Abstract
Examination of the oral soft and hard tissues may reveal signs of developmental anomalies, infectious or inflammatory conditions, and neoplastic processes. Alterations of the oral mucosa may also be associated with underlying systemic disorders and these oral findings may be the first sign of a systemic disease. This chapter explores a selection of both common and unusual, but significant, oral diseases. It emphasizes the important elements of the history, clinical presentation, and histopathologic findings that can lead to the correct diagnosis of an oral disease. The pathogeneses and treatment options for oral diseases will also be examined.
Keywords
oral pathology, oral disease, oral medicine, oral mucosa, oral manifestations of systemic disease, geographic tongue, mouth, Fordyce granules, fissured tongue, hairy tongue, central papillary atrophy, necrotizing ulcerative gingivitis, desquamative gingivitis, cutaneous sinus of dental origin, morsicatio buccarum, gingival enlargement, gingival hyperplasia, contact stomatitis, aphthous stomatitis, eosinophilic ulcer of the oral mucosa, orofacial granulomatosis, oral leukoplakia, oral erythroplakia, bite fibroma, proliferative verrucous leukoplakia, actinic cheilitis, mucocele, necrotizing sialometaplasia, nicotine stomatitis
Introduction
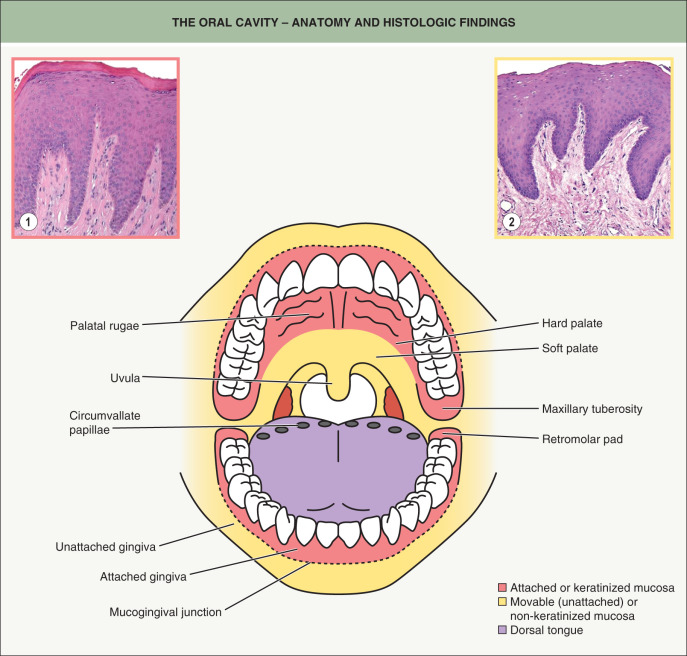
From both a clinical and histologic standpoint, the oral cavity is an amazingly diverse anatomic region. This is not surprising when one considers the various tasks and environmental interactions that take place within the mouth. Sites that are associated with the friction of mastication, such as the hard palate and attached gingivae, are bound to underlying periosteum and are referred to as attached mucosa or keratinized mucosa ( Fig. 72.1 ). Histologically, these anatomic areas show prominent rete ridge formation and a thick layer of orthokeratin. All other oral mucosal sites are “unattached” and are referred to as movable mucosa or non-keratinized mucosa . The soft palate, labial mucosa, and floor of the mouth exhibit only a modest degree of parakeratosis and are associated with numerous salivary glands; the buccal mucosa is similar, but variable numbers of heterotopic sebaceous glands (Fordyce granules) may be seen in this site. The dorsal tongue has a papillary architecture ( Fig. 72.2A ) with prominent parakeratin production, while the posterior lateral tongue has a surface epithelium with embedded taste receptors.
Common Benign Conditions of the Oral Cavity
Fordyce Granules
▪ Ectopic sebaceous glands ▪ Heterotopic or anomalous sebaceous glands ▪ Fordyce spots
- ▪
Common variation of normal sebaceous glands
- ▪
Multiple yellowish papules of the buccal mucosa and vermilion zone of the upper lip
Introduction
Fordyce granules are sebaceous glands that are located in the vermilion zone of the lips and the oral mucosa. Historically, these sebaceous glands have been considered ectopic; however, because they are such a common clinical finding in the oral cavity, Fordyce granules should be regarded as a variation of normal anatomy. “Free” sebaceous glands (see below) can also be found on the eyelids (meibomian glands), areolae (Montgomery tubercles), labia minora, and prepuce (Tyson glands).
Epidemiology
Fordyce granules are found in both sexes and are less common in children. The prevalence of Fordyce granules is as high as 70–85% in the adult population .
Clinical features
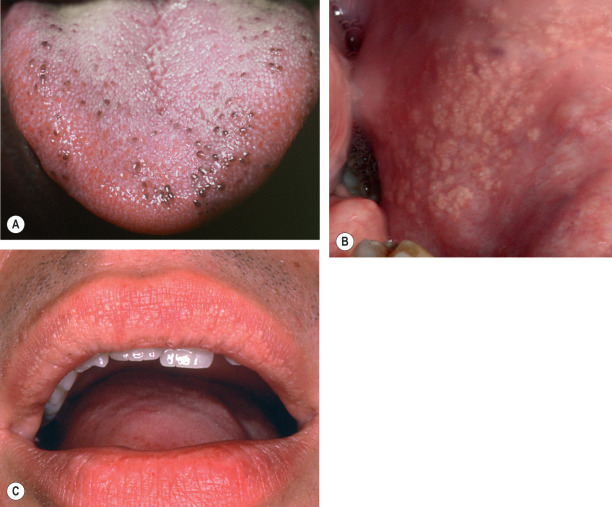
Fordyce granules characteristically present as asymptomatic, usually multiple, yellow to yellow–white papules ( Fig. 72.2B,C ). The papules are typically no more than 1–2 mm in diameter and often occur bilaterally. They are most commonly found on the buccal mucosa and the vermilion portion of the upper lip. Puberty appears to stimulate their development .
Pathology
Fordyce granules are similar to sebaceous glands found in the skin, but they are not associated with hair follicles, i.e. they are “free” sebaceous glands .
Treatment
Since Fordyce granules are considered a normal variation of anatomy, no treatment is required.
Geographic Tongue
▪ Benign migratory glossitis ▪ Oral erythema migrans ▪ Glossitis areata migrans ▪ Stomatitis areata migrans ▪ Benign migratory stomatitis
- ▪
Red, well-demarcated patches on the lateral and dorsal tongue with scalloped thin white borders
- ▪
Psoriasiform mucositis of unknown etiology
- ▪
No treatment is generally necessary
Introduction
Geographic tongue is a common, benign clinical condition of unknown etiology that primarily affects the tongue, although rarely it can occur in other sites within the mouth, such as the buccal mucosa, labial mucosa, and soft palate. When it occurs in sites other than the tongue, the tongue is almost always involved as well.
Epidemiology
Geographic tongue is a condition that affects both sexes, with a prevalence of 1–3% in the general population .
Clinical features
Geographic tongue typically presents on the dorsum of the tongue as multiple, well-delineated areas of erythema surrounded by a yellowish-white serpiginous border ( Fig. 72.3 ). Within the areas of erythema, there is atrophy of the filiform papillae. The lesions may begin as small white patches; these then develop a central red atrophic zone, which slowly increases in size. Many patients are unaware they have a geographic tongue, and it is often found on routine oral examination . Patients who are aware of their lesions frequently report that they appear to migrate. Geographic tongue is usually asymptomatic, although some patients may experience a burning sensation or sensitivity to hot or spicy foods. Some investigators have suggested that geographic tongue occurs with increased frequency in patients with psoriasis (including pustular psoriasis), in atopic individuals, and in patients with fissured tongue (discussed below).
Pathology
Because the microscopic features of geographic tongue are similar to those of psoriasis, the former is often termed “psoriasiform mucositis”. At the edge of the lesion, there is a characteristic loss of the papillary surface morphology of the tongue, elongation of rete ridges, varying hyperparakeratosis and spongiosis, and numerous, typically confluent, superficial intraepithelial neutrophilic microabscesses .
Treatment
Geographic tongue generally requires no treatment other than assuring the patient that it represents a benign condition. If symptoms such as burning, sensitivity, pain or tenderness are severe and interfere with the patient’s lifestyle, application of a potent topical corticosteroid gel may provide some relief.
Fissured Tongue
▪ Scrotal tongue ▪ Plicated tongue ▪ Lingua plicata ▪ Furrowed tongue
Introduction
Fissured tongue is a common, benign clinical condition of unknown etiology that affects the dorsum of the tongue. As the name implies, few to numerous furrows or grooves are seen on the dorsum of the tongue. Heredity appears to be a factor, with some evidence suggesting that the condition represents a polygenic trait, or an autosomal dominant trait with incomplete penetrance or expressivity. As mentioned previously, fissured tongue is often found in association with geographic tongue, suggesting that the same gene or genes may be associated with both conditions. Fissured tongue may be a component of the Melkersson–Rosenthal syndrome (see below). It is a common finding in Down syndrome and is occasionally seen in association with Cowden syndrome, pachyonychia congenita, and acromegaly (in the setting of macroglossia).
Epidemiology
Fissured tongue affects both sexes, with the reported prevalence ranging from 2–30% of adults, depending on the population studied .
Clinical features
Fissured tongue typically presents as multiple asymptomatic grooves on the dorsum of the tongue ( Fig. 72.4 ). The grooves are usually 2–3 mm in depth and can occur in isolated areas of the tongue (especially centrally) or involve the entire dorsal surface.
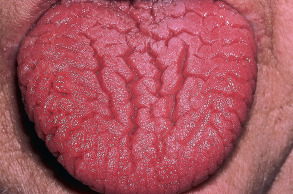
Treatment
Fissured tongue is a benign condition requiring no treatment.
Hairy Tongue
▪ Coated tongue ▪ Black hairy tongue ▪ Lingua villosa nigra
- ▪
Retention of keratin on the tips of filiform papillae
- ▪
Not due to yeast infection
- ▪
Best managed using a tongue scraper
Introduction
Hairy tongue is a common, benign clinical condition that represents accumulation of varying amounts of keratin on the dorsum of the tongue. Normally, the rate of keratin production on the dorsal tongue equals the rate at which keratin is naturally debrided during chewing and swallowing. If a patient has reduced food intake or a primarily soft or liquid diet, then the keratin simply accumulates. It is not a sign of any underlying systemic disease in an otherwise healthy and non-debilitated patient, as was historically believed. Factors that may be associated with increased production of keratin include smoking, poor oral hygiene, use of oxidizing mouthwashes, and hot beverages.
Epidemiology
Hairy tongue affects both sexes, with a prevalence reported as 0.5–5% of the population.
Clinical features
Hairy tongue presents as confluent hair-like projections on the dorsum of the tongue, especially its midportion ( Fig. 72.5 ). The elongated papillae are usually yellow to brown–black, although a wide range of colors may occur due to differential exogenous staining from food, tobacco, or chromogenic bacteria. Overgrowth of the latter may occur in the setting of systemic antibiotic therapy. Some patients complain of bad breath, bad taste, or a gagging sensation when the tongue contacts the palate. A distinctly different entity is pigmented papillae of the tongue, in which multiple, uniformly spaced, tiny brown papules (representing normal fungiform papillae) are seen on the tip and lateral aspects of the tongue in darkly pigmented individuals (see Fig. 72.2A ).
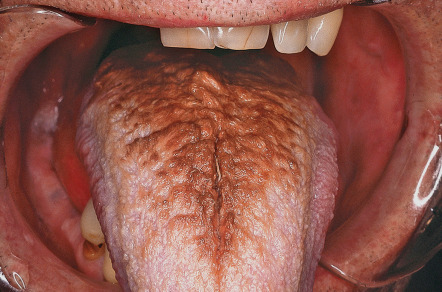
Pathology
Microscopic examination reveals pronounced accumulation of parakeratosis at the tips of otherwise normal filiform papillae. Often, bacteria can be seen colonizing the surface.
Treatment
Hairy tongue is a benign condition requiring no treatment. Some patients desire treatment due to the unaesthetic nature of the process, or because of altered taste, bad breath, or gagging sensation. In such cases, patients should be encouraged to scrape the tongue with a tongue scraper or brush it while tooth brushing.
Central Papillary Atrophy
▪ Median rhomboid glossitis ▪ Erythematous candidiasis
- ▪
Red, well-demarcated area in the midline of the posterior dorsal tongue
- ▪
Consistently associated with candidiasis
- ▪
Responds partially or completely to antifungal therapy
Introduction
Central papillary atrophy, previously referred to as median rhomboid glossitis, was originally thought to be a developmental defect resulting from failure of the embryonic tuberculum impar to be covered by the lateral processes of the tongue. However, it is not usually seen in children, but is found in up to 1% of adults. An association with chronic candidal infection has been demonstrated.
Clinical features
Central papillary atrophy appears as a well-demarcated, diamond- or oval-shaped area of erythema and atrophy on the dorsum of the tongue ( Fig. 72.6 ), anterior to the circumvallate papillae. The surface may be smooth or lobulated. It is usually asymptomatic but may also be associated with chronic multifocal candidiasis affecting the palate and angles of the mouth. While central papillary atrophy may represent a sign of HIV infection or other causes of immunosuppression, a multifactorial pathogenesis is now recognized and the condition is often seen in immunocompetent individuals .
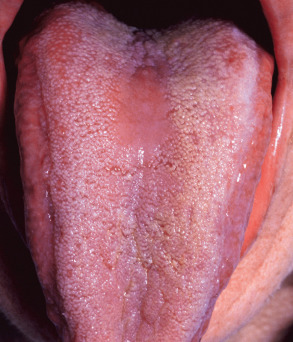
Pathology
Histologically, there is loss of the filiform papillae and tubular hyphae of Candida albicans are embedded within the parakeratin layer. However, the diagnosis is usually made clinically and confirmed by fungal culture or demonstration of pseudohyphae in a KOH preparation or a PAS-stained exfoliative cytologic preparation.
Treatment
The mucosal alteration resolves completely or partially after appropriate anticandidal therapy such as clotrimazole troches or oral fluconazole.
Syndromes of the Head and Neck
Basal Cell Nevus Syndrome
▪ Nevoid basal cell carcinoma syndrome ▪ Gorlin syndrome
Gardner Syndrome
▪ Familial colorectal polyposis
Osteomas most commonly develop in the maxilla and mandible, and along with bilateral CHRPE ( c ongenital h ypertrophy of the r etinal p igment e pithelium), represent early manifestations of this syndrome. Screening and treatment recommendations are discussed in Chapter 63 .
Multiple Endocrine Neoplasia Syndrome Type 2B
▪ Multiple endocrine neoplasia type 3 ▪ Multiple mucosal neuroma syndrome
The most distinctive feature of MEN 2B is the development of mucosal neuromas . Lesions often begin to appear during the first decade of life as soft, painless papules or nodules that favor the conjunctivae, lips and anterior tongue, but may also involve the buccal mucosa, gingiva, and palate. Medullary thyroid carcinoma develops in >90% of patients, usually during the first or second decade of life. Analysis of the RET gene should be performed in patients with clinical evidence of the disease as well as in asymptomatic individuals with an affected parent. Based upon the specific RET mutation detected, the recommended age for prophylactic thyroidectomy varies from infancy to 10 years.
Periodontal and Dental Disease
Necrotizing Ulcerative Gingivitis
▪ Trench mouth ▪ Acute necrotizing ulcerative gingivitis ▪ Necrotizing gingivostomatitis ▪ Vincent infection ▪ Fusospirochetal gingivitis
- ▪
Painful, hemorrhagic, necrotic gingivae with classic “punched-out” interdental papillae
- ▪
Mixed bacterial infection in susceptible hosts with predisposing risk factors
- ▪
Predisposing risk factors include immunosuppression, malnutrition, stress, smoking, and poor oral hygiene
Introduction
Necrotizing ulcerative gingivitis is a condition of the gums characterized by pain, bleeding, and necrosis of gingival interdental papillae. A mixed population of bacteria, including spirochetes, Prevotella intermedia , Fusobacterium , Treponema and Selenomonas spp., represents the primary etiology .
Epidemiology
Necrotizing ulcerative gingivitis affects both sexes and is seen primarily in young to middle-aged adults. Its prevalence in the normal population is <0.1%. Predisposing factors include immunosuppression, malnutrition, poor oral hygiene, psychological and physical stress, and smoking.
Clinical features
It is characterized by ulcerated, “punched-out” lesions of the gingival interdental papillae. There is generalized edema, erythema, hemorrhage, and necrosis of the gingivae. Other signs and symptoms include fever, malaise, lymphadenopathy, and often a foul odor from the mouth. Patients typically complain of “sore and bleeding gums”.
Pathology
Because the primary etiologic agents are bacteria belonging to the normal oral flora, bacterial culture results are nonspecific, as are the histopathologic findings. The diagnosis is based chiefly on characteristic clinical findings.
Treatment
In general, the most appropriate therapy is provided by a dentist or a dental specialist, such as a periodontist. Debridement after topical or local anesthesia should be the first step; this is often combined with broad-spectrum antibiotic therapy when systemic symptoms, such as fever and malaise, are present . Chlorhexidine oral rinses (0.12%) may reduce the bacterial load, and warm salt water rinses may be recommended for comfort.
Desquamative Gingivitis
- ▪
A clinical descriptor for oral manifestations of an underlying immune-mediated vesiculoerosive disease
- ▪
Characterized by diffusely smooth, red, and sometimes painful gingivae
- ▪
Histopathologic and immunofluorescence studies are required for appropriate diagnosis and subsequent treatment of the underlying condition
Introduction
The term desquamative gingivitis is used by clinicians to describe the gingival manifestations of several vesiculoerosive diseases. Clinically, the gingival epithelium is erythematous and spontaneously sloughs or can be removed with manipulation. Desquamative gingivitis is not a diagnosis; rather, it is a characteristic clinical pattern that may be seen with a number of immune-mediated conditions . Histopathologic and immunofluorescence (IF) studies are usually required to obtain a definitive diagnosis. This generally reveals a diagnosis of one of the following: lichen planus ( Ch. 11 ), mucous membrane (cicatricial) pemphigoid ( Ch. 30 ) or, less frequently, pemphigus vulgaris ( Ch. 29 ), lichenoid mucositis (idiopathic, drug-induced or foreign body-related), linear IgA bullous dermatosis, lupus erythematosus, epidermolysis bullosa acquisita, or chronic ulcerative stomatitis ( Fig. 72.7 ).
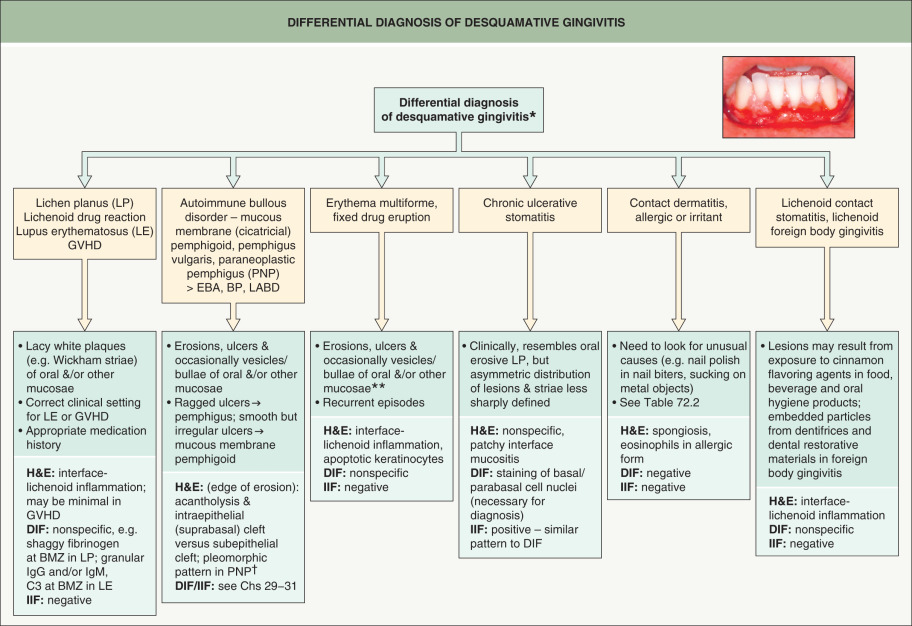
Epidemiology
The majority of individuals affected with desquamative gingivitis are >40 years of age, and most are women .
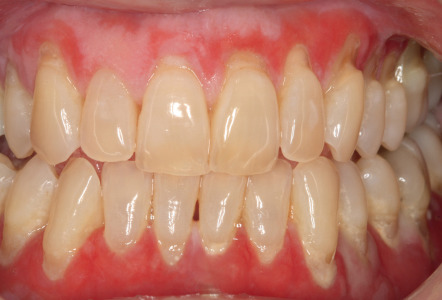
Clinical features
Patients with desquamative gingivitis usually present with the chief complaint of “sore gums”. The most common clinical manifestations include diffuse gingival erythema with varying degrees of mucosal sloughing and erosion ( Fig. 72.8 ). Vesicle or blister formation involving the gingivae may also be seen.
Pathology
The histopathologic and immunopathologic features of desquamative gingivitis depend on which of the aforementioned vesiculoerosive diseases it represents. Patients should be evaluated by incisional biopsy and specimens should be processed for both routine histology and direct IF microscopy . In addition, serum can be sent for indirect IF testing or ELISA to detect circulating autoantibodies. Specific findings will depend upon the underlying disease.
Treatment
Once a diagnosis is established, appropriate treatment for the responsible condition can follow. Meticulous dental prophylaxis (cleaning), oral hygiene instructions, and longitudinal evaluation are recommended. Reducing bacterial plaque often decreases the severity of lesions and is an important adjunct to medical therapy .
Sequelae of Dental Caries
- ▪
Dental infections can spread beyond the periapical region of teeth
- ▪
Periapical abscess, osteomyelitis, cellulitis, intraoral dental sinus, and cutaneous sinus are potential sequelae
- ▪
Emergent cases require immediate treatment to avoid airway obstruction or possible sepsis
Introduction
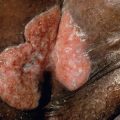
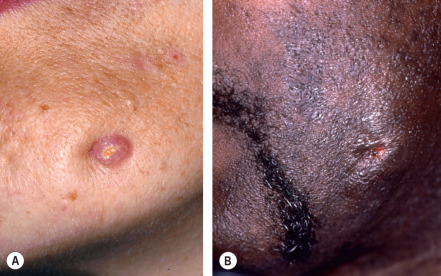
Bacterial infections involving carious teeth can sometimes spread beyond the pulp of the tooth. When they do, they often invade the periapical area of the tooth within the bone. The accumulation of inflammatory cells and bacterial debris at the apex of a non-vital tooth is termed a periapical abscess . Without resolution, there is the possibility that the abscess may extend further into medullary bone and cause osteomyelitis . If the host immune defenses are compromised or if the invading bacteria are particularly virulent, this lesion may progress, perforating the cortical bone and extending into the overlying soft tissues and fascial spaces as cellulitis . This usually manifests as a tender, diffuse swelling of facial soft tissues. If the bacteria are only moderately virulent, the infection may be contained but will eventually erode through to either an oral mucosal surface (resulting in an intraoral dental sinus tract ; “ parulis ”) or facial skin, producing a cutaneous sinus of dental origin ( Fig. 72.9 ). If a chronic path of drainage is achieved by an abscess, patients may be asymptomatic because of a lack of accumulation of purulent and inflammatory material. Without drainage, signs and symptoms of acute inflammation may become evident.
Clinical features
The clinical presentation of cellulitis includes diffuse, tender swelling of facial soft tissues, often accompanied by erythema. The areas involved and the severity of the infection can vary tremendously. Dysphagia, headache, fever, lymphadenopathy, and pain may be present. Sepsis is a less common complication. Two potential complications represent a particular danger to patients and demand immediate attention. Ludwig’s angina presents as swelling of the submandibular, submental, and sublingual spaces. Urgent attention is required for this form of cellulitis because it can rapidly spread to the retropharyngeal space, obstructing breathing, and even spread to the mediastinum. Cavernous sinus thrombosis due to a dental abscess may initially present as swelling of the canine space. Infraorbital, ocular, and eventually CNS involvement may develop.
An intraoral dental sinus tract is characterized by a soft, nontender, erythematous papule that develops on the alveolar process, typically in the same region as the apex of the offending non-vital tooth. Such sinus tracts are frequently asymptomatic, but patients may describe periodic episodes of a salty or bitter taste that probably coincides with the discharge of purulent exudate. A cutaneous sinus of dental origin appears as an erythematous papule, often with an ulcerated or umbilicated center, or as a depression with associated fibrosis (see Fig. 72.9 ). Cervicofacial actinomycosis must be excluded (see Ch. 74 ).
Treatment
Treatment involves elimination of the focus of infection, which often requires incision and drainage, endodontic “root canal” therapy or extraction of the involved tooth, appropriate antibiotic therapy, and airway management, if necessary.
Physical and Chemical Injuries
Fibroma
▪ Irritation or traumatic fibroma ▪ Fibrous hyperplasia or nodule
- ▪
Smooth, pink, soft nodule, typically on the buccal mucosa
- ▪
Asymptomatic unless persistently irritated or traumatized
Introduction
Fibroma is the most common “tumor” of the oral cavity, but it most likely represents reactive hyperplasia of fibrous connective tissue in response to local trauma.
Epidemiology
Irritation fibromas most commonly occur during the fourth to sixth decades. Women outnumber men 2 : 1, based on biopsy specimens submitted for examination.
Clinical features
The typical lesion is a sessile, soft, smooth-surfaced pink papulonodule similar in color to normal mucosa and located along the bite line of the buccal mucosa. They also occur on the labial mucosa, tongue and gingiva and are usually <1.5 cm in diameter.
Pathology
Microscopic examination shows an unencapsulated mass of hyperplastic fibrous connective tissue with minimal inflammation. The overlying epithelium may be atrophic or hyperkeratotic secondary to friction or biting; sometimes there is also ulceration.
Treatment
Irritation fibromas are cured by conservative surgical excision. It is important to submit the tissue for histopathologic examination because other benign or malignant tumors may clinically resemble fibromas.
Chemical Burn
▪ Aspirin burn ▪ Mucosal burn
- ▪
“Burns” can occur when caustic chemicals come into contact with the oral mucosa
- ▪
A superficial, white, sloughing membrane represents coagulative necrosis of the epithelium
- ▪
Treatment consists of removal of the offending agent and palliative care
Introduction
Numerous chemicals and drugs come into contact with the oral mucosa, and some of these are caustic enough to cause superficial burns . A variety of agents have been reported as being responsible for these burns, including aspirin, vitamin C tablets, hydrogen peroxide, silver nitrate, phenol, eugenol, rubbing alcohol, battery acid, gasoline, various dental materials, and many over-the-counter toothache medicaments . Aspirin (acetylsalicylic acid, pH 3.5) is one of the most commonly recognized medications responsible for mucosal damage . Patients often place a tablet on the oral mucosa overlying a sore tooth in an attempt to alleviate dental pain, and the resultant continuous contact produces injury.
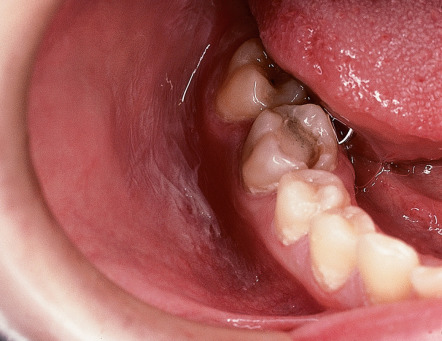
Clinical features
Most caustic agents cause characteristic damage to the mucosa. This may appear initially as just an erythematous zone. With greater concentration or longer duration of exposure to the chemical, a wrinkled white membrane that represents superficial necrosis becomes evident ( Fig. 72.10 ) .
Pathology
Microscopic evaluation reveals coagulative necrosis of the epithelium. The necrosis begins on the surface and may extend partially into the epithelium, or it may involve its entire thickness.
Treatment
A chemical burn is a self-resolving condition requiring no treatment except for discontinuing exposure to the offending agent. Application of emollient pastes or topical anesthetics as a palliative measure may provide temporary relief.
Morsicatio Buccarum
▪ Chronic cheek chewing ▪ Chronic cheek biting
- ▪
Factitial habit
- ▪
Bilateral shaggy white lesions of the anterior buccal mucosa
- ▪
No treatment indicated
Introduction
Chronic irritation or injury to the buccal mucosa from repetitive chewing, biting, or nibbling produces a characteristic appearance clinically . Similar changes may also be observed on the labial mucosa and lateral tongue, referred to as morsicatio labiorum and morsicatio linguarum , respectively.
Clinical features
Morsicatio buccarum typically presents as bilateral shaggy white lesions of the anterior buccal mucosa that approximate the area where the upper and lower teeth meet (occlusal plane). The white areas are ragged and shredded ( Fig. 72.11 ). Foci of erythema, erosion, or traumatic ulceration are rarely identified.
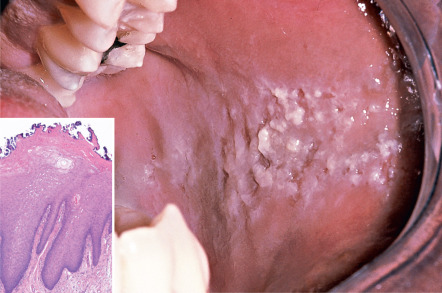
Pathology
Microscopic evaluation reveals marked hyperparakeratosis of the surface epithelium, which demonstrates a ragged morphology (see Fig. 72.11 inset). Varying degrees of vacuolization and spongiosis are observed in the superficial portion of the epithelium. Surface colonization by bacteria is also a frequent feature. Although these findings are relatively nonspecific, in the appropriate clinical setting, they are highly suggestive of this factitial condition.
Treatment
Morsicatio buccarum is a benign disorder requiring no treatment.
Traumatic Ulcer
- ▪
Acute or chronic injury to the oral mucosa may cause a traumatic ulcer
- ▪
Commonly occurs on the tongue, lips, or buccal mucosa
- ▪
May mimic oral squamous cell carcinoma
- ▪
Self-limited process that heals within 3–6 days following removal of etiologic agent
Introduction
The etiology of a traumatic ulcer is related to acute or chronic injury to the oral mucosa. The injury is often mechanical in nature, such as biting the tongue or cheek accidentally, or lacerating the mucosa with sharp foods or objects (e.g. the sharp edge of a fractured tooth).
Clinical features
Traumatic ulcers are moderately painful lesions. They are characterized by mild erythema surrounding a central ulcer that is covered by a yellow fibrinopurulent membrane. Often there is a white border of hyperkeratosis surrounding the ulcer ( Fig. 72.12 ). Traumatic ulcers occur most commonly on the tongue, lips and buccal mucosa, although other sites may be involved. They are usually self-limiting, if the etiologic agent is removed, and generally heal within 3–6 days. Since traumatic ulcers can clinically mimic oral squamous cell carcinomas (SCCs), biopsy is indicated if the ulcer does not resolve within 2–3 weeks following elimination of potential causes.
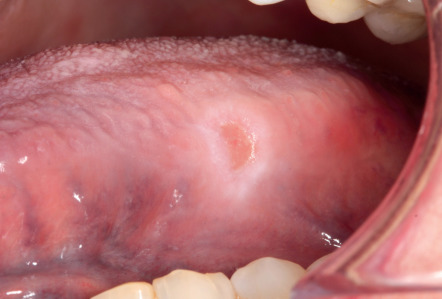
Treatment
Remove any obvious causative agent. Application of a thin protective film of cellulose compound, with or without a topical anesthetic as a palliative measure, may provide some relief.
Drug-Related Gingival Enlargement (Hyperplasia)
- ▪
Enlargement of the gums occurs during the first year of drug administration
- ▪
Most commonly associated with phenytoin, nifedipine, and cyclosporine
- ▪
Severity correlated with patient susceptibility and level of oral hygiene
Introduction
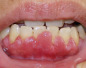
Multiple systemic medications have been reported to be associated with gingival enlargement ( Table 72.1 ), but a high prevalence has only been noted with phenytoin (50%), nifedipine (25%), and cyclosporine (25%). Although it is still controversial whether the risk of developing this side effect is dose-related, there is a definite correlation with poor oral hygiene.
 | CAUSES OF GINGIVAL ENLARGEMENT (HYPERPLASIA, HYPERTROPHY) | 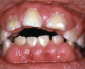 |
| Systemic medications | ||
| Common | ||
| Anticonvulsants ( phenytoin , phenobarbital, lamotrigine, valproate, vigabatrin, ethosuximide, topiramate, primidone) | ||
| Calcium channel blocking agents ( nifedipine , amlodipine, diltiazem, felodipine, verapamil) | ||
| Cyclosporine * | ||
| Uncommon | |
|---|---|
| Antibiotics (erythromycin, trimethoprim-sulfamethoxazole [co-trimoxazole]) | Sertraline |
| Lithium | Oral contraceptives, estrogens |
| Amphetamines | |
| Additional causes | |
| |
Clinical features
During the first year of drug administration, gingival enlargement commences in the interdental papillae of the anterior teeth on the labial side. The hyperplasia may extend over part or all of the involved teeth. Edentulous areas are usually spared, but significant hyperplasia under poorly maintained dentures has been reported. The enlarged gingiva is usually normal in color, firm, and nodular or lobulated. However, associated inflammation can lead to a red, edematous, or friable appearance.
Pathology
Microscopically, redundant tissue of normal composition or an increased amount of collagen with a normal density of fibroblasts is seen. When the specimen is clinically inflamed, there is increased vascularity and a chronic inflammatory cell infiltrate.
Treatment
A rigorous regimen of oral hygiene consisting of frequent professional cleaning and home plaque control utilizing chlorhexidine rinses helps to prevent drug-associated gingival enlargement. Discontinuation of the suspected offending drug or even substitution with another drug of the same class may result in cessation or some regression of the gingival hyperplasia.
If all else fails, extirpation of the excess gingival tissue, a procedure normally performed by a periodontist, remains the treatment of choice. Samples of excised tissue should be submitted for histopathologic evaluation as there are other causes of gingival enlargement (see Table 72.1 ).
Allergic and Immunologic Diseases
Contact Stomatitis
▪ Oral mucosal cinnamon reaction ▪ Lichenoid amalgam reaction ▪ Contact lichenoid reaction
- ▪
Cinnamon flavoring and dental amalgam (silver-containing filling material) may cause lichenoid changes of the oral mucosa
- ▪
Red, white or mixed lesions are seen, with varying degrees of erosion, ulceration, or striae formation
- ▪
Lesions may be located where the offending agent contacts the mucosa for a prolonged period of time
- ▪
Removal of the offending agent should be curative
Introduction
Numerous foods, artificial flavorings, food additives, and materials used in dentistry have been implicated as etiologic agents of allergic or irritant contact stomatitis ( Table 72.2 ). Two of these, namely cinnamon flavoring (found in many foods, gums, candies, and dentifrices) and dental amalgam (“silver” dental fillings), may lead to unique clinicopathologic features.
| POTENTIAL CAUSES OF CONTACT STOMATITIS OR CHEILITIS |
| Allergic |
|
| Irritant |
|
Epidemiology
The frequency of true allergic contact reactions is probably not as high for the oral mucosa as for the skin. The reduced reactivity of the oral mucosa has been attributed to many factors, including the brief period of contact of agents with the mucosa, more rapid dispersal and absorption of antigens, and the diluting and neutralizing properties of saliva.
Clinical features
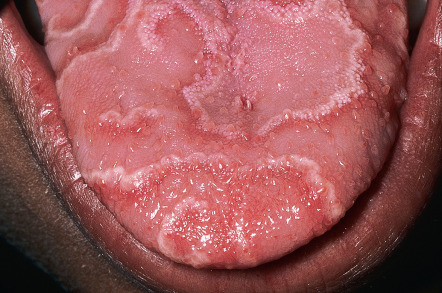
The clinical features of contact stomatitis from cinnamon flavoring can vary; sometimes it produces localized lesions on the buccal mucosa or lateral borders of the tongue that appear as shaggy white keratotic areas ( Fig. 72.13 ). The buccal lesions may resemble oral lichen planus or morsicatio buccarum clinically, while the tongue lesions may resemble oral hairy leukoplakia or SCC. Erosion, ulceration, and erythema can also be seen. Gingival involvement appears as swelling and erythema.
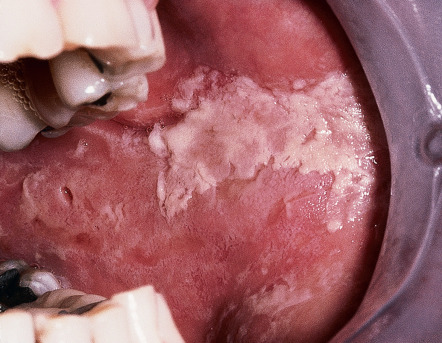
Contact stomatitis from dental amalgam occurs mainly at sites adjacent to amalgam restorations, such as the posterior buccal mucosa and ventrolateral surface of the tongue . When the mucosa is relaxed, the lesion characteristically directly contacts the amalgam restoration, which is often on the buccal or lingual (rather than occlusal) surface of the tooth. The lesions may be white or erythematous, with or without peripheral striae, and can resemble oral lichen planus. Ulceration or erosion may also be observed. With both amalgam and cinnamon reactions, pain is not a universal finding although it may be present.
Pathology
Biopsy specimens of contact stomatitis from dental amalgam or cinnamon flavoring often demonstrate a lichenoid mucositis. The cinnamon reaction may have a psoriasiform pattern, whereas, in general, patients with contact stomatitis exhibit spongiosis histologically. In lichenoid mucositis, the epithelium may have hyperkeratosis, basilar crowding and atypia, atrophy of the spinous layer, lymphocytic exocytosis, and, sometimes, ulceration. Immediately subjacent to the epithelium is a band of chronic inflammatory cells that, in some areas, effaces the rete ridge architecture. A perivascular infiltrate composed primarily of lymphocytes with occasional plasma cells frequently surrounds small blood vessels. Longstanding amalgam reactions are characterized by a much broader band of lichenoid inflammation compared to lichen planus, and it is not unusual to find well-defined germinal centers within the lymphocytic infiltrate.
Treatment
Contact stomatitis due to cinnamon flavoring resolves within a week of discontinuing the offending product, but may reappear quickly with reuse of the product or other cinnamon-containing agents, confirming the cause. When contact stomatitis secondary to amalgam is suspected, patch testing may be performed and should include organic and inorganic mercury as well as amalgam and the other dental metals. The offending restoration is typically treated conservatively by polishing, smoothing and recontouring (in addition to cleaning the teeth to remove plaque), but if these measures fail, appropriate alternative dental materials, such as composite (a white plastic-like material), porcelain fused to metal or gold, can be used to replace the amalgam restoration .
Recurrent Aphthous Stomatitis
▪ Recurrent oral aphthae ▪ Aphthous ulcers ▪ Canker sores ▪ Simple or complex aphthosis
- ▪
The three forms of simple recurrent aphthous stomatitis are minor, major, and herpetiform
- ▪
Lesions are painful, typically <5 mm in diameter, round to oval, and appear creamy-white with an erythematous halo
- ▪
The pathomechanism is unknown, but immunologic factors may be involved
- ▪
Usually controlled through the use of topical corticosteroids
Introduction
Recurrent aphthous stomatitis is a relatively common ulcerative disorder of the oral cavity. While the etiology remains obscure, it is likely a multifactorial disorder influenced by a host of predisposing factors. Recurrent aphthous stomatitis is believed by some to be the result of immunologically mediated damage to epithelial cells . This abnormal immune response may be triggered or influenced by trauma, hematinic (iron, folate or vitamin B 12 ) deficiencies, hormonal fluctuations, psychological stress, infectious agents, food hypersensitivities, genetic factors, and HIV infection. Complex aphthosis, the nearly constant presence of more than three oral aphthae or oral and genital aphthae, requires the exclusion of Behçet disease, inflammatory bowel disease, cyclic neutropenia, vitamin deficiencies (e.g. B 12 , folate), HIV infection, and other disorders listed in Table 72.3 .
| RECURRENT APHTHAE – ETIOLOGIES AND DIFFERENTIAL DIAGNOSIS |
| Etiologies |
|
| Differential diagnosis |
|
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree