(1)
Professor of Plastic Surgery, Director of Diabetic Wound Center, Director of Cell Therapy Laboratory, Korea University College of Medicine and Korea University Guro Hospital, Seoul, Republic of Korea (South Korea)
Abstract
Negative-pressure wound therapy (NPWT) or vacuum-assisted closure (VAC) is a mode of therapy used to encourage wound healing. In 1997, Morykwas et al. first described applying controlled suction through a foam to create a favorable environment for wound healing, which they defined as NPWT. This therapy has been widely and successfully used over the last 15 years although the physiologic basis of its effects is not yet fully understood. NPWT has dramatically changed the treatment of chronic and complex wounds. It can be also used as an adjunct treatment for temporary closure and wound preparation preceding surgical procedures such as skin grafts and flap surgery. Randomized controlled clinical trials have shown that NPWT increases the wound healing rate. In this chapter, composition of NPWT device, mechanism of action, application method, benefits, indications, contraindications, adverse effects, and innovations are described. However, although NPWT is a useful modality for wound healing, there is a potential problem for the treatment of diabetic foot ulcers. NPWT decreases tissue oxygenation in wound beds by the external compression exerted by the foam and pad of the connecting tube. Moreover, the foot is the most sensitive region in the body to ischemia, and diabetes is often accompanied by atherosclerosis. Therefore, when an NPWT is applied to a diabetic foot ulcer, care should be taken to minimize compression pressure. The author’s applying method of NPWT for diabetic foot ulcers is presented in this chapter.
Keywords
Negative pressureVACDiabetic ulcerComposition of NPWT Device
NPWT is a relatively new technology in wound management. The general system is made up of 5 major components: (1) a filler material placed into the wound (most commonly a foam sponge with pores), (2) a semipermeable film to isolate the wound environment and allow the vacuum system to transmit subatmospheric pressures to the wound bed, (3) a connecting suction (drainage) tube, (4) a suction device, and (5) an exudate-collecting canister. The device permits the negative pressure to be distributed evenly throughout the foam and improves exudate drainage.
The structure of the filler material packed into the wound is important in the efficacy of NPWT. The traditional polyurethane ether foam is hydrophobic, whereas the polyvinyl alcohol and polyurethane ester foams are more hydrophilic. The traditional polyurethane ether foams have been often used for wounds with heavy exudates and for stimulating granulation tissue formation. In contrast, the polyvinyl alcohol sponges have been used in cases when delicate underlying structures, such as tendons or blood vessels, need to be protected. The polyurethane ester devices are designed for use with instillation therapy. In addition, a foam with smaller pores can help to restrict ingrowth of granulation tissue, thereby diminishing pain associated with dressing changes and reducing risk when hypergranulation is a concern.
Mechanism of Action
Numerous controversies exist regarding the mechanism of action, especially with regard to tissue perfusion. Action mechanisms of NPWT can be broadly classified as primary mechanisms and their associated secondary effects. Four primary mechanisms of action have been proposed: (1) wound shrinkage or macrodeformation, (2) microdeformation at the wound bed, (3) exudate/fluid removal, and (4) creation of the favorable wound environment. There are also several secondary effects including angiogenesis and neurogenesis.
Wound Shrinkage or Macrodeformation
Wound shrinkage is caused by collapse of the pores of a foam and centripetal forces exerted onto the wound bed by the negative pressure. Polyurethane foams exposed to −125 mmHg suction can significantly decrease the foam volume leading to decrease in wound surface area. The extent of shrinkage is largely dependent on the mobility of the wound area. For example, in areas such as the abdomen, significant shrinkage can be observed, but the low elasticity of scalp skin over the rigid calvarium is likely to less contract.
Microdeformation
Microdeformation occurs on the wound bed by the porous filler material when exposed to negative pressure. Based on a computer simulation, NPWT using −110 mmHg causes an average tissue strain of 5–20 % over most of the wound bed. These mechanical forces appear to be transmitted to individual cells. The cells are subjected to a variety of mechanical forces, including shear and hydrostatic pressure from wound exudates, stretch and compression from their surrounding extracellular matrices (ECMs), and the pull of gravity. Microdeformation is the morphologic result of these integrated mechanics. Cell shape has been well established to govern its behavior. Cellular deformation and associated cell stretch caused by NPWT induce cell proliferation, migration, and differentiation, thereby promoting wound healing. This is supported by evidence that short (6 h) intermittent applications of NPWT to a diabetic mouse model caused an extended proliferative cell response.
Exudate/Fluid Removal
Excessive fluid in the extracellular space, which is commonly seen as edema, is a contravening factor in wound healing, owing to the compressive effect it can exert on local cells and tissues. Individual cells generate intrinsic tension via their cytoskeleton and interactions with the ECMs, inducing a proliferative response. Elevated fluid pressures in the interstitium diminish this response by dampening intrinsic tension buildup. Fluids from the extracellular space appear to communicate with the wound surface. Applying negative pressure to this surface results in fluid removal from the extracellular space. Fluid removal likely reduces compression of the microvasculature, optimizing tissue perfusion potentially by allowing increased blood flow to the area. Toxins from the wound, bacteria, and exudate can also be removed with the fluids.
NPWT also likely reduces the amount of fluids that must be cleared through the lymphatic system. In addition, NPWT induces a gradual increase in lymphatic density at wound edges, thereby improving drainage.
Creation of the Favorable Wound Environment
The filler material and semiocclusive film act as thermal insulators to maintain wound warmth. Removal of excess extracellular fluid reduces the required diffusion distance at the capillaries, resulting in optimized tissue perfusion and aiding in the healing process. Semiocclusive polyurethane film maintains subatmospheric pressures at the wound bed and prevents evaporative water losses. The dressing is impermeable to bacteria, decreasing the risk of wound infection. In addition, the film dressing exhibits limited permeability to water vapor and other gases, helping to maintain a stable moist wound environment. Various types of NPWT can also improve the wound environment. Instillation of bioactive factors and using a silver-impregnated foam are examples of how the favorable wound environment can be created.
Secondary Effects
The application of NPWT actually generates a paradoxical increase in pressure below the wound bed. It has been suggested that the temporary reduction of blood flow at the wound edge stimulates angiogenesis. NPWT demonstrates increased microvessel density during chronic wound treatment. In vitro studies using intermittent NPWT also achieved similar results of angiogenic response stimulation.
NPWT promotes neural growth and neuropeptide expression (substance P, calcitonin gene-related peptide, and neurotrophin nerve growth factor).
The influence of NPWT on wound bioburden remains controversial, particularly in terms of the variety of responses that may be elicited by different bacterial strains.
Clinical Application
Application Method
NPWT should be applied after the wounds are debrided and are clean of eschar, slough, and nonviable tissue. Sterile foam is cut to the wound size and placed in a wound bed. Any areas of undermining or tunneling should also be filled with foam pieces. Skin sealant is used with padding of periwound area to prevent excessive pressure from tubing and skin irritation. The wound is covered with film drape to ensure an airtight seal. The tube clamps are opened, and the pump is turned on to allow wound fluid and bacteria to travel into the collecting canister. The dressing is left in place for 48–72 h (12–24 h, if infected). Most studies have used −125 mmHg of pressure.
NPWT can be used until the wound is closed or used in preparation for surgical closure such as a skin graft or flap. Many suction pumps have been designed to allow continuous or intermittent application. Although there is basic research showing a greater response using intermittent therapy, the optimal mode of application has not been determined.
NPWT should be applied in conjunction with treatment of other comorbid diseases such as diabetes, hypertension, peripheral vascular disease, and venous stasis disease.
Benefits
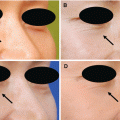
When using NPWT devices in wounds, significant shrinkage of the wound can be observed as the wound edges come together by negative pressure applied on a foam. At the wound interface, the foam creates microdeformations that stretch cells on wound bed and activate molecular pathways for cell migration, proliferation, ECM synthesis, and angiogenesis. NPWT also removes a large amount of exudate or fluid, resulting in decreased tissue edema. In addition, a semiocclusive film covering the wound reduces contamination from outside bacteria and provides closed moist, warm wound healing environment. NPWT also reduces cell death caused by dehydration. Additionally, NPWT has the advantage in that it often only need to be changed every 2–3 days, reducing the number of dressing changes for patients (Fig. 8.1).
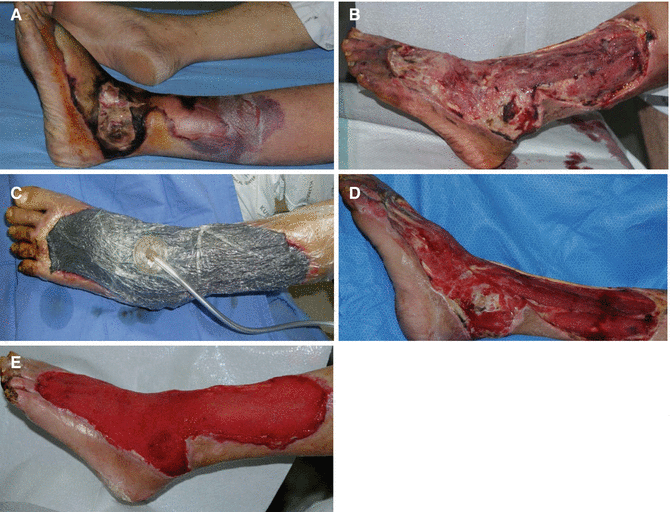

Fig. 8.1
NPWT is useful to encourage wound healing. (A) A severely infected wound with spreading cellulitis, which originated from the lateral malleolus. (B) The wound was first surgically debrided to remove infected tissues. (C) NPWT was applied after the wound was clean of eschar and nonviable tissue. (D, E) The wound bed was successfully prepared for skin graft
Indications
NPWT can be used for a wide range of wounds, including poorly healing ulcers. These devices have been commonly used to treat pressure sores, diabetic ulcers, venous or arterial ulcers, open abdomens, sternal wounds, traumatic wounds, burn wounds, dehisced wounds, and skin graft recipient sites.
In the treatment of pressure ulcers, serial randomized controlled trials (RCTs) demonstrated a reduction in wound area and volume, improved granulation, and a reduced frequency of hospitalization in patients undergoing NPWT. In a retrospective cohort study evaluating chronic diabetic, arterial, and venous ulcers in high-risk patients, treatment with NPWT was shown to increase the incidence of closure. Earlier application of NPWT to these wounds also results in faster healing times. In the treatment of diabetic foot ulcers, NPWT promotes wound area reduction, wound bed granulation, and microbial clearance, thereby enabling a higher rate of limb salvage, especially in Wagner grade 3 and grade 4 ulcers.
Additional indications for NPWT include wounds with exposed bone or tendons and wounds with exposed hardware.
Contraindications
Because inappropriate use of NPWT devices has the potential for patient harm, the clinician should carefully weigh the risks, benefits, and alternatives of using NPWT. NPWT is contraindicated for use on severely ischemic wounds, dry wounds, necrotic wounds or eschar, wounds in body cavities, over malignancies, untreated fistulas, untreated osteomyelitis, and exposed organs. Caution should be taken when NPWT is applied around exposed nerves or vascular structures such as the heart or large blood vessels because erosion may occur, resulting in massive blood loss. Caution must also be taken when placing the foam in contact with visceral organs because erosion can occur, leading to fistula. The porous filler material is highly efficient at removing blood; therefore, hemostasis should be nearly complete before application of negative pressure, and NPWT must be used with caution in patients with coagulopathies. Suction devices should have appropriate overflow alarms to alert clinicians when there is excessive blood loss. NPWT should not be applied immediately after an incision and drainage procedure to allow for adequate wound visualization.
However, in some cases, clinicians have used NPWT despite the established contraindications. For example, some studies have successfully implemented NPWT in cases of exposed organs,
Adverse Effects
Skin irritation can be developed from the adhesive drape. Odor may be emitted from the dressing or canister or both. They can be mitigated by a brief interruption of therapy for a few days. Dermatitis can be treated with topical creams or ointments, depending on the etiology. Some patients do not tolerate the therapy because of pain or sensitivity to the adhesive drape or the foam. Pain may be relieved by decreasing the pressure on the device.
Innovation
There is much innovation in the NPWT device. Instillation devices have been designed that allow delivery of a variety of irrigation solutions, including antimicrobials. NPWT devices can be combined with other wound advances such as artificial/allogenic dermis, processed allogeneic or xenogenic materials, or antimicrobial dressings.
Instillation therapy provides a methodology to intermittently add fluid to the wound through either the same or a different port on the NWPT connecting tubing. It is possible to instill normal saline or other agents to help wound healing such as antimicrobial agents. Bioactive factors which may be introduced through the continuous-instillation system include doxycycline, dilute Betadine, platelet gel, and activated protein C, an anticoagulant.
Combination of NPWT with dermal scaffolds provides excellent immobilization and contact between the scaffold and wound bed. A multicenter RCT demonstrated related long-term effects when combining dermal scaffolds with NPWT; scars were observed to have greater elasticity and more natural skin pigmentation 12 months postoperatively, while the rate of postoperative wound contamination decreased.
Traditional NPWT uses polyurethane ether foam; however, its effect on bacteria in the wound has been inconsistent. To attempt to reduce the amount of bacteria in the wound, silver has been added to the coating of the foam. Stinner et al. investigated this potential therapy by simulating proximal leg wound infection in a goat model; silver dressings were placed beneath negative pressure dressings in wounds inoculated with bacteria. The authors observed a decline in bacterial load (particularly S. aureus) with this treatment compared with standard NPWT.
NPWT in Diabetic Foot Ulcers
Diabetic foot ulcers present a difficult treatment problem and respond poorly to conventional treatment. The main reason that wound healing often cannot be achieved is that the pathophysiology of this condition involves many factors. For example, fibroblasts isolated from diabetic patients have lower migration and proliferation potentials and cause changes associated with cellular senescence in the presence of high glucose concentrations. Wound fluid from diabetic ulcers also inhibits fibroblast proliferation in many cases because of excessive metalloproteinase (MMP) levels and depressed levels of their natural inhibitors and tissue inhibitors of metalloproteinases (TIMPs). In addition, even low concentrations of microbes in wound beds can delay or prevent wound healing because many diabetic patients do not have appropriate defense mechanisms against bacterial invasion due to an immunocompromised status or a poor systemic profile.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree