement. A young heavy machinery operator, baseball outfielder, or pilot may warrant more aggressive surgical intervention to minimize the risk of posttraumatic diplopia.
The initial physical examination of any trauma patient should focus on ensuring adequate ventilation, hemodynamic stability, and prompt assessment of any life-threatening injuries. Once stable, every patient should undergo at least a basic eye exam, pending more thorough ophthalmologic evaluation. An initial eye exam in the emergency room should at the least include visual acuity testing, extraocular movements, pupillary exam, and an external exam. Evidence of diminished visual acuity, proptosis, or afferent pupillary defect all raise the suspicion for orbital compartment syndrome, which should prompt an emergent cantholysis to decompress the orbit. Bradycardia and defects in extraocular motility should raise concern for muscle entrapment. True muscle entrapment is rare in the adult population owing to diminished orbital wall bone elasticity, and significant periorbital edema may sometimes limit extraocular movements. Forced duction testing may be performed at the bedside to rule out entrapment. The external exam should assess any point tenderness, bony step-offs, crepitus, and sensory deficits; trauma to the infraorbital nerve may frequently lead to dysesthesia. Globe position should be assessed for inferior displacement, enophthalmos, or exophthalmos. A flat anterior chamber, misshapen pupil, or circumferential subconjunctival hemorrhage should raise the suspicion for globe injury. Lateral or medial canthal deformity may indicate either zygomatic involvement or a naso-orbital ethmoid fracture, respectively.
Full ophthalmological evaluation and clearance is critical for all orbital fracture patients prior to surgical repair. In addition to the aforementioned basic eye exam, ophthalmological evaluation should include a more thorough assessment of visual acuity, globe pressure, visual fields, a slit lamp ocular exam, and a retinal exam. Approximately 22%–29% of orbital fracture patients will present with concomitant ocular injuries. In one series, 79% of open globe injuries resulted in blindness. Several studies have demonstrated higher incidence of globe injuries associated with blow-out fractures compared to fractures with rim involvement.
Any evidence of muscle entrapment on exam should warrant early surgical intervention within 24–48 hours. Several studies have demonstrated increased risk of late diplopia with delayed repair in the setting of muscle entrapment. It has been postulated that true muscle incarceration leads to occlusion of the artery running in the central portion of the inferior rectus muscle, resulting in scarring and fibrosis within the muscle. Persistent bradycardia and hemodynamic instability secondary to the oculocardiac reflex are additional indications for immediate surgical repair. Additionally, many authors advocate immediate repair of large wall defects that result in early enophthalmos on presentation.
The timing of surgical repair of orbital fractures in the setting of concomitant globe injury remains controversial. On one hand, early intervention risks further trauma to a ruptured globe, compromising long-term vision outcomes. On the other hand, delaying repair of facial fractures may be detrimental to long-term cosmetic outcomes. Within the reconstructive surgery literature, many authors support careful early repair of orbital fractures with associated globe injury, particularly in the setting of high-energy penetrating trauma, where return of visual acuity is unlikely.
In the absence of globe injury or indications for immediate repair, many authors advocate repair of orbital fractures within a 2-week window. The degree of scarring and adhesions tend to increase after 2 weeks, complicating and decreasing the effectiveness of surgical repair because of fixed scar tissue. Conversely, waiting several days following initial injury may help with resolution of periorbital edema, which might otherwise complicate access during repair. Some feel that the immediate post-injury period is more vulnerable to spasm in the circulation to the optic nerve.
Radiological Evaluation
Thin-cut non-contrast CT of the orbits and face are the gold standard imaging modality for detecting orbital fractures. Cuts of 1–2 mm are generally recommended for adequate visualization, and the entire face should be imaged to assess for other associated facial injuries. Three-dimensional reconstructions may be helpful in assessing craniofacial fracture patterns following trauma. The orbital floor is best visualized on coronal and sagittal views, while the medial orbital wall is best seen on axial and coronal cuts. The sagittal view is particularly useful for assessment of the posterior ledge for orbital floor implant support. The coronal view provides good visualization of the orbital soft tissues and may be used to assess fat herniation or muscle entrapment. Rounding of the inferior rectus muscle has been associated with late enophthalmos if left untreated.
Classification
Orbital wall fractures are generally categorized based on involvement of the orbital floor, roof, or medial wall. Considering the specific fracture pattern is critical in selecting the appropriate surgical approach and implant. Isolated orbital floor fractures are most frequently repaired with an onlay implant with or without an orbital rim component. In isolated medial orbital wall fractures, the inferonasal bone strut is frequently preserved, serving as support for implant reconstruction. Traditional onlay reconstruction is an option if surrounding bone is strong enough to support an implant. In cases of poor exposure or lack of sturdy support surrounding the fracture segments, an inlay technique may be used. Porous polyethylene may be inserted into the ethmoid sinuses after reduction of orbital contents. Lastly, in the setting of an intact medial orbital wall fracture fragment, the defect may be repaired via endoscopic repositioning. The orbital contents are reduced, and the wall reconstructed by rotating the fracture fragment into stable position.
Combined medial wall and orbital floor fractures present a unique set of challenges. They are frequently associated with loss of the inferonasal bony strut support (a shallow bulge), hindering secure implant placement and increasing the risk of enophthalmos and diplopia. Further, dissection adequately exposing both the orbital floor and medial wall is frequently complicated by the origin of the inferior oblique muscle near the lacrimal fossa. Several implant choices exist for combined orbital floor and medial wall fractures. Some authors support the use of a rim-fixed orbital floor implant with a medial wing; single wraparound sheets are also available to cover both defects with one implant. Alternatively, each defect may be reconstructed separately, with two individual floor and medial wall implants.
Orbital fractures may additionally be categorized as either pure blow-out fractures or those involving the orbital rim. Two primary mechanisms have been described to explain isolated orbital wall fractures without associated rim involvement. The hydraulic theory holds that a blow to the orbital contents transiently increases the intraorbital pressure, leading to outfracture of the orbital wall. According to the buckling theory, a high-energy impact to the orbital rim is directly transmitted to the more fragile orbital wall, leading to the fracture. Many authors contend that both theories are likely at play in most orbital floor fractures. Given the anatomy of the medial orbit and neighboring ethmoid air cells, the hydraulic theory is likely most pertinent in medial orbital wall fractures. A third possibility is that the globe itself is driven posteriorly, fracturing the orbital walls when the globe diameter exceeds the diameter of the orbit.
Surgical Indications
Indications for surgical repair remain controversial and are largely based on predicting long-term cosmetic and visual sequalae of orbital trauma. The aforementioned cases of persistent bradycardia, optic nerve compression, periorbital muscle entrapment, and enophthalmos greater than 2 mm on initial presentation warrant early surgical repair within 24–48 hours. In the absence of indications for acute intervention, patients may be reevaluated and scheduled for surgery within 1–2 weeks. Evidence of extraocular movement restriction or diplopia, particularly in primary and downward gaze, are indications for repair when limitation of muscle movement can be proven. Certain patient populations dependent on intact extraocular motility, such as athletes, pilots, or heavy machinery users, may warrant more aggressive intervention. Significant globe malposition or orbital rim involvement leading to bony step-offs and disfigurement should likewise be reduced to prevent long-term cosmetic sequelae.
Predicting which injury patterns will lead to long-term enophthalmos and visual restrictions remains challenging. Fractures involving 50% of the orbital floor area are frequently used as a cut-off for non-surgical management. Fractures up to 3 cm 2 have been managed conservatively with optimal outcomes. The medial wall appears to be less forgiving, warranting repair for defects over 2 cm. Other imaging findings predictive of long-term globe malposition include rounding of the inferior rectus muscle on CT imaging.
Surgical Techniques
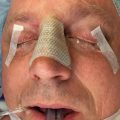
Repair of orbital wall fractures is performed under general anesthesia after infiltration of the surgical site with a local analgesic. Several commonly used surgical approaches exist to expose the orbital floor and medial wall ( Fig. 1.9.2 ). The subtarsal approach involves a cutaneous incision just below the tarsal plate, along a natural crease line. The orbicularis oculi muscle is dissected, and the orbital rim approached in the preseptal plane. The closely related infraorbital rim incision involves a cutaneous incision directly overlying the orbital rim. The latter approach has come out of favor due to less favorable scar placement. Some of the benefits of the subtarsal approach include lower risk of lower lid malposition and ectropion, as well as easier exposure without the need for a lateral canthotomy.
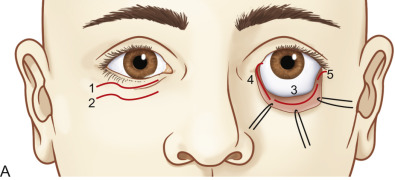
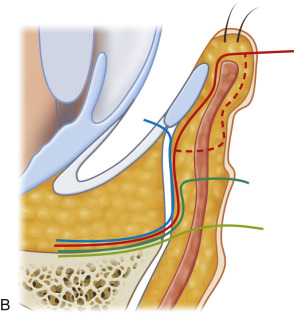
The subciliary approach includes an incision several millimeters below the lash line. Dissection is then either continued directly through the orbicularis oculi muscle, or carried in a step fashion in the subcutaneous plane for a few millimeters before muscle division just below the tarsal plate. The rim is then exposed in a preseptal plane. A major disadvantage of the subciliary approach is the high risk of scar contracture leading to ectropion. Extensive subcutaneous dissection may even lead to skin necrosis.
The transconjunctival approach involves a mucosal incision through the conjunctiva below the level of the tarsus ( Fig. 1.9.3 ). The rim is then exposed via either a preseptal or retroseptal dissection plane ( ). The transconjunctival approach has gained popularity in recent years due to its lower risk of ectropion and lack of cutaneous scar. Some authors have even noted favorable results without the use of suture. One downside is its limited exposure, frequently requiring the use of lateral canthotomy for larger fractures.