Abstract
This chapter discusses leprosy, tuberculosis, and non-tuberculous mycobacterioses that can present with cutaneous lesions. Leprosy, a chronic infection with Mycobacterium leprae , is a slowly progressive disease with a predilection for the skin and peripheral nerves. M. tuberculosis infection may lead to cutaneous lesions that are due to exogenous exposure (e.g. tuberculous chancre, tuberculosis verrucosa cutis) or endogenous infection (e.g. scrofuloderma, miliary tuberculosis, lupus vulgaris) as well as reactive tuberculids. Non-tuberculous mycobacterioses are caused by mycobacterial species that are found in nature. These organisms can produce cutaneous disease by inoculation into the skin and disseminated infection may occur in immunocompromised hosts. Diagnosis and treatment of all these infections will be addressed.
Keywords
leprosy, Hansen disease, tuberculosis, cutaneous tuberculosis, mycobacteria, mycobacteriosis, tuberculous chancre, tuberculosis verrucosa cutis, scrofuloderma, lupus vulgaris, tuberculous gumma, non-tuberculous mycobacteria, atypical mycobacteria, Buruli ulcer, swimming pool granuloma, tuberculids, papulonecrotic tuberculid, lichen scrofulosorum, erythema induratum
In 1896, the name “mycobacteria” was given to a large group of bacteria that produce filamentous pellicles similar to molds when grown in liquid media. These organisms are thin, slightly curved to straight, non-motile and non-spore-forming, with a waxy coating that makes them resistant to most stains. Mycobacteria are also acid-fast, which means they are not readily decolorized by acid after staining . A number of the >50 species in the genus Mycobacterium may or may not be pathogenic depending on the immunologic state of the host , whereas M. leprae and M. tuberculosis are virulent and constitute an important world health problem. Research on leprosy has been greatly hampered by the inability to culture the organism, which has the longest doubling time of all known bacteria, but the discovery of the genomic sequence of M. leprae in 2001 was a significant advancement.
Although the classic mycobacterioses, leprosy and tuberculosis, have been known since antiquity, in the past few decades there has been a worldwide explosion in the incidence of mycobacterial infections. Several factors have played a role in this increase, including the emergence of resistant strains of M. tuberculosis and a rise in the number of immunocompromised hosts, including those with AIDS . In Table 75.1 , the different species of mycobacteria that are associated with cutaneous disease are outlined utilizing Runyon’s classification scheme .
| MYCOBACTERIA THAT CAUSE CUTANEOUS DISEASE | ||
|---|---|---|
| Group and pigment | Rate of growth | Pathogens |
| Slow growers | ||
| Photochromogens * | 2–3 weeks | M. kansasii, M. marinum, M. simiae |
| Scotochromogens † | 2–3 weeks | M. scrofulaceum, M. szulgai, M. gordonae, M. xenopi |
| Nonchromogens ‡ | 2–3 weeks | M. tuberculosis, M. avium, M. intracellulare, M. ulcerans, M. haemophilum, M. malmoense, M. terrae, M. genavense, M. bovis § , M. nonchromogenicum |
| Rapid growers | 3–5 days | M. fortuitum, M. chelonae, M. smegmatis, M. abscessus ** , M. immunogenum, M. goodii, M. wolinskyi, M. cosmeticum, M. mucogenicum , M. mageritense |
| Non-cultured (to date) | M. leprae | |
* Capable of yellow pigment formation upon exposure to light.
† Capable of yellow pigment production without light exposure.
‡ Incapable of pigment production.
§ Including bacillus Calmette–Guérin.
** Including M. abscessus subsp. massiliense and M. abscessus subsp. bolletii .
This chapter addresses the mycobacterial species that are of greatest interest to dermatologists: M. leprae, M. tuberculosis , and non-tuberculous mycobacteria that cause cutaneous infections.
Leprosy
▪ Hansen disease
- ▪
Chronic infectious disease caused by the bacillus Mycobacterium leprae , an intracytoplasmic parasite of macrophages and Schwann cells
- ▪
Slowly progressive condition characterized by granulomas and neurotropism, with a predilection for the skin as well as peripheral nerves
- ▪
The primary skin lesion is erythematous or hypopigmented, and it is often anesthetic
- ▪
Based upon the clinicopathologic findings, which reflect the degree/type of immunity, leprosy is divided into two major forms – lepromatous with a predominantly Th2 response and tuberculoid with a predominantly Th1 response
- ▪
In lepromatous leprosy, multiple organisms are present in the dermis, whereas in tuberculoid leprosy, there are only a few organisms
Introduction
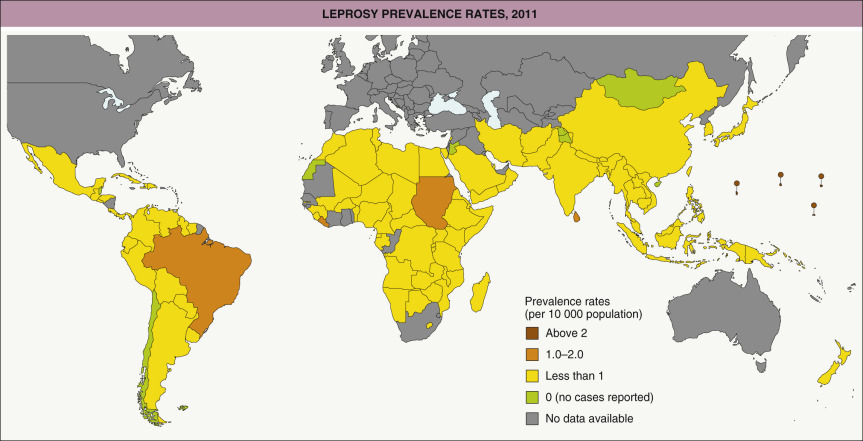
Leprosy is a chronic infectious disease with prominent involvement of the skin and nerves that is caused by the bacillus Mycobacterium leprae . It is divided into two major forms that depend upon the degree and type of immunity – lepromatous with a predominantly Th2 response and tuberculoid with a predominantly Th1 response. Leprosy is a serious health issue in a number of low-income countries. Although it seldom kills, leprosy represents a deforming, disabling, and stigmatizing disease . Once worldwide in distribution, leprosy is now seen primarily in tropical and subtropical regions of Asia, Africa, and Central and South America ( Fig. 75.1 ). The geographic distribution is probably related more to a lower standard of living and poorer hygiene than to a warmer climate . Early diagnosis and prompt therapy are key components in the strategy to control this chronic infectious disease .
History
The first written descriptions of leprosy may date back to as early as 600 BC in India, and to 200 BC in China and Japan. Many of the cases mentioned in the Bible may actually have been psoriasis, vitiligo, or other skin disorders rather than leprosy. The earliest scientific evidence of leprosy has come from an Egyptian skeleton and two Coptic mummies from the second century BC and the fifth century AD, respectively.
One theory was that leprosy was brought from India to the Mediterranean basin by Alexander the Great’s soldiers between 327 and 326 BC and then spread throughout the Greek and Roman Empires. Recent studies utilizing comparative genomics have shown that all extant cases of leprosy are attributable to a single clone, and the disease appears to have originated in the Near East or eastern Africa. During the Middle Ages, leprosy reached epidemic proportions in Europe and then spread to the New World via the wave of exploration in the late fifteenth and sixteenth centuries. Independent of geographic location, the afflicted have frequently been ostracized by their communities and families .
During the nineteenth century, Danielssen and Boeck provided the first modern description of leprosy, and in the 1870s, a third Norwegian, Gerhard Henrik Armauer Hansen, conducted landmark studies on the microbiology and epidemiology of M. leprae . Mitsuda created the skin test for leprosy in 1919 , and the value of sulfones in the treatment of leprosy was reported in 1942 . The Brazilian dermatologist Rabello advanced the notion of polar forms of leprosy in 1938, with a formal presentation in 1953 at the International Congress of Leprosy. In 1966, Ridley and Jopling suggested a classification of leprosy based on the immunologic status of the patient (see below).
Epidemiology
In the early 1980s, 11–15 million people worldwide were estimated to have leprosy . Curative multidrug therapy has substantially reduced the number of affected individuals . The number of new cases per year decreased from >500 000 in 2003 to ~210 000 in 2015, with only ~175 000 patients requiring treatment remaining worldwide . The goal of the World Health Organization (WHO) is a prevalence rate of <1 case per 10 000 persons, which has been achieved in all but a few countries .
In the Americas, the highest incidence is seen in Brazil, especially in western Amazonia (see Fig. 75.1 ). Although men and women are equally affected, the lepromatous form of leprosy is seen twice as often in men as in women. Leprosy affects all races and ages; however, its peak incidence occurs in individuals between 10–15 and 30–60 years of age .
Three requirements for the spread of leprosy are: a contagious patient, a susceptible person, and close or intimate contact. In endemic countries, the vast majority of new cases are in children and young adults who have close relatives with contagious forms of the disease. The risk of acquiring the disease from household contacts is 25%, emphasizing the public health importance of domicile control . The incubation period varies widely, from months to >30 years, but it is usually 4–10 years . M . leprae is spread predominantly via nasal and oral droplets from the bacilliferous patient and much less often from eroded skin. Even after 1–7 days, the bacillus is still viable in dried secretions. Inoculation is via the nasal mucosa or, less commonly, through breaks in the skin barrier. Transmission also depends on the infectivity of the contagious patient . An unusual mode of transmission, reported from India, was via reused tattoo needles.
Although leprosy primarily affects humans, it can be found or reproduced in nine-banded armadillos (southern US from Texas to Florida), red squirrels (British Isles), and mice . In one recent study, ~40% of leprosy patients from the southeastern US were infected with 1 of 2 M . leprae genotypes associated with wild armadillos, suggesting that leprosy represents a zoonosis in this region .
Pathogenesis
The bacillus M. leprae is a very small, slightly curved rod that is characteristically acid-fast. It is an obligate intracellular microorganism that has a predilection for macrophages and Schwann cells. The major sites of involvement are the peripheral nerves, skin, mucous membranes, bones, and viscera (e.g. testes, liver) . The bacillus requires a temperature of ~35°C to grow, and it thus prefers cooler regions of the body (e.g. nose, testicles, ear lobes) as well as regions where the peripheral nerves are close to the skin .
The fact that the majority of exposed individuals do not develop disease implies that there is variability in susceptibility/resistance, depending upon genetic and environmental factors. For example, susceptibility and the type of immune response appear to correlate with specific HLA types, such that individuals with HLA-DR2 and HLA-DR3 are more likely to develop the tuberculoid form and those with HLA-DQ1 the lepromatous form . Polymorphisms in several genes, including those that encode tumor necrosis factor (TNF), interleukin (IL)-6, 10 and 12, lymphotoxin-α (a low-producing allele associated with early-onset leprosy), the vitamin D receptor (VDR), and Toll-like receptor (TLR) 1 and 2, have also been shown to be related to either overall susceptibility or the particular form of leprosy that develops (although there is variability in different populations) . In Vietnamese and Brazilian populations, a leprosy susceptibility locus on chromosome 6q25–q26 contains 17 markers in a block of ~80 kilobases that overlaps the 5′ regulatory region shared by the Parkinson disease gene PARK2 (encoding Parkin, an E3 ubiquitin protein ligase) and the co-regulated gene PACRG . Genome-wide association studies in Chinese populations have linked variants in genes encoding proteins in the NOD2 (nucleotide-binding oligomerization domain containing 2) pathway with susceptibility to leprosy, especially the multibacillary form . The NOD2 pathway, along with the VDR, TLRs, and Parkin, play key roles in the innate immune response, as do several additional proteins encoded by leprosy-associated genes, e.g. MBL (mannose-binding lectin), MRC1 (mannose receptor C-type 1), NRAMP1 (natural resistance-associated macrophage protein 1), and KIR (killer cell immunoglobulin-like receptor) .
Depending upon the level of specific cell-mediated immunity, as reflected in the lepromin test (see below), the disease can progress, limit itself, or resolve spontaneously. Humoral immunity is increased in forms that are associated with minimal cell-mediated immunity, i.e. the lepromatous end of the spectrum ( Fig. 75.2 ) . For example, serum levels of antibodies against phenolic glycolipid-1 (PGL-1), an M. leprae -specific antigen, are highest in patients with lepromatous leprosy .
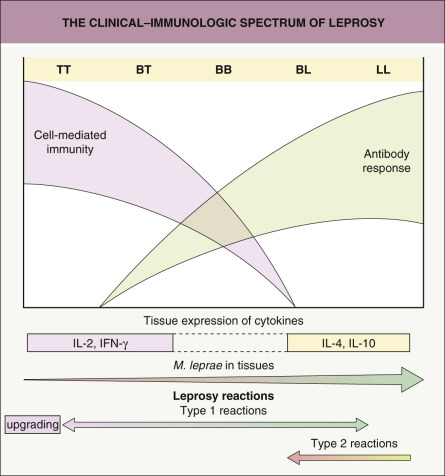
Macrophages play an important role in the body’s attempt to eliminate M. leprae , producing IL-1, TNF, and IL-12 when they encounter this organism. These cytokines then stimulate the number and activity of other macrophages. Studies in humans with tuberculoid leprosy have shown that a predominantly Th1 CD4 + T-cell response which produces IL-2, interferon (IFN)-γ, and lymphotoxin-α maintains inflammation. In patients with lepromatous leprosy, a predominantly Th2 response leads to the release of different cytokines – IL-4, IL-5, IL-10 and IL-13; these cytokines suppress macrophage activity . The cell wall of M. leprae contains lipid complexes, including PGL-1, which may suppress T-cell responses and IFN-γ production as well as the organism’s invasion of Schwann cells .
CD4 + T cells that express the antimicrobial protein granulysin have been detected at a sixfold greater frequency in tuberculoid as compared to lepromatous skin lesions . Tuberculoid lesions also have stronger expression of TLR2 and TLR1, activation of which can induce differentiation of macrophages and dendritic cells. CD1b + dendritic cells, which promote T-cell activation and secretion of proinflammatory cytokines, were not detected in lesions from patients with progressive lepromatous leprosy ; instead, these lesions had upregulation of genes belonging to the leukocyte immunoglobulin-like receptor (LIR) family. LIR-7 has been shown to suppress innate host defense by several mechanisms, including blocking antimicrobial activity triggered by TLRs .
Clinical Features
There is a wide spectrum of clinical findings in leprosy and Rabello divided it into four types: (1) lepromatous, associated with depressed cell-mediated immunity; (2) tuberculoid, associated with intact cell-mediated immunity; (3) dimorphous; and (4) indeterminate. The dimorphous form is “unstable” and capable of evolving into lepromatous or tuberculoid forms, depending upon whether there is improvement or worsening of cell-mediated immunity (see Fig. 75.2 ). The Ridley and Jopling classification is also based on polar forms, with lepromatous (LL) at the immunodepressed end, tuberculoid (TT) at the immunocompetent end, and three types of borderline leprosy in between: borderline lepromatous (BL), borderline-borderline (BB, in the middle), and borderline tuberculoid (BT) .
In 1997, the WHO created an operational system to facilitate the classification and therapy of leprosy in endemic areas with no access to laboratory facilities. This scheme divides leprosy into three groups: (1) paucibacillary, single-lesion leprosy (one skin lesion); (2) paucibacillary leprosy (two to five skin lesions); and (3) multibacillary leprosy (more than five skin lesions). This simplified classification is based solely on the number of cutaneous lesions, independent of their size, location or histologic features. However, on a global basis, the classification scheme outlined in Table 75.2 is still the one most commonly employed .
| CLASSIFICATION OF LEPROSY | ||||||
|---|---|---|---|---|---|---|
| Clinical findings | LL | BL | BB | BT | TT | I |
| Type of lesions | Macules, papules, nodules, diffuse infiltration | Macules, papules, plaques, infiltration | Plaques and dome-shaped, punched-out lesions | Infiltrated plaques | Infiltrated plaques, often hypopigmented | Macules, often hypopigmented |
| Number of lesions | Numerous | Many | Many | Single, usually with satellite lesions, to more than 5 | One or few (up to 5) | One or few |
| Distribution | Symmetric | Tendency to symmetry | Evident asymmetry | Asymmetric | Localized, asymmetric | Variable |
| Definition | Vague, difficult to distinguish normal versus affected skin | Less well-defined borders | Less well-defined borders | Well-defined, sharp borders | Well-defined, sharp borders | Not always defined |
| Sensation | Not affected | Diminished | Diminished | Absent | Absent | Impaired |
| Bacilli in skin lesions | Many (globi) | Many | Many | Few (1+), if any, detected | None detected | Usually none detected |
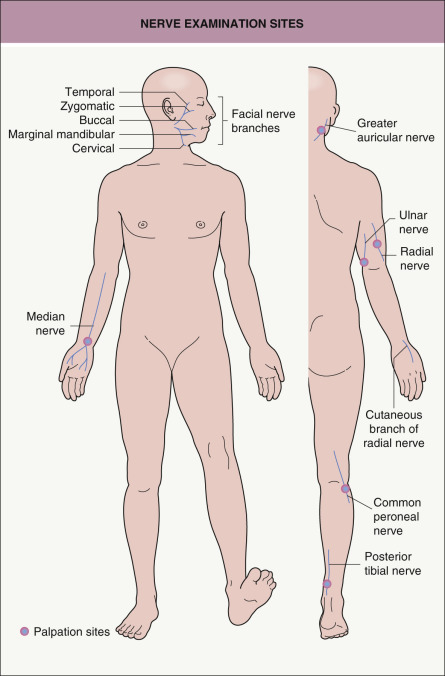
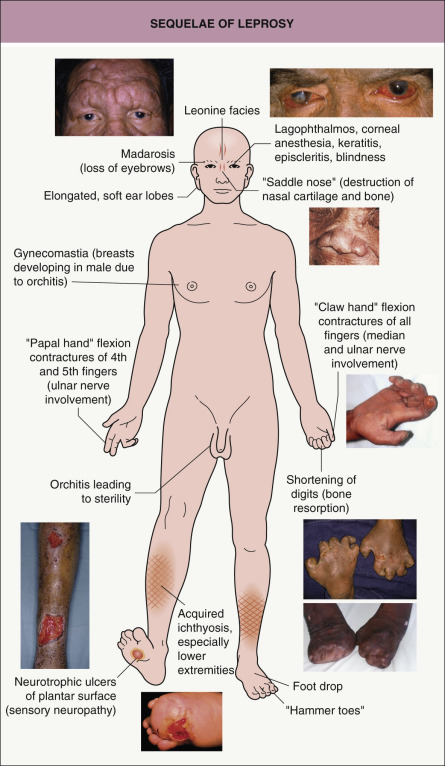
The clinical manifestations of leprosy involve primarily the skin and the nervous system (see Table 75.2 ). In addition to anesthesia or hypesthesia of individual cutaneous lesions, peripheral nerves can become enlarged and palpable. Certain peripheral nerves are more commonly affected, based primarily upon their more superficial location. Palpation of these nerves is an integral component of the physical examination of a patient with leprosy ( Fig. 75.3 ). The clinician should also determine via a complete neurologic examination whether there is a decrease in sensation to pain, temperature, and/or touch. This is in addition to inspection for neuropathic changes (e.g. muscle atrophy, flexion contractures of the fourth and fifth fingers), vasomotor alterations, and secretory disturbances (e.g. dry eyes and nasal mucosa) .
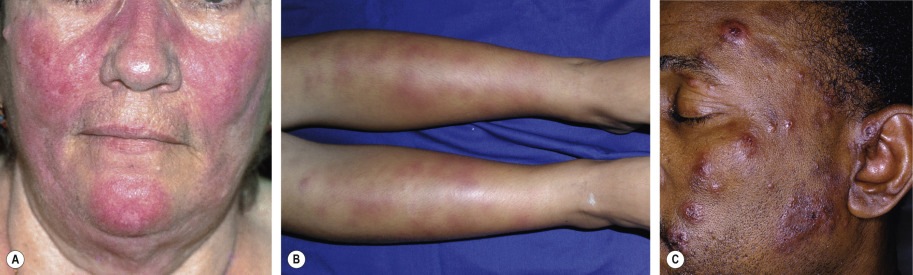
Lepromatous leprosy , the form with the least cellular immunity and greatest number of bacilli, is characterized initially by multiple, poorly defined, erythematous macules, papules, nodules, and plaques ( Fig. 75.4A ). Lesions are widespread and usually symmetric in distribution. The most common sites of involvement are the face, buttocks, and lower extremities. Infiltration of the skin of the forehead can lead to leonine facies ( Fig. 75.4B ). Additional signs and late sequelae include madarosis, saddle nose, infiltration of both earlobes, and acquired ichthyosis on the lower extremities ( Fig. 75.5 ). Anesthesia in a stocking or glove distribution may develop, often together with enlarged peripheral nerves and neuropathic changes. Ocular manifestations such as lagophthalmos (inability to completely close the eyes) and corneal and conjunctival anesthesia due to involvement of branches of the facial and trigeminal nerves, respectively, are sometimes present in severe cases . Histoid leprosy is a clinically distinct form of multibacillary disease that some authors classify as a variant of lepromatous leprosy. It is characterized by the development of dermatofibroma-like papules and nodules.
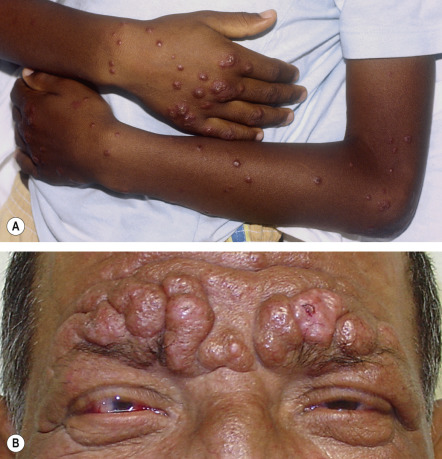
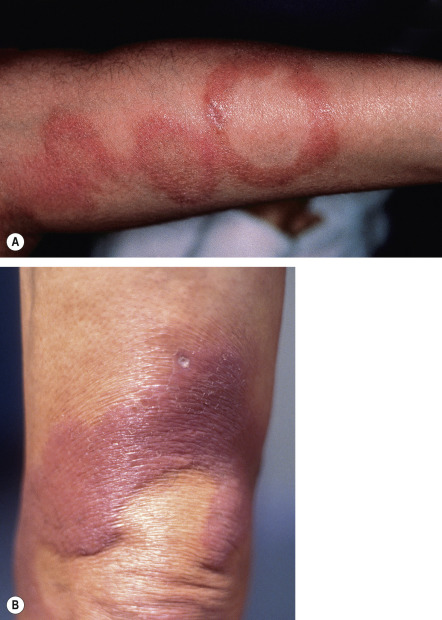
Borderline leprosy , as its name implies, has features that are intermediate between the two ends of the spectrum. Cutaneous lesions are usually asymmetric ( Fig. 75.6 ), e.g. there may be unilateral earlobe swelling. The severity of the cutaneous and peripheral nervous system abnormalities depends upon whether the patient is “leaning” towards the lepromatous pole (BL) or the tuberculoid pole (BT; see Table 75.2 ). Within the cutaneous plaques, hair is usually absent.
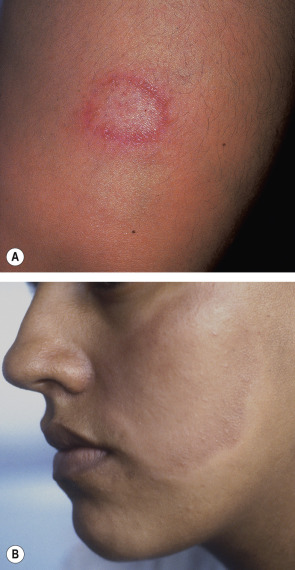
In tuberculoid leprosy , only a few well-demarcated plaques are seen and sometimes only neural involvement is present. The borders of the skin lesions are often slightly elevated, and this represents the preferred site for histologic examination. In some patients, the papules or plaques are erythematous, while in others, especially in darkly pigmented individuals, they are hypopigmented ( Fig. 75.7 ). There is typically partial, but not complete, pigment loss. The plaques should be examined for alopecia as well as anesthesia or hypesthesia. Unilateral neuropathic changes, especially of the extremities (e.g. digital resorption), may be seen .
One of the major problems faced by clinicians and patients, especially during treatment, is the development of reactions (see Fig. 75.2 ). These are characterized by acute inflammation that appears suddenly ( Fig. 75.8 ). Type 1 reactions can affect patients with any form of leprosy (except for the early indeterminate form), but there is a predilection for those in borderline categories. Type 2 reactions most often occur in patients with lepromatous or borderline lepromatous leprosy. In addition to the administration of antimicrobial drugs, the most frequent causes of reactions are pregnancy, other infections, and mental distress. The immunopathogenesis, clinical manifestations, and treatment of the two major types of inflammatory reactions are outlined in Table 75.3 . Type 1 (reversal) reactions are due to a change in the immunologic state of the patient and are often associated with neuritis. When there is an increase in cell-mediated immunity, this is referred to as an “upgrading” reaction.
| TWO MAJOR TYPES OF LEPROSY REACTIONAL STATES | ||
|---|---|---|
| Type 1 – reversal reaction with “upgrading” | Type 2 – vasculitis (most commonly erythema nodosum leprosum) | |
| Immune mechanism | Enhancement of cell-mediated immunity with a Th1 cytokine pattern | Excessive humoral immunity with a Th2 cytokine pattern and formation of immune complexes; may be accompanied by increased cell-mediated immunity |
| Pathogenic process | Delayed-type hypersensitivity reaction | Cutaneous and systemic small vessel vasculitis |
| Leprosy category | Borderline (BT, BB, BL) or tuberculoid with immunologic recovery during or after treatment | Lepromatous and BL > BB; especially patients with a high bacterial index who are undergoing treatment |
| Clinical characteristics |
|
|
| Treatment | Prednisone | Thalidomide |
Type 2 reactions are due to the formation of immune complexes in association with an excessive humoral reaction. They typically occur when patients with lepromatous forms of leprosy undergo treatment. A type 2 reaction represents a cutaneous and systemic small vessel vasculitis, and the most common clinical manifestation is erythema nodosum leprosum (see Fig. 75.8B,C ) . Patients with the diffuse form of lepromatous leprosy, who are usually from Central or South America, may develop the Lucio phenomenon , a reactional state characterized by thrombotic phenomena in addition to necrotizing cutaneous small vessel vasculitis. On a worldwide basis, leprosy is a common cause of cutaneous vasculitis, especially in low-income countries .
Pathology and Laboratory Evaluation
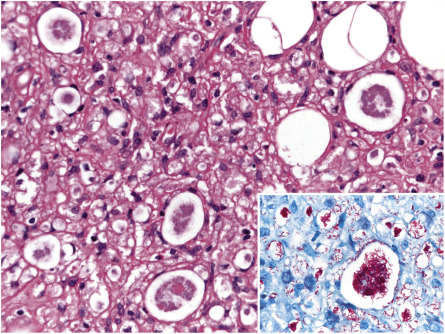
There are three basic histopathologic patterns observed in leprosy: lepromatous, tuberculoid, and borderline. In the lepromatous pattern, an infiltrate is seen in the dermis, subcutis, lymph nodes, abdominal organs (e.g. kidney, liver), testicles, and bone marrow. The infiltrate contains Virchow cells, which are macrophages with numerous bacilli as well as lipid droplets in their cytoplasm. In H&E-stained sections, these cells have a foamy appearance. The bacilli in leprosy can be detected by a Gram, Ziehl–Neelsen, or Fite (the most commonly used) stain, all of which stain the bacilli a bright red color. Methenamine silver stains are also useful for detecting fragmented acid-fast bacilli. Sudan III and Sudan IV (also known as scarlet red) were used historically and stain the bacilli black and red, respectively. For lesions in which bacilli are usually scant, it is recommended that at least six sections be examined before declaring them negative .
A band of normal-appearing dermis (Unna band or Grenz zone) separates the epidermis from the infiltrate, which is composed of plasma cells and lymphocytes in addition to Virchow cells ( Fig. 75.9 ). Both isolated bacilli and globi (clumps of bacilli) are seen in the dermis; when the patient is undergoing successful therapy, the organisms fragment and become granular. Cutaneous nerves demonstrate lamination of the perineurium, producing an onion-skin appearance. In the histoid variant, a well-circumscribed proliferation of spindle cells contains numerous bacilli, which typically line up along the long axis of the cell.
In the tuberculoid pattern of leprosy, a dermal granulomatous infiltrate is seen and may have a linear pattern following the course of a nerve ( Fig. 75.10 ). Epithelioid cells and Langhans giant cells are surrounded by lymphocytes. The cutaneous nerves are edematous, and there is an absence of organisms, even with special stains. Inflammation and fragmentation of nerve fibers in tuberculous leprosy differentiate it from sarcoidosis and other granulomatous disorders.
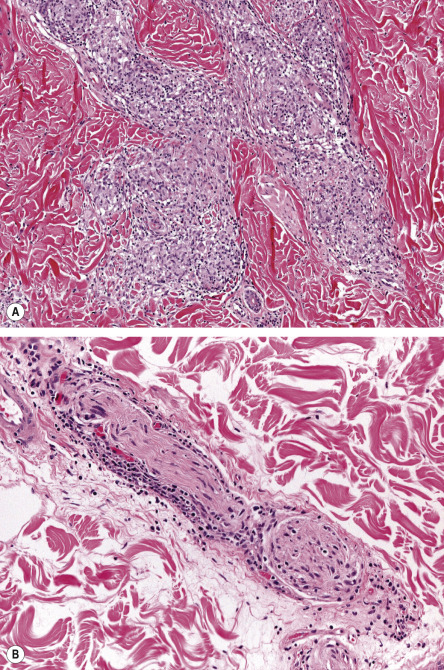
The borderline pattern contains histologic features of both the lepromatous form (e.g. Virchow cells) and tuberculoid form (e.g. granulomas). Predominance of the former versus the latter is dependent upon whether the patient has the BL, BB, or BT form of borderline leprosy. There are three different histologic “scenarios” in borderline leprosy: (1) some lesions have a lepromatous pattern while others have a tuberculoid pattern; (2) lepromatous and tuberculoid patterns are present in the same lesion; and (3) there is a mixture of foamy and epithelioid cells.
Indeterminate leprosy is usually difficult to diagnose histologically. Only a patchy infiltrate of lymphocytes or histiocytes around blood vessels or appendages is seen . There are no granulomas or Virchow cells , and organisms are usually absent.
When leprosy is suspected, the diagnosis of several forms (e.g. LL, BL) can be confirmed by finding bacilli in cutaneous scrapings, lymph nodes, or nasal secretions. Samples for bacilloscopy may be obtained from the earlobes, forehead, chin, extensor forearms, and dorsal fingers, as well as the buttocks and trunk. To avoid bleeding, a fold of skin is firmly squeezed between the finger and thumb of the examiner or with forceps, and a small incision is made with a scalpel blade. The liquid obtained is smeared onto a slide and allowed to dry. The smear is usually stained by the Fite (or Ziehl–Neelsen) method and a search is made for red rods (against a blue background) at 100× with oil immersion . Organisms are found in 100% of patients with lepromatous leprosy, 75% of those with borderline leprosy, and only 5% of those with tuberculoid leprosy. A biopsy specimen of the skin lesions should be obtained, especially in patients with suspected tuberculoid leprosy (see above).
By PCR, a number of genes encoding antigenic proteins (e.g. 36 kDa proline-rich antigen, Ag85B) can be amplified, as can M . leprae -specific repetitive repeat sequences (RLEP region). This molecular technique is of particular help in paucibacillary leprosy and can be performed on slit-skin smears and fresh, frozen , or paraffin-embedded skin biopsy specimens. In a study utilizing RLEP real-time PCR, M . leprae DNA was detected in 38 (75%) of 51 paraffin-embedded skin biopsy specimens from patients with paucibacillary leprosy . In addition, immunohistochemical staining of biopsy specimens for the PGL-1 antigen may prove helpful in paucibacillary disease.
Serologic assays for anti-PGL antibodies are only sensitive for the diagnosis of leprosy in the setting of multibacillary disease. However, measurement of these antibodies can help to classify patients, monitor the response to treatment, and predict leprosy reactions . Serum levels of anti-PGL-1 IgG and IgM antibodies are highest in those with lepromatous disease and lowest (or absent) in those with BT or tuberculous disease, therefore representing a marker of mycobacterial “load”. Elevation of anti-PGL-1 IgM antibody levels is also associated with reactions and impairment of nerve function .
Nowadays, three classic skin tests for leprosy – histamine, pilocarpine, and lepromin (Mitsuda) – are performed infrequently. In the first, a drop of 0.001% histamine is placed on the suspect patch and another one on normal skin. Both sites are then pricked with a needle and, after 10 minutes, the intensity and extent of the wheal and flare are recorded. This response is dependent upon the integrity of sympathetic nerves fibers; in tuberculoid lesions of leprosy, it will be decreased, delayed, or absent .
In the pilocarpine test, tincture of iodine is applied to the suspect lesion and normal skin prior to injection of pilocarpine. These areas are then dusted with a starch powder that will turn blue if there is normal sweating (see Ch. 39 ). Because sweating depends on the integrity of parasympathetic nerve fibers, it is decreased in lesions of leprosy. Quinizarin, which turns from white to blue with sweating, can be used instead of iodine and starch .
The lepromin (Mitsuda) test consists of an intradermal injection of 0.1 ml of a suspension of heat-killed M. leprae . The response is positive when a nodule forms at the site of injection 3 to 4 weeks later, indicating that the patient can mount a specific cell-mediated response to the bacilli. The test provides prognostic, but not diagnostic, information. It is positive in TT and BT leprosy .
Lastly, leprosy, especially LL and BL, is one of the causes of biologically false-positive Venereal Disease Research Laboratory (VDRL) and fluorescent treponemal antibody absorption (FTA-ABS) assays for syphilis (see Ch. 82 ).
Differential Diagnosis
Skin diseases that may be confused with the different cutaneous presentations of leprosy are outlined in Table 75.4 .
| CLINICAL DIFFERENTIAL DIAGNOSIS OF LEPROSY | |
|---|---|
| Hypopigmented lesions |
|
| Circinate (annular) plaques |
|
| Infiltrated plaques/nodules |
|
| Neurologic findings |
|
| Deforming acral features |
|
| Type 1 reaction |
|
| Type 2 reaction |
|
* Lesions favor the shins and are longer-lived than erythema nodosum leprosum lesions, which are widespread, appear in crops and then resolve relatively rapidly.
Treatment
The WHO recommends multidrug regimens that are safe, effective, and easily administered ( Table 75.5 ). When bacilloscopy is available and no bacilli are found, patients are considered to have paucibacillary leprosy; they are treated with rifampin (rifampicin) monthly plus a sulfone (usually dapsone) daily for 6 months, and then observed for 2 years. For patients with paucibacillary disease and a single lesion, single-dose therapy with r ifampin, o floxacin, and m inocycline (“ROM scheme”) has produced good results. If at least one bacillus is detected via bacilloscopy, the patient should receive rifampin, clofazimine, and dapsone for 1 to 2 years and then be observed for 5 years. A second option is to combine a sulfone with clofazimine .
| MULTIDRUG THERAPY/WHO SCHEME FOR THE TREATMENT OF LEPROSY | ||||||
|---|---|---|---|---|---|---|
| Rifampin | Clofazimine | Dapsone | Ofloxacin | Minocycline | Therapy duration | |
| MB (>5 lesions * ) | 600 mg once monthly | 300 mg once monthly and 50 mg daily | 100 mg daily | − | − | 12 blister packs over 12 to 18 months |
| PB (2–5 lesions * ) | 600 mg once monthly | − | 100 mg daily | − | − | 6 blister packs over 6 to 9 months |
| PB (single lesion * ) | 600 mg × 1 | − | − | 400 mg × 1 | 100 mg × 1 | Single dose |
| Dose adjustments for children | ||||||
| 10 to 14 years of age, MB † | 450 mg once monthly | 150 mg once monthly and 50 mg every other day | 50 mg daily | − | − | 12 blister packs over 12 to 18 months |
| <10 years of age, MB † | 300 mg once monthly | 100 mg once monthly and 50 mg twice weekly | 25 mg daily | − | − | 12 blister packs over 12 to 18 months |
| 5 to 14 years of age, single lesion PB | 300 mg × 1 | − | − | 200 mg × 1 | 50 mg × 1 | Single dose |
* WHO classification for endemic areas when no facilities for performing bacilloscopy are available.
† For PB disease (2–5 lesions), clofazimine is not required and the duration of therapy is 6 to 9 months.
Multidrug therapy is extremely effective. Importantly, after the first dose, the patient is no longer infectious to others. All patients who complete the prescribed regimen are considered cured, as there are virtually no relapses. Bacilli may be found but they are non-viable. Drugs that may play a significant role in the treatment of leprosy in the future include other quinolones (e.g. moxifloxacin), clarithromycin, and ansamycins . Rifapentine (a rifampin derivative) has higher peak serum concentrations, a longer serum half-life, and more bactericidal activity against M. leprae than does rifampin. In addition, moxifloxacin appears to be more bactericidal than ofloxacin. Thus, the combination of rifapentine, moxifloxacin, and minocycline (PMM) may be superior to rifampin, ofloxacin, and minocycline (ROM) .
For the two major inflammatory reactions, additional medications are often required. Oral prednisone (20–60 mg per day) is used for type 1 (reversal) reactions, while thalidomide (100–200 mg per day) is the principal therapy for type 2 reactions (erythema nodosum leprosum). Systemic corticosteroids are also recommended for the Lucio phenomenon. Although thalidomide remains the treatment of choice for type 2 reactions, teratogenicity is a major limitation and it is unavailable in many countries where leprosy is endemic. Lenalidomide and pomalidomide are thalidomide analogues with different side-effect profiles (e.g. more myelosuppression) that could have potential utility in the treatment of type 2 reactions. Additional drugs with possible benefit in type 2 reactions include cyclosporine, which has been used for corticosteroid-resistant type 1 reactions, as well as clofazimine, chloroquine, pentoxifylline, and phosphodiesterase type-4 inhibitors (e.g. roflumilast) .
Although leprosy is now considered a curable disease with a good prognosis and excellent survival rate, it can still be incapacitating and stigmatizing. It is very important to make the diagnosis as soon as possible and to examine contacts, since early treatment prevents disability. Recognition of leprosy in its initial phases by healthcare professionals as well as the general population in endemic countries is essential in order to reduce the impact of this disease .
Cutaneous Tuberculosis
- ▪
Infection of the skin due to Mycobacterium tuberculosis , an acid- and alcohol-fast bacillus
- ▪
A resurgence of tuberculosis (TB) in the late 1980s and early 1990s resulted from the HIV epidemic, emergence of resistant strains of M. tuberculosis , and a decline in TB control efforts
- ▪
Exogenous exposure produces primarily the tuberculous chancre and TB verrucosa cutis
- ▪
Endogenous infection can lead to scrofuloderma, miliary TB, and lupus vulgaris
- ▪
Tuberculids include a papulonecrotic eruption, lichen scrofulosorum, and erythema induratum
Introduction
Tuberculosis (TB) has affected humans since prehistoric times. There was, however, a worldwide explosion in its incidence in the late 1980s and early 1990s, and consequently a resurgence of cutaneous TB . This phenomenon has been attributed to the HIV epidemic, the emergence of resistant strains of Mycobacterium tuberculosis , increased use of immunosuppressive therapies, more migration of people around the world, and a decline in TB control efforts , superimposed on the pre-existing factors of poverty and malnutrition.
History
There are paleopathologic findings suggesting M. tuberculosis infections as early as 3700 BC in Egypt, and 2500–1500 BC in Europe. Utilizing PCR, M. tuberculosis DNA has been detected in mummified remains from ancient times. In 1826, Laennec first reported cutaneous TB in the form of a “prosector’s wart”. Rokitansky and Virchow subsequently described the histologic features of cutaneous TB in detail. The discovery of M. tuberculosis by Koch in 1882 and the advances in descriptive pathology during the nineteenth century helped establish cutaneous TB as part of the spectrum of this infectious disease .
Epidemiology
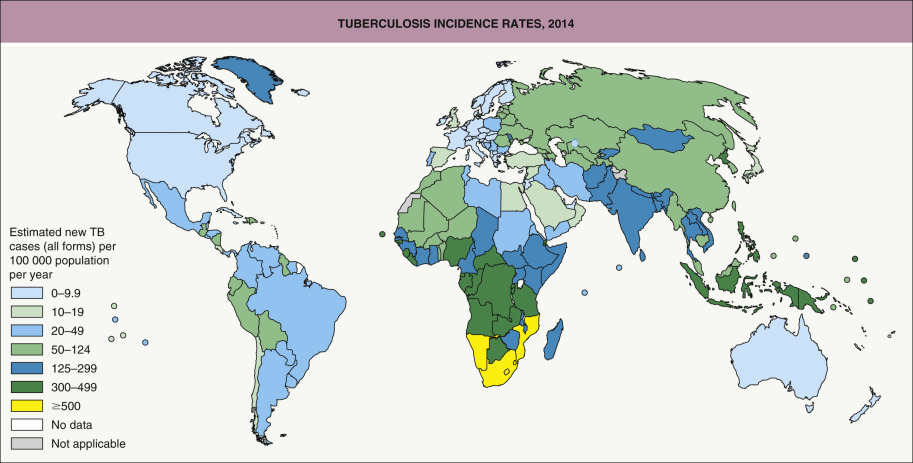
M. tuberculosis is not particularly virulent, as only 5% to 10% of infections lead to clinical disease. This bacterium has a worldwide distribution; it is more prevalent in regions with a cold and humid climate but can also occur in the tropics ( Fig. 75.11 ) . The incidence of cutaneous TB parallels that of pulmonary TB and is therefore higher in low-income countries and impoverished populations. HIV infection, immunosuppressive therapies (especially TNF inhibitors; see Ch. 128 ), and innate defects of the IL-12/IFN-γ axis (see Ch. 60 ) place individuals at greater risk for the development and reactivation of TB.
Pathogenesis
M. tuberculosis is the predominant etiologic agent of cutaneous TB. Occasionally, M. bovis and bacille Calmette–Guérin (BCG), an attenuated strain of M. bovis , may produce skin lesions .
M. tuberculosis is a slender, non-motile, aerobic, non-spore-forming, filamentous rod. It is an acid- and alcohol-fast bacillus that has a waxy coating with a high lipid content, which makes it resistant to degradation after phagocytosis .
M. tuberculosis disseminates primarily via inhalation of aerosolized droplets of saliva from individuals with active disease; transmission by ingestion or inoculation can also occur . Intact skin provides an effective protective barrier against invasion of the organism, but a break in the mucocutaneous barrier can facilitate entrance . Untreated infected individuals who do not have underlying medical problems have a 5% to 10% lifetime risk of progression to active TB; this risk increases substantially with immunosuppression, e.g. due to HIV infection or treatment with medications such as TNF inhibitors.
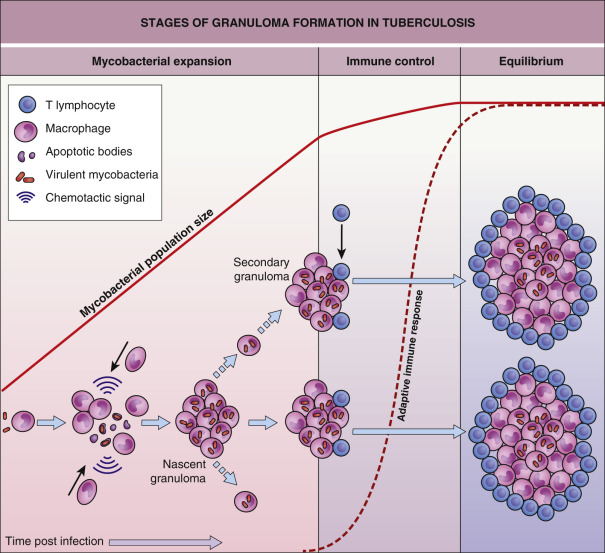
Studies using a model of zebrafish infected with Mycobacterium marinum have demonstrated that granulomas, which are classically considered as host-protective structures formed to control infection, actually contribute to early bacterial growth and facilitate spread of infection ( Fig. 75.12 ). Mycobacteria induce granuloma formation by releasing the early secreted antigenic target 6 (ESAT-6) protein, which stimulates neighboring epithelial cells to produce matrix metalloproteinase-9 (MMP-9). MMP-9 enhances recruitment of macrophages that contribute to bacterial proliferation and spread as well as maturation of the nascent granuloma . Increased production of other MMPs (e.g. MMP-1, MMP-3) has been shown to drive tissue destruction in TB . Eventually, development of adaptive immunity results in the curtailment, but not eradication, of bacterial growth by CD4 + and CD8 + effector T cells. The mature granuloma therefore represents an equilibrium between mycobacterial growth and the host immune response, and MMPs represent potential targets for antimycobacterial therapy.