Chapter 48 Minimal scar breast augmentation using autologous fat grafting
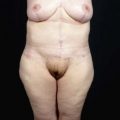
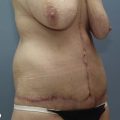
• Moderate augmentation (100–200 ml) of the breast is successfully achieved by autologous lipoinjection without major complications if appropriately performed in selected patients.
• Breasts augmented with fat injection are soft and show natural texture and appearance, and patients are free from concerns associated with foreign materials (and related complications and potential implant replacement or removal).
• The surviving fat volume varies substantially among patients, and multiple factors likely affect clinical results; patient factors include skin redundancy of the breast and availability of fat.
• Surgeons need to improve quality (such as tissue viability and progenitor/adipocyte ratio) of fat grafts and infiltration techniques.
• Aspirated fat tissue should be appropriately harvested and stored, and quickly processed and infiltrated to place aliquots or noodles of fat grafts as diffusely as possible.
• Supplementation of vascular stromal fraction containing adipose progenitor cells may boost the efficacy and safety of lipoinjection to the breasts.
Introduction
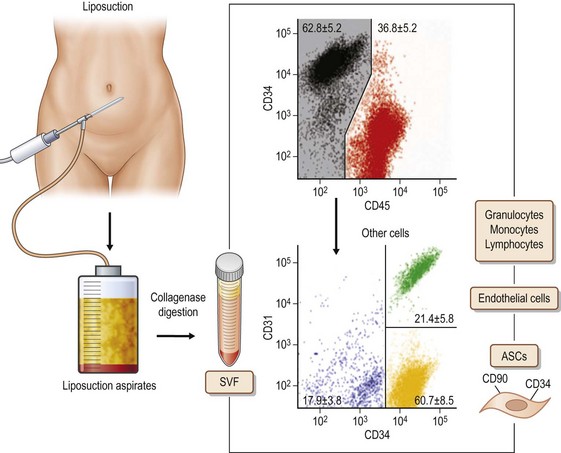
Adipose tissue has been considered an organ of energy storage, the largest endocrine organ, a soft tissue filler (augmentation by micro-fat grafting), and a cosmetically unnecessary tissue discarded by liposuction. Adipose tissue physiologically turns over very slowly. Adipocytes have a life span of 10 years;1 several thousands of adipocytes die and are replaced with new adipocytes every second in our body. Adipose tissue has many cells other than adipocytes, though adipocytes constitute more than 90% of adipose tissue volume. Through collagenase digestion, cellular components other than adipocytes are extracted as a cell pellet, which is called stromal vascular fraction (SVF). SVF contains adipose stromal cells (ASC), vascular endothelial cells (VEC), pericytes, adipose-resident macrophages, etc. (Fig. 48.1). It was found that the adipose stromal cell population contains not only monopotent progenitor cells responsible for the tissue turnover, but also multipotent mesenchymal stem cells,2 which are now called adipose (-derived) stem cells. These are regarded as a potent tool for cell-based therapies, comparable to bone marrow-derived mesenchymal stem cells, because they can be obtained in a large amount through a less invasive approach. For adipose tissue engineering/regeneration, it is a critical point to understand the physiological roles and functions of ASC; this population, working together with VEC and stem/progenitor cells recruited from bone marrow, is a main player for adipose tissue engineering, which means adipogenesis and angiogenesis.
Autologous fat transplantation is one of the promising cosmetic treatments for facial rejuvenation and soft-tissue augmentation due to minimal incisional scars and lack of complications associated with foreign materials. However, certain problems remain, such as unpredictability and a low rate of graft survival due to partial necrosis. Implantation of prostheses has been a golden standard for breast augmentation, but complications such as capsular contracture remain to be resolved. Recently, autologous fat injection has been re-evaluated as a potential alternative to artificial implants for breast augmentation.3 This re-evaluation may reflect recent advances in autologous fat transfer and the radiological detection of breast cancer.
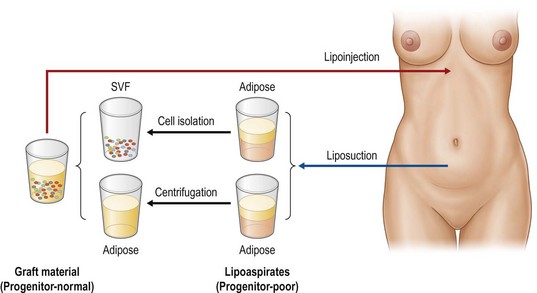
In this chapter, biological principles and surgical techniques of fat injection for breast augmentation are discussed as well as our novel approach of autologous fat grafting called cell-assisted lipotransfer (CAL),4,5 which is concurrent transplantation of aspirated fat tissue and adipose progenitor cells (Fig. 48.2).
Preoperative Preparation
There seem to be several patient factors that may affect the clinical result of lipoinjection; skin redundancy of the breasts, age, BMI, personal quality or character of fat tissue, adhesive scars, breast implant and its capsule, systemic disease such as autoimmune disease, oral corticosteroid, etc.4,6 Good candidates are those who have sufficient fat at donor sites and the redundant breast skin with healthy vascularity and without scars. Lipoinjection can be performed in any patients from teens to 70s, but patients with low BMI (less than 18) are not good candidates due to difficulty in harvesting a sufficient volume of fat tissue without severe morbidity. Patients who want a large-volume (250–400 ml) augmentation, are not good candidates because augmentation volume achieved by a single session of lipoinjection is limited (100–200 ml).
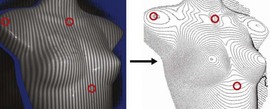
For preoperative examination, physical measurements (maximum and bottom breast circumferences, etc.), mammogram, MRI, echogram, photograph, and videograph are performed. We also adopted a 3-dimensional measurement system that enables a volumetric evaluation of the breast mound in a standing position (Fig. 48.3). An echogram is easy to perform at every patient’s visit and sensitive enough to detect small cyst formation. Long follow up with an annual mammogram up to 5 years is recommended to detect abnormal signs such as calcifications.
Surgical Technique
Breast Augmentation
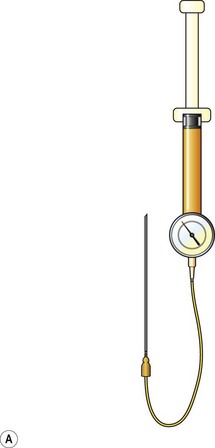
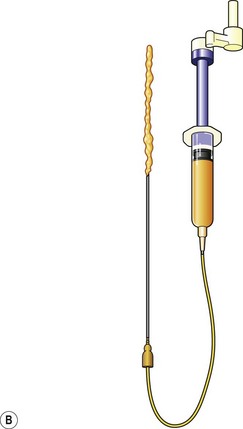
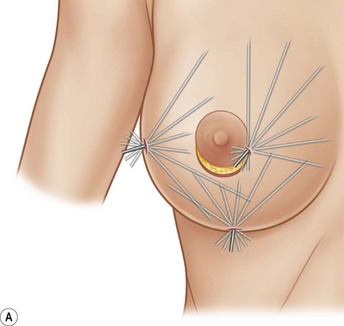
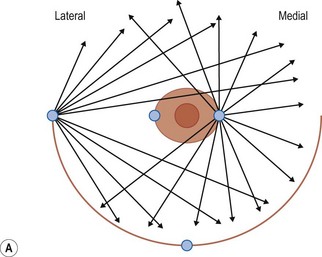
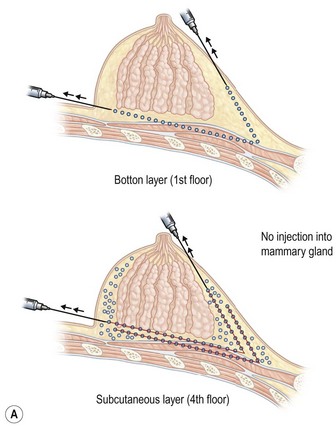
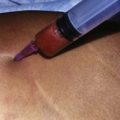
Donor sites are usually the thighs alone or the thighs and the abdomen or flanks, decided according to the patient’s preference and BMI. After the liposuction site is infiltrated with saline solution with epinephrine (0.001%) under general anesthesia, adipose tissue is suctioned using a cannula with 2–3 mm inner diameter and a conventional liposuction machine. We use screw-type syringes (with a threaded plunger) to allow for precise control and avoid a flushing injection (Fig. 48.4). To reduce the time of the procedure, two syringes are used; while one syringe is being used for an injection, the other is filled with the graft material in preparation for the next injection. A 16- or 18-gauge needle (150 mm long) is used for lipoinjection and inserted subcutaneously from the inframammary fold or areolar margin (Fig. 48.5A). The operator takes care to insert and place the needle horizontally (parallel to the body), in order to avoid causing a pneumothorax. The needle is inserted in several layers and directions, and is continuously and gradually retracted as the plunger is advanced (Fig. 48.5B). This technique is used to obtain a diffuse distribution of the graft material. The grafts are placed into the fatty layers on, around, and under the mammary glands (but not into the mammary glands), and also into the pectoralis muscles. After training, it is not hard for an operator to recognize the mammary gland or pectoralis fascia as a harder tissue than the fat or muscle tissue. Injection is discontinued when the skin tension becomes tense; injection volume is usually 200–300 ml for each breast in Asian patients.
Breast Implant Replacement
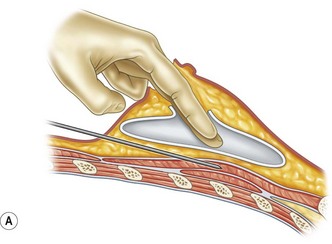
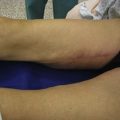
For patients with implants, lipoinjection can be performed simultaneously with implant removal.7 Breast implants are removed through a periareolar incision, which is placed at a caudal third of the areolar margin. The lipoinjection is begun at the deepest layer (under the implant capsule when the implant was subglandular) and completed with the injection into the most superficial subcutaneous layer. During the injection, the operator can insert fingers into the implant capsule and recognize the location of the injection needle (Fig. 48.6). Injection into the mammary glands or into the capsular cavity is not performed. Finally, the capsular cavity is irrigated with saline to remove any fat tissue injected and the periareolar incision is closed.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree