Contributor’s Statement
Dr. Alexandre Mendonça Munhoz is the principal investigator of this study. The principal investigator made significant contributions to the conception and design of this study and made substantial contributions to the acquisition, analysis, interpretation of data, and manuscript preparation. The author revised the article for intellectual content, gave final approval of the version to be published, and has sufficiently participated in the work to take public responsibility for appropriate portions of the content.
Introduction
Breast-conserving surgery (BCS) for early breast cancer treatment continues to be one of the most frequently performed oncologic surgeries worldwide. The introduction of the oncoplastic approach as well as new surgical techniques have led to widespread acceptance of immediate and delayed reconstruction following BCS.
Advances in oncoplastic surgical techniques have reduced surgical morbidity and can thus preserve breast shape and lead to better aesthetic outcomes. Even though most partial mastectomy defects can be treated with primary closure, the outcome may be aesthetically unpredictable and result in contour abnormalities. Oncoplastic techniques are classified as volume displacement or replacement procedures. There is no consensus as to the best approach, and the criteria for selecting an optimal technique are determined by the surgeon’s experience and the size of the defect relative to the remaining breast. The advantages of oncoplastic breast reconstruction should include reproducibility, low interference with oncologic treatment, and acceptable long-term results. Surgical planning should include an assessment of patient preference, addressing individual reconstructive requirements and to custom tailor each individual reconstruction.
Even though it is widely used today, lipofilling or lipomodeling (more specifically described as autologous fat grafting/transfer, AFG) is an old concept. Despite the advantages AFG offers, its use in reconstructing BCS defects is controversial, particularly with regard to aesthetic results and oncologic outcomes. As of this writing, few clinical studies have assessed outcomes after AFG to an unfavorable recipient site, and studies focused on breast conservation with immediate AFG are similarly lacking. Some authors maintain that this is because AFG is usually performed by plastic surgeons, whereas oncologic/breast surgeons perform BCS. Today, a new generation of oncoplastic surgeons is emerging with training in both breast and plastic surgery, and the number of studies on this topic is expected to increase in the coming years.
Although reconstruction following BCS has a high rate of patient satisfaction, some patients may present unsatisfactory results and require surgical revision. In our experience, many of these reoperations are required for problems related to the soft tissue such as local irregularities and implant visibility/rippling rather than reconstruction failure. As with total breast reconstruction, there has been a resurgence in the use of AFG following BCS for a variety of indications over the past 10 years. Although refinement in AFG procedures has improved reproducibility, a standardized technique is lacking, and its relevance as an associated technique has yet to be investigated. It can be assumed that if AFG, BCS, and oncoplastic reconstruction are equally reproducible and involve similar risk and surgical time, the feasibility of combining all of these techniques is now realistic.
The objective of this chapter is to provide an overview of BCS reconstruction incorporating oncoplastic techniques and AFG. Although all these techniques are well-studied procedures, few detailed clinical reports specifically address the operative planning, outcomes, and complications following AFG. As a result, this chapter presents a detailed description of our method, including the preoperative evaluation and intraoperative care for patients undergoing primary and secondary reconstruction. The surgical technique, advantages, and limitations are also discussed. When combined with clinical expertise, the evidence provided in this chapter will help plastic surgeons better counsel and educate patients to achieve reproducible and predictable aesthetic outcomes.
Patient Selection
Preoperative History and Information
Breast conservation and AFG should only be performed after full informed consent, primarily because this is a novel investigative technique. Although most clinical studies to date have demonstrated this to be effective, the majority are retrospective with a low level of evidence. The initial consultation should clarify the patient’s medical history and expectations. Each patient is informed in detail about the technical aspects, advantages, disadvantages, and potential early and late complications. In our practice, we emphasize three main aspects: the possibility of fat resorption, multiple fat grafting procedures may be necessary, and that aesthetic outcome will vary with changes in body weight. It is crucial to discuss the risk of local recurrence and discuss studies that have demonstrated that local recurrence has not been linked to AFG. Although complications in the donor area are infrequent, the patient must be advised that ecchymosis, hematoma, prolonged swelling, and minimal scarring may occur.
Patient Evaluation and Considerations
Before AFG, all patients should undergo ultrasound, mammogram, and breast magnetic resonance imaging (MRI). Following AFG, patients should have breast ultrasound and mammogram 6 months after the final AFG procedure. Before surgery, an accurate physical examination is performed with the patient in a standing position. The cancer-affected breast is compared with the contralateral breast for planning in the regions requiring repair. It is important to evaluate symmetry, shape, volume, the position of the nipple–areolar complex (NAC), and the presence of local radiotherapy (RT) effects including fibrosis and retractile scars. Physical examination and manipulation of the entire breast and the quadrant area permits calculation of the volume of fat that needs to be harvested as well as planning for associated procedures like percutaneous fasciotomies and contralateral symmetrization. The abdomen is usually the preferred donor site because it does not require changes in patient position and tends to contain an adequate volume of fat. An alternative choice is the outer or inner thigh.
Indications and Contraindications
Indications and Timing
In patients having immediate reconstruction, the surgical process is facilitated because the ablative and reconstructive procedures occur in the same operative setting. Because there is no scar or fibrosis at the initial operation, breast reshaping is simplified and the aesthetic outcome is usually improved. Kronowitz et al demonstrated that immediate repair following partial mastectomy is preferable over a delayed procedure because the incidence of complications is usually decreased. Our experience has demonstrated that the complication rate following radiation (for delayed BCS reconstruction) is higher when compared with reconstruction without RT. Clinical studies have demonstrated that patients with larger breasts present more radiation-related complications than patients with breasts of normal volume. Another point is that immediate reconstruction permits wider local tumor excision and may reduce the incidence of margin involvement.
Despite the benefits of the immediate approach, there are also limitations. The surgical time may be longer, and specialist training is required to learn and properly apply these procedures. As a result, for some specific patient groups, delayed reconstruction should be considered. Radiation is known to generally result in some degree of breast fibrosis, and in some cases the final breast contour cannot be predicted at the time of the initial BCS. The delayed approach allows the plastic surgeon to wait until the postoperative changes in the deformed breast stabilize. Our experience has demonstrated that there is insufficient space for AFG in most cases of immediate reconstruction primarily because the skin flaps have been undermined compromising the infiltration of fat.
Despite these limitations, studies have described immediate BCS reconstruction through AFG injection into the tissues beyond the margins of the wide excision site. Khan et al evaluated the aesthetic outcomes of patients after BCS and immediate AFG reconstruction compared with BCS alone. In a series of 71 patients with median follow-up of 36 months, the authors demonstrated significantly better aesthetic results from AFG ( p < 0.001) and fewer local breast symptoms ( p = 0.0045) compared with the BCS only group. Other authors performing studies without a comparison group also observed satisfactory outcomes following immediate AFG reconstruction.
Contraindications and Limitations
There are no formal contraindications to AFG as an adjunct to oncoplastic reconstruction. Ideal candidates are at normal weight or overweight and have small to moderate breast defects. Thin patients are sometimes challenging because of the lack of sufficient fat donor sites; however, adequate volumes of fat can often be harvested from the hip and flank regions. It is important to emphasize that an additional 70% of the volume of fat needed to reconstruct the BCS must be harvested to compensate for the portion of fat lost during decantation/filtration process ( Table 14.1 ).
| Indications | Contraindications |
|---|---|
|
|
|
|
|
|
|
|
Autologous fat grafting following RT can be challenging due to the effects of radiation that include skin retraction, severe fibrosis, and distortion of the NAC. In some situations, and depending of the severity of the deformity, AFG is not an adequate technique, and several procedures may be required. Additionally, the tissues in irradiated areas lack elasticity that can limit the volume of fat that can be injected. In this scenario, the patient should be informed that a 4–6 month interval between procedures is recommended. When the radiation changes are severe, alternative strategies such as local or distant flaps should be considered. Mastectomy is sometimes the best option in refractory cases.
Operative Approach
Breast Defect Classification
Several classification schemes have been developed to define breast deformities and proposed reconstructive techniques. These strategies involve primary closure, breast reshaping, and local and distant flaps. The role of AFG is sometimes omitted from these strategies based on the tissue deficit and the presence of RT effects. Most articles include these options within a broader category of complex breast defects with few clinical studies having been published that propose an algorithm for reconstruction.
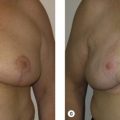
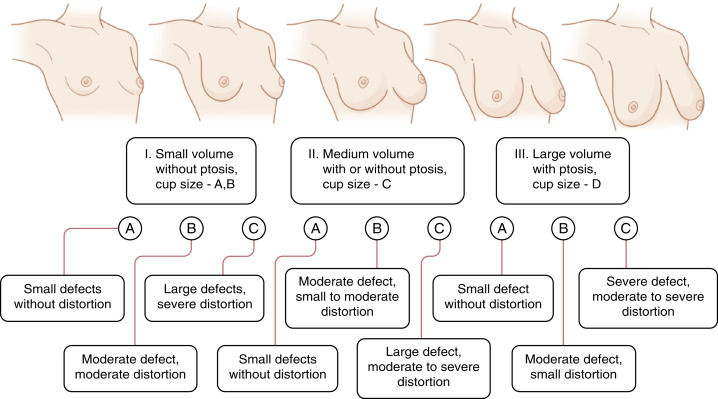
Our 20 years of experience with oncoplastic surgery has enabled us to identify various patterns of acquired breast deformities and develop an algorithm for immediate and delayed BCS reconstruction based on initial breast volume, the extent/location of glandular tissue resection, and the remaining available breast tissue. Each defect is unique and has its own specific reconstructive requirement to achieve a desired aesthetic outcome. With this in mind, partial breast defects can be classified into one of three types ( Fig. 14.1 ):
Type I: Defects include resection in a smaller breast without ptosis. Type IA defects involve minimal defects that do not cause volume distortion, with resected tissue accounting for less than 10–15% of total breast volume. Type IB defects involve moderate defects that generate moderate volume distortion, and resected tissue is between 15–40% of total volume. Type IC defects are large and cause significant volume distortion, with resected tissue comprising more than 40% of total breast volume.
Type II: This group includes tissue resection in medium-sized breasts with/without ptosis. Type IIA involves small defects that do not cause significant volume distortion. Type IIB defects are moderate and cause minor/moderate volume alteration. Type IIC defects cause moderate/large volume variations in the breast shape.
Type III: This group includes tissue resection in large breasts with ptosis. Type IIIA defects are small and do not cause significant aesthetic deformity. Type IIIB involves moderate defects leading to minor/moderate volume alterations. Type IIIC defects are large and cause significant volume alteration ( Scheme 14.1 ).
Operative Techniques: Classic Oncoplastic Procedures and AFG
Theoretically, most oncoplastic techniques can be associated with AFG as a complement or surgical refinement. Surgical planning should include the breast volume, tumor location, extent of glandular tissue resected, and should especially address individual reconstructive requirements, providing an individualized reconstruction tailored to each patient. Evaluation of BCS reconstruction must subsequently consider these important points, and only then should the proper technique or a combination of procedures (AFG+mammaplasty, AFG+local flaps, etc.) be chosen. In our experience, most reconstruction techniques incorporate one of six surgical options: breast tissue advancement flaps (BAF), lateral thoracodorsal flaps (LTDF), bilateral mastopexy (BM), bilateral reduction mammaplasty (BRM), latissimus dorsi myocutaneous flaps (LDMF), and lateral/anterior intercostal perforator flaps (AICAP, LICAP).
Types IA, IIA, and IIIA: Repairs generally include BAF when the defect is spherical or rectangular. The breast tissue is advanced along the chest wall or beneath the breast skin flap to fill the tumor defect. To achieve a better aesthetic outcome without significant skin retraction, superficial undermining can be performed between the breast tissue and skin flap, preserving the skin blood supply. Usually no contralateral breast surgery is required in these patients. In both immediate and delayed reconstructions, AFG can be indicated as a complementary procedure. The major limitation of AFG is related to the degree of retraction and local fibrosis, a fact that can determine the need for serial percutaneous fasciotomies and multiple surgical procedures. Patients with skin laxity, no previous RT (rarely observed, except in benign cases), and no fibrosis can be treated in one or two stages. In patients with moderate fibrosis and local RT effects, external expansion, and more surgical steps with serial percutaneous fasciotomies may be necessary to achieve a satisfactory result.
Type IB: In patients with lateral defects, LTDF can be indicated. As described elsewhere, this local flap is planned as a wedge-shaped triangle located entirely on the lateral aspect of the thorax and then rotated to the lateral breast defect. Although additional scars are necessary, these are placed in the lateral region with a satisfactory outcome. In very thin patients who do not have sufficient volume in the flap area, AFG can be performed to increase flap volume. In this situation, care should be taken with the volume injected to avoid ischemia of the flap and loss of results. In patients with central and medial tumors, LDMF or lateral and anterior intercostal perforator flaps (AICAP, LICAP) can be indicated. Local flaps and especially LTDF are useful techniques for upper outer or lower outer defects. Using tissue located adjacent to the defect provides matching color and texture for the breast, and when this technique is associated with AFG large volumes can be achieved. In our practice we have used LDMF to replace skin and glandular tissue resected during oncologic surgery. This is frequently indicated for severe defects where there is not enough breast tissue to perform the reconstruction. As with LTDF flaps, AFG can be performed carefully to increase the volume of the flap and optimize reconstruction. The LDMF flap provides the possibility of intramuscular fat grafting, which permits safer transfer of a larger volume of fat than non-muscular flaps (LTDF and perforator flaps).
Type IC: Defects are converted to a skin-sparing mastectomy (SSM) and reconstructed with an appropriate technique. AFG can be indicated as a complement for total reconstruction with alloplastic tissue (expanders and implants) or refinement for autogenous tissue (abdominal flaps).
Type IIB: Defects are frequently reconstructed with BM techniques when there is sufficient breast tissue to perform the reconstruction. The preoperative appearance can be improved with smaller and more proportional breasts. This technique facilitates radiation therapy in the remaining breast tissues with acceptably low complication rates. Surgery is usually performed on the opposite breast to obtain appropriate symmetry, particularly in breasts with severe ptosis. With a well-trained surgical team, the procedure can take place simultaneously on both sides, consequently reducing operative time. AFG can usually be indicated to refine BM reconstruction to treat tissue irregularities frequently observed after adjuvant RT treatment. Mild cases may require only percutaneous fasciotomies and AFG can be performed ; in more severe cases, previous expansion (using the Brava system), serial percutaneous fasciotomies, and several surgical stages may be necessary.
Type IIC: Defects are analyzed individually according to the size of the breast defect in relation to the remaining breast tissue available. During evaluation, the patient is positioned upright to assess the amount of remaining glandular tissue. Type IIC can be subclassified into favorable and unfavorable defects; if there is enough tissue to create an adequate breast mound, the defect is classified as favorable. For lateral defects, extended LTDF or LTDF associated with AFG may be indicated. In patients with central and medial defects, extended LDMF or perforator flaps can be indicated. When insufficient breast tissue remains, the breast defect is classified as unfavorable and SSM and total reconstruction are indicated, and AFG may be indicated as a complement to total reconstruction.
Type IIIB: Defects are frequently reconstructed with BRM techniques. The most favorable tumor location is in the lower breast pole, where a conventional superior pedicle or superior-medial technique can be utilized. In patients with central tumors, an inferior pedicle is used to carry parenchyma and skin into the central defect.
Type IIIC: Breast defects are analyzed individually. When the defect is favorable the deficiency is most frequently reconstructed with BRM. Marked reshaping of the breast with available tissue and similar contralateral breast reduction are then performed. In patients with an unfavorable relation, SSM and total breast reconstruction with an appropriate technique can be indicated. As mentioned for Types IC and IIC, AFG can be performed to refine total reconstruction with alloplastic or autogenous tissue.
Operative Techniques: AFG Procedure
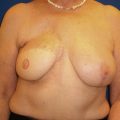
AFG is extensively used in reconstructive surgery to restore volume and contour defects with technical variations on fat harvesting, preparation, and grafting. Although various surgical procedures have been described, including washing, centrifugation, decantation, and previous expansion, there is still discussion on intake, outcome, complications, and long-term results. Debate also continues not only on the optimal AFG technique but also the preparation regime for the recipient site. Some authors advocate the preoperative use of negative-pressure systems ; basically, progressive external breast skin expansion results in increased local vascularization, less pressure, and more free space for AFG. Mirzabeigi et al evaluated safety and outcomes following external expansion of the breast and AFG following BCS in a series of 27 AFG sessions on 20 patients with mean follow-up of 2.3 years. These authors found no cases of local relapse following AFG and no difference in complication rates between patients where the external expansion was and was not used. External expansion permitted a significantly greater initial fill volume than those that were not externally expanded (219 mL vs 51 mL). Despite these results, other authors have demonstrated that one or two AFG sessions were sufficient to obtain a satisfactory outcome in most patients following BCS reconstruction without previous external expansion. As of this writing, no comparative controlled randomized study has made a detailed comparison of AFG with previous external expansion and AFG alone. Furthermore, there are limited clinical data indicating the use of external negative-pressure systems for treatment of BCS defects, and consequently more research is needed on the role of the external expansion, local effects of negative pressure, and whether this therapy has an effect on angiogenic stimulation and cell proliferation.
As for harvesting and preparation in AFG, Coleman and Saboeiro introduced the “structural fat grafting” concept and pointed out the relevance of extracting nonviable aspirate components by centrifugation. The present technique has gained clinical application and has become important in a variety of procedures described in many other studies. Although it is currently our preferred technique (Coleman-modified associated with closed systems), this procedure is controversial in the literature. Khater et al observed that more active preadipocytes were maintained in noncentrifuged adipose tissue, which could potentially lead to enhanced fat survival. Similarly, Rohrich et al performed a quantitative analysis of the role of centrifugation and harvest site and found that the fat survival rate after centrifugation was no better than after filtration.
Several fat-processing techniques have recently become available to boost the predictability of AFG. Some of the new procedures offer much faster fat processing, which is especially productive for large-volume fat grafting. One such system uses the PureGraft device (Cytori Therapeutics, San Diego, CA, USA), which involves washing and filtering the fat to prepare the grafts.
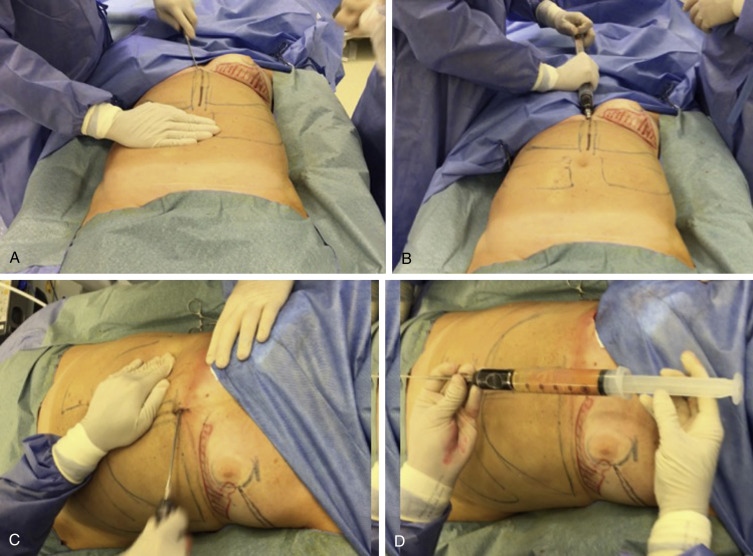
AFG Technique
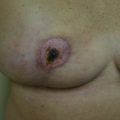
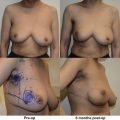
The AFG procedure applied after BCS reconstruction is derived from the technique applied for total breast reconstruction and aesthetic breast augmentation. We usually begin by identifying the best donor site areas, which are marked before the procedure with the patient in an upright position. Possible donor sites include the abdomen, flanks, inner thigh, trochanteric region, and distal thigh. For most patients, the abdominal wall serves as the preferred donor site followed by the lateral thigh and flank regions. Abdominal fat is harvested through a single periumbilical and suprapubic incision, whereas suprailiac incisions are used for the flank (one on each side) and trochanteric area. We use a modified Coleman technique for AFG harvesting. Following injection of local anesthesia (40–00 cc/area of 1% lidocaine and 1:80,000 epinephrine), fat is harvested using a blunt 3-mm cannula connected to a 60-cc syringe (Byron Medical, Inc., Tucson, AZ, USA). To reduce the surgical trauma involved in AFG, mechanical aspiration is not used (see Fig. 14.1 ). Since 2016, our practice has been using closed fat filtration systems such as PureGraft (Cytori Therapeutics, San Diego, CA, USA), which is thought to have some advantages over simple decantation or centrifugation. First, it is a closed system, which avoids graft contamination and possible infection. The second advantage is that processing time is accelerated as only about 10 minutes are needed to obtain 300 cm 3 of AFG. After this step, the purified fat is transferred into 3-cc syringes for injection through blunt cannulas. Strict sterility is maintained, and prolonged exposure to air is avoided throughout the entire process ( Fig. 14.2 ). Two to four small incisions are made in each breast, and the fat is injected into the subcutaneous tissue near the defect area of the flap. We suggest using a 3.0-cc syringe connected to a 1.9–2.1-mm cannula, which allows the controlled precision necessary to graft 0.1–0.5 cc of fat per centimeter of cannula movement. In our experience, large cannulas and syringes do not provide the necessary precision for this maneuver. The AFG technique relies on preoperative topographic markings, grafting small amounts of fat through multiple passes along several planes from deep to subcutaneous tissue; this process requires use of blunt cannulas in the subdermal region. In some cases, a blunt cannula is utilized before this stage, depending on the degree of fibrous tissue observed ( Fig. 14.3 ). The fat is slowly injected with a retrograde technique in small volumes without putting too much pressure on the syringes, following the principle of the “spaghetti” technique. We always avoid overfilling in AFG to limit ischemia and necrosis. With this in mind, care must be taken in cases with severe fibrosis associated with the local effects of RT, which result in noncompliant, hard tissue with limited distensibility ( Scheme 14.2 ). In these cases, as the fat is grafted the tissue compliance becomes tense, and smaller volumes of fat can lead to a major increase in pressure. This is very important, because injecting too much fat (“fat lakes”) into one small region can result in ischemia because the revascularization is insufficient for large-volume fragments. In patients who have previously undergone RT, BCS defects are generally much less compliant and much less tolerant of large volumes of AFG. Therefore, to improve vascularization and AFG intake, we strongly recommend that fat be spread carefully as a fine mist of small droplets using the sprinkler principle, avoiding higher tissue pressure ( Scheme 14.3 ). If necessary, percutaneous aponeurectomy with an 18-gauge needle can be an important way of releasing scar tissue. Care must be taken to avoid wide subcutaneous undermining during percutaneous aponeurectomy. The goal of this maneuver is to create a mesh-like pattern within the fibrous tissue and not to create an open space or lake-like effect. After the fat injection, the injected area is carefully reshaped to adapt to the outline of the desired surface ( Figs. 14.4 and 14.5 ).