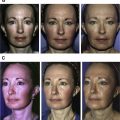
The author provides his preferred approach to fat grafting for facial rejuvenation. The preferred donor sites include low abdomen and inner thigh. Fat grafts should be harvested with low negative pressure to ensure the integrity and viability of adipocytes. Fat grafts can be processed with proper centrifugation that can reliably produce purified fat with concentrated growth factors and adipose-derived stem cells, all of which are beneficial to improve graft survival. The approach described in this article is supported by the most scientific studies and thus may provide a more predictable long-lasting result if performed properly.
Key points
- •
The preferred donor sites for fat grafting should be the lower abdomen or inner thigh.
- •
Fat grafts should be harvested with low negative pressure to ensure the integrity and viability of adipocytes.
- •
Fat grafts can be processed with proper centrifugation that can reliably produce purified fat and concentrated growth factors and adipose-derived stem cells.
- •
Preinjection of local anesthetic with epinephrine to planned fat grafting sites prevents intravascular injections and possible fat embolism.
- •
Fat grafts should be injected with gentle injection of a tiny amount per pass in multiple tissue planes and levels and with multiple passes to ensure maximal contact of the graft with vascularized tissue in the recipient site.
Introduction
Fat grafting is considered an excellent option for facial rejuvenation because fat is abundant, readily available, inexpensive, host compatible, and can be harvested easily and repeatedly. Compared with any available synthetic filler, fat can be an ideal filler for facial rejuvenation because the clinical result after fat grafting can be permanent without any concerns or complications related to fillers. However, the overall survival rate after fat grafting may still be less optimal. To improve fat graft survival has therefore been constantly the driving force for clinicians to search for better techniques of fat grafting.
Since mid-1990s, Dr Sydney R. Coleman from New York City has championed and popularized the technique primarily for facial fat grafting. His technique, also referred to as the Coleman technique, emphasizes proper harvest, process, and placement of fat grafts.
For the last 10 years, fat grafting has become a popular procedure in plastic surgery, especially for facial rejuvenation either with or without other surgical procedures. In this article, the author introduces his preferred and more scientifically sound technique for fat grafting to the face. He also describes why each step should be performed based on most recent scientific studies by many investigators. Several case examples are presented to highlight those important principles in facial fat grafting.
Preoperative evaluation and special considerations
Each patient’s general health and past medical or surgical history should be reviewed first. Concerns about his or her facial aging from each patient should carefully be evaluated. The quality of facial skin and the anatomy of the face, including symmetry, signs of facial aging such as excess skin, ptosis of structures, prominent lines, and soft tissue atrophy in certain anatomic areas, are analyzed and documented. The potential donor sites for fat graft harvest are also examined. The detailed plan for facial fat grafting can be formulated and communicated with the patient.
Anesthesia for harvest of fat grafts can be performed under general anesthesia or local anesthesia with intravenous sedation. The tumescent solution used for donor site analgesia or hemostasis should contain the lowest concentration of lidocaine possible because its high concentration may have detrimental effect on the adipocyte function and viability. In general, the author uses 0.03% of lidocaine in 1 L of Ringer lactate solution. The tumescent solution also contains epinephrine with a concentration of 1:200,000. Epinephrine can precipitate vasoconstriction in the donor sites as well as the recipient sites, which may decrease blood loss, bruising, hematoma, and the possibility of intraarterial injection of the transplanted fat especially when injecting around periorbital or temporal area.
Whether overcorrection is necessary or not for fat grafting remains unclear. Because the viable fat grafts are only observed in the peripheral zone approximately 1.5 mm from the edge of the grafts and the percentage of graft viability depends on its thickness and geometric shape, overcorrection for “better” graft survival in the recipient site seems to be lack of scientific support. In addition, significant overcorrection may increase the incidence of fat necrosis and subsequent calcification or even severe infection. Therefore, “significant” overcorrection should be avoided until its necessity and safety can be confirmed by future studies.
Because overall take rate of fat grafting by even experienced surgeons ranges from 50% to 80%, additional procedures are always necessary to achieve an optimal outcome. However, there is no scientific study that has addressed the timing of subsequent fat grafting. So far, only one “expert” opinion has been mentioned in the literature regarding this specific issue : “the timing of additional fat grafting sessions should be deferred until 6 months postoperatively to diminish the “inflammatory response” in the grafted area.”
It is often difficult to assess the surgical outcome during the first few weeks after fat grafting. In general, the extent of swelling and the waiting period that it needs to resolve is also volume dependent. It has been observed that the transplanted fat gradually loses its volume with time and usually becomes stabilized at 3 months postoperatively if surgical recovery is uneventful. Therefore, the timing of a subsequent fat grafting procedure should be deferred to at least 3 months after previous transplantation.
Surgical procedures
Donor Site Selection
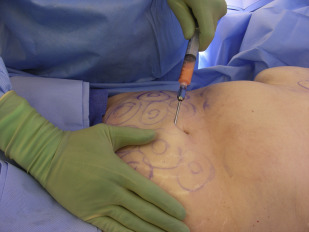
As a rule, donor sites are selected that enhance body contour and are easily accessible in the supine position, which is the position that is used for almost all facial fat grafting procedures. Although there is no evidence of a favorable donor site for harvest of fat grafts because the viability of adipocytes within the fat grafts from different donor sites may be considered equal, higher concentration of adipose-derived stem cells (ADSCs) is found in the lower abdomen and inner thigh in one study. In addition, in younger age group (<45 years old), fat grafts harvested from both lower abdomen and inner thigh have higher viability based on a single assay test. With what is known about the potential role of ADSCs in fat grafting, the lower abdomen and inner thighs should, therefore, be chosen as the “better” donor sites for fat grafting to the face ( Fig. 1 ). These donor sites are not only easily accessible by the surgeons with a patient in the supine position but also scientifically sound as long as patients have adequate amount of adipose tissue in those areas. In the author’s practice, a total of 30 cc is usually needed for most facial rejuvenation cases.
Fat Grafting Harvesting
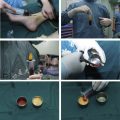
The syringe aspiration, as a relatively less traumatic method for harvest of fat grafts, is supported by the more recent studies and should be considered as a standardized technique of choice for harvest of fat grafts. However, this technique can be time consuming even for experienced surgeons and the large quantity of fat grafts may not easily be obtained with this technique.
Placement of incisions can be done with a No. 11 blade in the locations where the future scar can easily be concealed. The size of incision is about 2 to 3 mm. A small clamp is used to dilate the underlying subcutaneous tissue through the incision to allow insertion of the harvesting cannula with ease. The aesthetic solution is then infiltrated to the donor site 10 to 15 minutes before fat extraction, which makes harvesting of fat graft easier and less traumatic. The tip of the infiltration cannula is usually blunt and has one opening on the side. The ratio of aspirated fat to tumescent solution should be about 1:1 so that each pass of fat extraction can be more efficient.
A 10 cc Luer lock syringe is used and connected with a harvesting cannula. For harvesting fat grafts from the lower abdomen or inner thigh, a newly designed harvesting cannula with multiple side holes is used ( Fig. 2 ). This kind of cannula can be more efficient for fat extraction. A gentle pull back on the plunger creates a 2 cc space vacuum negative pressure in the syringe. With gentle back and forth movement of the syringe, the fat is gradually collected inside the syringe ( Fig. 3 ). After harvest, all incision sites are closed with interrupted sutures once excess tumescent fluid or blood is milked out.

Fat Graft Processing
Several methods have been proposed to effectively remove the infiltrated solution and cell debris within the lipoaspirates and to obtain more concentrated fat grafts. However, it is the most controversial and disagreeable issue in fat grafting even among many experts in the field. Common methods for processing fat grafts include centrifugation, filtration, or gravity sedimentation.
Centrifugation, as proposed by Coleman, is the author’s preferred method to process fat grafts. There are several advantages of centrifugation of fat grafts. More viable adipocytes are found at the bottom of middle layer after centrifugation even with a force of 50 g for 2 minutes base on viable cell counts, and this makes manipulation of fat graft for use easier but with better viability. Recent studies have shown that proper centrifugation can concentrate not only on adipocytes and ADSCs but also on several angiogenic growth factors within the processed fat grafts. Because higher content of stem cell or angiogenic growth factor positively correlated with fat graft survival both in experimental and clinical studies, centrifugation at 3000 rpm (about 1200 g) for 3 minutes seems to offer more benefits for this effectively concentrating adipocytes and ADSCs and should be a valid method of choice for processing fat grafts, especially for small volume fat grafting.
The Luer lock aperture of the 10 cc syringe locked with a plug at completion of harvest is ready for centrifugation ( Fig. 4 ). After careful removal of the plunger, all lipoaspirate-filled 10 cc syringes are placed into a centrifuge and are then centrifuged with 3000 rpm (about 1200 g) for 3 minutes. Greater g-force or longer duration of centrifugation may be harmful to adipocytes and is therefore not recommended.
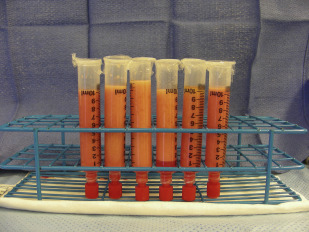
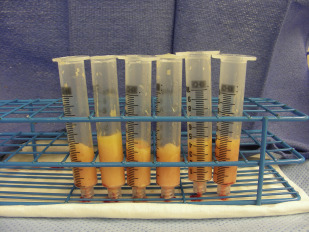
Attention should be made to avoid prolonged exposure of fat grafts to air and to avoid bacterial contamination. After being centrifuged, lipoaspirates with the syringe are divided into 3 layers: the oil content in the upper layer, fatty tissue in the middle layer, and the fluid portion at the bottom ( Fig. 5 ). The oil can be decanted from the Luer lock syringe. The residual oil is wicked with a cotton strip or swab. The fluid at the bottom can be easily drained out once the plug at the Luer lock aperture is removed.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree