Management of the Microphthalmic or Anophthalmic Orbit
Sarah M. Jacobs
Christopher B. Chambers
DEFINITION
A spectrum exists ranging from small eyes to complete absence of any ocular tissue:
Nanophthalmos is defined as a small eye with otherwise normal internal structures. The axial length of the eye is ≥2 standard deviations shorter than average.
Microphthalmos is an eye with axial length less than 20 mm and additional structural abnormalities.1
Clinical anophthalmos describes some cases of microphthalmia in which the eye is so small that the patient appears anophthalmic. However, histopathologic examination of the tissue does identify products of optic vesicle formation.
ANATOMY
Nanophthalmos: In addition to short axial length, the sclera is thickened relative to the eye, the anterior chamber angles are narrow, and the refractive error is highly hyperopic. The small eye may or may not result in a hypoplastic orbit.3,4
Microphthalmos: In addition to short axial length, the internal structures of the eye are abnormal. Microphthalmic eyes can be associated with coloboma of the uvea and/or lens, persistent fetal vasculature, Peters anomaly, glaucoma, and anterior polar cataracts. An uncommon variant, microphthalmia with cyst results from failure of the ocular embryonic fissure to close, leading to a small rudimentary eye joined to a larger cystic cavity along the colobomatous inferonasal sclera.5 The orbit on the affected side is hypoplastic.
Anophthalmos: There is complete absence of any ocular tissue. The eyelids have a small palpebral fissure, and beneath the lids a rudimentary conjunctival membrane is present covering the hypoplastic orbit.
PATHOGENESIS
Embryologic failure of the optic vesicle to develop normally from the cerebral vesicle during the 4th to 8th week of gestation can lead to congenital absence or malformation of one or both eyes.
Because the globe influences orbital growth, these ocular anomalies can result in orbital and facial maldevelopment as well.
Nanophthalmos is typically sporadic, but autosomal recessive or autosomal dominant forms have been described (NNO1 on chromosome 11p and NNO3 on chromosome 2q). It can occur as an isolated disorder, or as part of a syndrome such as oculo-digital-dental syndrome (ODD), or autosomal dominant vitreoretinochoroidopathy with nanophthalmos (ADVIRC).6
Microphthalmos occurs in approximately 10 per 100 000 live births. It can occur sporadically, with some suspected association to fetal alcohol syndrome or maternal infections during gestation, especially rubella, cytomegalovirus, and herpes simplex virus. It is estimated that over half of cases occur in conjunction with systemic abnormalities, including those occurring in the setting of chromosomal anomalies, including Trisomy 13 (Patau syndrome), Trisomy 18 (Edwards syndrome), or Wolf-Hirschhorn syndrome.7 In inherited cases, mutations of the SOX2 gene on chromosome 3q are frequently encountered.7 This condition can be associated with CHARGE syndrome (Coloboma, Heart defect, Atresia choanae, Retarded growth, Genital abnormality, and Ear abnormality), and PHACE syndrome (Posterior fossa anomalies, Hemangioma, Arterial lesions, Cardiac abnormalities, and Eye anomalies).
Anophthalmos occurs approximately 2 per 100 000 live births. It is most often sporadic but can be associated with autosomal dominant defects of the SOX2 or RBP4 genes. Maternal deficiency of vitamin A during pregnancy can also pose a risk of anophthalmia.
NATURAL HISTORY
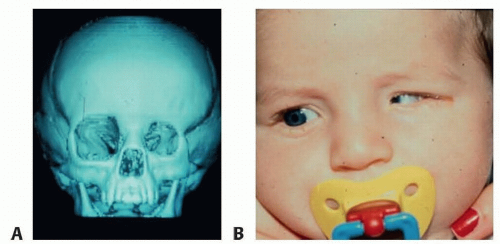
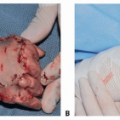
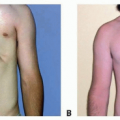
Ocular volume drives the growth of the eyelids, fornices, and bony orbit. In turn, hemifacial development parallels orbital development. Thus, a small or absent eye leads to a hypoplastic orbit that can result in deformity of the hemifacial skeleton (FIG 1).
At birth, the normal newborn eye is approximately 70% of its adult size. In contrast, the newborn face is only 40% of its adult size. Rapid growth of the facial skeleton and soft tissues occurs during early childhood, with the face reaching 70% adult size by 2 years and 90% adult size by 5.5 years.8 The degree of orbital and facial maldevelopment is different for each microphthalmic child, depending on the amount of ocular tissue present and the trophic influence that tissue exerts.
PATIENT HISTORY AND PHYSICAL EXAMINATION
As part of the initial assessment, the following questions should be considered as they will guide further management throughout childhood:
Is the eye present or absent?
Is this a seeing eye?
Is the other eye present? If so, is it normal?
Are the orbits symmetric and appropriate in size?
Are the periocular and facial soft tissues symmetric and appropriate in size?
A coordinated multidisciplinary approach is needed for evaluation of the microphthalmic or anophthalmic child, as approximately 50% have associated systemic abnormalities and 25% have defined systemic syndromes. This approach should include:
Comprehensive assessment by a pediatrician is needed, with particular attention to the face, ears, and palate; feeding difficulties, which can indicate neurologic or esophageal abnormalities; the cardiac system and genital anatomy (due to association with CHARGE); and metabolic disturbances, which may indicate pituitary dysfunction due to midline brain abnormalities.
Referral to subspecialists such as neurologist, cardiologist, endocrinologist, nephrologist, and audiologist should be arranged if indicated.
In cases with systemic findings or a syndromic diagnosis, evaluation and counseling by a pediatric geneticist is also recommended.
Ophthalmologic exam is recommended as soon as possible within the first 2 to 3 weeks of life. This should involve:
Complete anterior and posterior assessment of both eyes to assess orbital and ocular anatomy as well as visual function. In unilateral cases, the fellow eye may have subtle findings such as coloboma, cataract, retinal dystrophy, or optic nerve hypoplasia.9
Full cycloplegic refraction should be prescribed to maximize visual acuity of seeing eyes. Eyeglasses with polycarbonate lenses should be used in order to protect the globes from trauma.
IMAGING AND OTHER DIAGNOSTIC STUDIES
Preferential gaze testing (eg, Teller cards) or pattern visual evoked potential (pattern VEP) can assess basic visual acuity in preverbal patients.
Flash visual evoked potential (VEP) testing can evaluate for any visual-cortical function in patients with severe microphthalmia or apparent anophthalmia.9
Facial photographs should be taken, including a ruler in the photo for scale.
Imaging with ultrasound of the eye and orbit to evaluate its structure and detect ocular remnants or cysts.
MRI for detailed assessment of the soft tissues of the orbit and brain
Maxillofacial CT for assessment of the orbital and facial bones
NONOPERATIVE MANAGEMENT
Management focuses on utilizing the remaining vision in one or both eyes, promoting appropriate growth of the orbit and face, and optimizing ocular appearance.
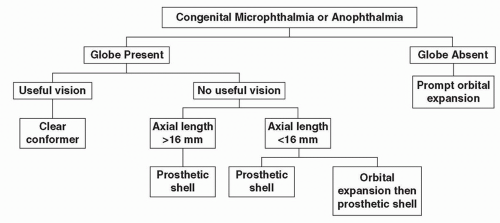
FIG 2 outlines a treatment algorithm to accomplish these goals according to varying degrees of severity. Parents should be counseled that a series of procedures and/or devices will be needed over time to help the orbit expand in parallel to the growth of the rest of the face.
Certain cases can be managed nonoperatively with placement of progressively larger conformers:
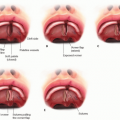
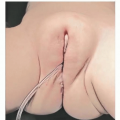
Children with useful vision in the affected eye(s) should be fitted with a series of clear conformers to progressively expand the lids, fornices, and orbit while still allowing the eye to see through the device so that visual development can occur (FIG 3).
Blind eyes measuring greater than 16 mm in length can be fitted with a series of ocular prostheses to maintain the socket while also improving the appearance of the eye.
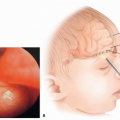
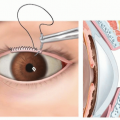
If an eye is unable to tolerate wearing a prosthetic shell due to mechanical irritation of the corneal surface, a graft can be placed to cover the cornea (FIG 4).
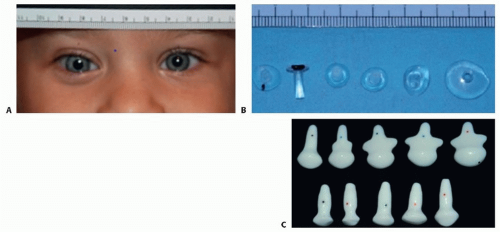
FIG 3 • A-C. A series of progressively larger conformers can be made to maintain the socket, exerting pressure to help the orbit expand and promote appropriate facial growth. For eyes with meaningful vision, clear material is used in order to avoid deprivational amblyopia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree

 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access