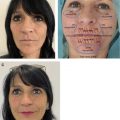
Craniofacial deformities represent a great challenge for the patient and the plastic surgeon. Fat grafting has allowed a shift in paradigm for craniofacial reconstruction by providing a less invasive and safer alternative than traditional reconstructive options. Increasing evidence supports its use with optimal results. This article examines the evidence and practical aspects involved in the decision making and technique of fat grafting to treat secondary craniofacial deformities.
Key points
- •
Careful facial analysis is crucial for optimal outcomes.
- •
Fat grafting provides a minimally invasive technique to improve facial contouring and symmetry in craniofacial patients with secondary deformities.
- •
The Coleman technique for harvesting, processing, and grafting provides a reliable strategy for consistent results.
Introduction
Craniofacial deformities represent a great challenge for the patient and the plastic surgeon. They occur in the setting of congenital, traumatic, or oncological etiology and may lead to psychological distress and functional impairment. Reestablishment of normal craniofacial morphology is important to allow patients to return to their normal activities and better reintegrate into society. Furthermore, improvement of the quality of injured soft tissues is important to improve the mobility of the facial region and to prevent sequelae like wound breakdown and worsening fibrosis.
The transfer of autologous adipose tissue has emerged as a suitable alternative for the treatment of craniofacial deformities. Fat grafting was first described as a cosmetic facial filler in the 1980s. Coleman, however, was responsible for developing a fat grafting technique that allowed more reproducible and reliable results, with quoted graft retention rates as high as 90%. The key steps involve the aspiration of small fat aliquots, fat centrifugation, separation of unwanted lipoaspirate components (local anesthetic, oil, blood, and other noncellular material) and injection of fat as tiny aliquots.
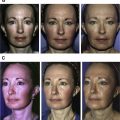
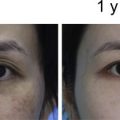
Fat grafting is a minimally invasive, efficacious, safe, and cost-effective technique that has low complication rates and high patient satisfaction rates. It has been used to treat deformities all over the body to improve areas with significant volume deficits. Fat grafting has traditionally been applied to aesthetic surgery but increasing evidence is demonstrating its benefits in breast reconstruction and congenital craniofacial deformities like hemifacial microsomia. The adaptation of these techniques to craniofacial deformities from surgery or trauma seems simple but various factors come into play making its translation challenging with uncertain resorption rates. First, the facial recipient bed is now scarred and fibrotic, reducing its vascular supply and compliance to support fat engraftment. Second, the base of the zone of injury often is lined by a mesh or other avascular foreign body, which further reduces the recipient bed vascular supply and thus engraftment. A paucity in the literature exists describing the role of fat grafting to correct traumatic or surgical craniofacial deformities. Only a few small case series and cohort studies have been published, but they group congenital, traumatic, and surgical craniofacial deformities together. Nonetheless, these studies confirm that fat grafting is safe, leading to no major complications and to high patient satisfaction rates and favorable aesthetic outcomes, based on photographs.
The authors’ group recently conducted a prospective cohort study assessing the safety and efficacy of fat grafting for the treatment of posttraumatic and postsurgical craniofacial deformities. The study confirmed the safety of fat grafting with absence of major complications. Volume retention averaged 63% at 9 months, with the 3-month point strongly predicting the final result. There was no correlation between the total volume injected and retention rate. Subsequent grafting procedures had similar volume retention as the first round, leading to a significant correlation between volume retention percentage at the first round of injection to retention of subsequent injections. An improvement was noticed in satisfaction with physical appearance, social relationships, and social functioning quality of life.
The authors believe, therefore, that autologous fat grafting for craniofacial deformities is a great alternative because it is less invasive and safer than traditional reconstructive options, it reaches volume stability at 3 months, and leads to positive patient-reported outcomes. This article examines the practical aspects involved in the decision making and technique of fat grafting to treat secondary facial deformities.
Preoperative evaluation and special considerations
A thorough history and physical examination are the basis for successful outcomes in fat grafting. Care should be taken in understanding the deformity etiology, prior surgical treatment, and reconstructive techniques previously attempted. Evaluation of the patient’s preoperative medical status must include conditions that can affect the survival of fat grafting, such as smoking and tobacco use and history of prior infection or postsurgical infection. A surgical history should be taken, including prior surgeries, complications to anesthesia, and prior liposuction or fat grafting. A personal or family history of clotting or bleeding disorders should be noted. If there is an allergy to lidocaine, it should not be used for in the infiltration solution. A thorough medication history should be obtained to determine if the patient takes anticoagulants or supplements that affect coagulation. After a general examination, a detailed analysis of the face should be conducted focusing on basic principles of facial aesthetics, assessment of texture and quality of the skin, identification of contour deformities, volume deficiencies, scars, fibrotic tissue, and presence and/or exposure of plates or other foreign bodies. A through assessment of potential fat harvesting donor sites to determine presence of sufficient fat and assess for prior liposuction deformities to avoid worsening of those sites during harvesting.
All medications that interfere with the platelet function should be stopped 2 weeks prior to surgery. Smoking should be stopped at least 1 month prior to surgery. Throughout the preoperative planning, the surgeon should gauge goals of the patient, ensure that the expectations are realistic, and ensure that fat grafting is the appropriate treatment modality.
Indications for fat grafting include a wide range of cosmetic and reconstructive deformities. Although the procedure is well tolerated, some contraindications exist, including hematologic abnormalities and anticoagulant medication use.
Surgical procedure
Donor Site Selection
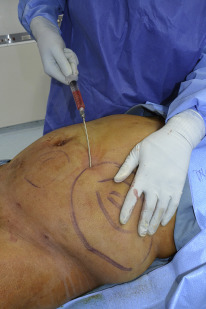
Fat can be harvested from anywhere in the body. Because studies have shown that the site has no impact on postprocessing fat yield, the decision of harvest site is usually based on safety, ease of access, and preference of the patient. Patients normally are placed in a supine position to provide easy access to the inner thighs, flanks, and abdomen for harvesting and the face for transfer. The donor and recipient sites are then prepared with povidone-iodine and the patient is draped in a sterile fashion.
Harvesting
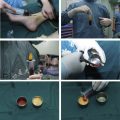
Infiltration solution is placed through 2-mm incisions sites. If the surgery is performed under local anesthesia, the authors’ preference is a solution composed of 0.5% lidocaine with 1:200,000 epinephrine buffered with sodium bicarbonate infused with a Lamis infiltration cannula (Mentor Worldwide, Santa Barbara, California). If the surgery is performed under intravenous sedation or general anesthesia, the authors’ preference is a solution composed of 0.2% lidocaine, Ringers lactate with 1:400,000 epinephrine. The volume infiltrated is lower than the expected volume of lipoaspirate and is left in for 10 minutes. Tumescent solution is not used because of the potential for mechanical damage to fat parcels and because of prolongation of the procedure due to an excessive fluid fraction in the aspirate. Manual liposuction is performed using a 10-mL syringe and a 9-hole harvesting cannula (12- gauge or 14-gauge, 15 cm or 23 cm long, depending on the volume needed) as shown in Fig. 1 .
Processing
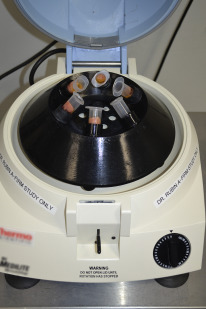
The syringe is then connected to a Luer lock cap (Becton, Dickinson and Company, Franklin Lakes, New Jersey) and the plunger is removed. The syringes are then spun in a sterilized centrifuge at 1286 g for 3 minutes ( Fig. 2 ). The oil in the top supernatant layer is decanted and the lower aqueous layer is allowed to be drained from the bottom of the syringe by removing the Luer lock cap. A neurosurgical cotton patty is used to wick away the remaining superior oil ( Fig. 3 ). The processed fat is then transferred into 1-mL Luer lock syringes ready for injection.