Autologous fat graft has limitations, especially long-term unpredictability of volume maintenance. The mechanical enrichment of fat graft with adipose-derived stem cells (ADSCs) could guarantee the survival of fat grafts. After decantation, washing, and centrifugation of lipoaspirate, the authors carried out histochemical analysis and flow cytometry to determine the best layers for preparing ADSC-enriched fat. After centrifugation, the stromal vascular fraction (SVF) was separated by mechanical dissociation and mixed with another layer of intact adipocytes, which was injected into patients. All patients showed volumetric improvement after a single lipotransfer section, without overcorrection. The method is safe, has low cost, and is easily reproducible.
Key points
- •
The preferred donor site for lipotransfer in face should be the lower abdomen. The fat is harvested with syringe, creating a light negative pressure by slowly withdrawing the plunger in a manual gradual manner.
- •
Part of the aspirated fat is allowed to set for decantation and washing with saline. Other part is processed in a centrifuge for mechanical isolation of the stromal vascular fraction (SVF) to prepare the mechanically SVF-enriched fat.
- •
The fat is injected using a multichannel technique, retrogradely in each movement until achieving volume augmentation, avoiding overcorrection.
- •
The postoperative absorption index of the injected enriched fat is low compared with traditional fat grafting.
- •
The mechanically SFV-enriched lipotransfer technique does not use enzymatic process, does not involve any expensive machinery or devices, and is easily reproducible.
Introduction
Fat is an ideal tissue as an autologous substitute with which tissue deficiencies can be treated . It is available in easily accessible subcutaneous depots but also can be molded to reconstruct defects. Unfortunately, despite more than 100 years of clinical use, the clinical longevity of the fat graft is highly variable, is operator-dependent, and has a high ratio of absorption; in particular, the volume of large grafts decreases significantly over time. The presumed mechanisms of tissue loss seem primarily insufficient vascularity, cell death, and other mechanisms, such as mechanical disruption of cells, lipid-induced membrane damage, and apoptosis.
Nowadays, it is known that adipose tissue is rich in regenerative cells, such as stem cells, more than bone marrow. Adipose-derived stem cells (ADSCs) were identified by Zuk and colleagues in 2001. These investigators defined the ADSC characteristics by their ability to differentiate into several mesenchymal lineages. ADSCs can be isolated from the stromal vascular fraction (SVF) through enzymatic process and culturing on plastic because, unlike the other cell types in SVF, they adhere to plastic. The recognition that fat contains multipotent stem cells that can be harvested through liposuction without altering their viability has driven further examination into the potential uses of fat and its ADSCs. The aspirated fat has approximately half the number of the adipose mesenchymal stem cells found in excision whole fat. The relative deficiency of ADSC yield may be one of the reasons for long-term atrophy of transplanted aspirated adipose tissue. The clinical potential of supplementing fat grafts with more ADSCs has been reported as a novel method of autologous tissue transfer, termed cell-assisted lipotransfer. The potential benefits of ADSCs are promising, because they have several advantages over fat grafting alone.
In this article, the authors introduce a structural fat grafting procedure associated with a new model of cell-assisted lipotransfer technique in the management of volumetric deficit of the face in pathologies, such as Romberg syndrome and trauma sequelae, and also in the aged face, to determine an easy and reproducible method that can yield higher concentrations of preserved adipocytes and ADSCs that could guarantee long-lasting results. The enrichment of fat presented in this article is based on another study that compared the effects of the 3 most common fat processing techniques used in plastic surgery (decantation, washing, and centrifugation) on the viability and number of cellular components of aspirated adipose tissue, determining a method that can yield higher concentrations of viable adipocytes and mesenchymal stem cells. The whole surgical lipotransfer was performed bedside, and enzymatic digestion process was not used to prepare the enriched fat, called by the authors mechanically SVF-enriched (MeSe) lipotransfer technique.
Preoperative evaluation and special considerations
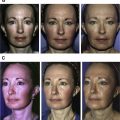
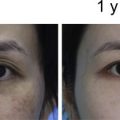
For the management of volumetric deficit of the face, in cases of facial deformities, such as Romberg syndrome and trauma sequelae, the preoperative work-up included laboratory studies, blood test, cardiac examination, photography, and sliced 3-dimensional (3-D) tomography. The amount of fat to be implanted was assessed based on degree volume deficit. In the cases of Romberg syndrome and trauma sequelae, the parameter was the contralateral hemiface that was normal. After obtaining successful results in these unilateral deformities, the mechanically SVF-enriched lipotransfer technique was applied in the treatment of the aged face.
All patients were clearly informed of the benefits, risks, operative complications, and postoperative care and gave their informed consent to the intervention. Furthermore, all patients were submitted to the surgery in hospital environment.
The patients consented under approved guidelines set for human clinical trials by the Brazilian investigation ethics committee board (protocol no. 28063) and the Brazilian Clinical Trials Registry (Rebec. UTN: U1111–1145–3081).
Surgical procedure
Donor Site Selection
When the lipotransfer is executed in the face, donor sites are selected considering the easy accessibility in the supine position, avoiding changes in the position of the patient during the surgery. In general, the authors prefer the lower abdomen as a donor area. In addition to avoiding the change of decubitus, a higher concentration of ADSCs was found in the lower abdomen.
Fat Harvesting
After antiseptic cleaning of the skin with chlorhexidine, the fat was collected from the lower abdomen using the super wet technique. A solution containing normal saline and 0.5% lidocaine with 1:500,000 of epinephrine was injected through a 22-gauge spinal needle before aspiration. It was infiltrated into the area of liposuction at a ratio of 1 mL of solution per milliliter of aspirated tissue. The mean quantity of 200 mL of harvested fat was aspirated using blunt cannulas of 3 mm in diameter and 20 cm in length (tip model 3B, Richter, São Paulo, Brazil) attached to a 10-mL Luer lock syringe, creating a light negative pressure by slowly withdrawing the plunger in a manual gradual manner.
Fat Purification and Mechanical Stromal Vascular Fraction Enrichment of the Fat
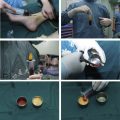
After collection, the adipose tissue was separated into 2 groups. Half of the syringes were allowed to set for decantation under the action of gravity for 15 minutes. After this, the inferior layer, composed mostly of blood, was thrown out and the remaining fat was washed with saline to remove blood and cell elements and left to stand; 3 washes yielded a layer of saline and a supernatant predominantly comprising bright yellow adipocytes. This procedure was done using the same 10-mL Luer lock syringe used during fat harvesting, and the piston was never removed from the harvesting syringe during washing to avoid tissue exposure to air. The other half of the volume of the aspirated fat was processed in a centrifuge for mechanical isolation of the SVF. This half of the collected fat that was placed in capped 10-mL syringes was put in a centrifuge (IEC Medilite Microcentrifuge, Thermoelectron Corporation, Byron Medical, USA) and spun at 3000 rpm for 3 minutes, equivalent to 1286 g. In the final centrifugation process, 4 basic layers were observed macroscopically. The fourth layer, called the pellet, at the bottom of the centrifuged sample, corresponds to the SVF, a pool of regenerative cells that was separated by mechanical dissociation without enzymatic process ( Fig. 1 ).

To prepare the mechanically SVF-enriched fat, each 10-mL syringe with washed fat was enriched with the pellet of 1 centrifuge sample and gently mixed, after connecting to fill Luer lock syringes via syringe adapter (female Luer lock adapter).
One syringe of 10 mL of washed fat and 1 pellet of the centrifuge sample collected from each patient were sent to the laboratory and analyzed by the same group of biotechnicians, less than 24 hours after the collection. After discarding the superior and inferior layers of washed lipoaspirate, the middle layer, which is routinely used for adipose tissue graft, was kept for analysis.
Implant Site of the Mechanically Stromal Vascular Fraction–Enriched Lipotransfer Technique
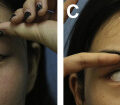
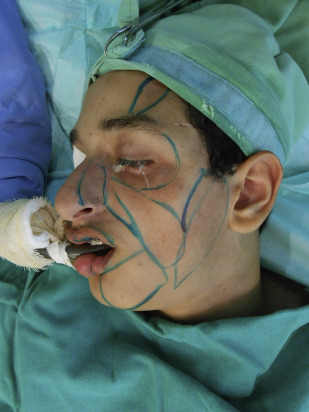
Preoperative markings were done in face, identifying the areas of volumetric deficit ( Fig. 2 ). Supraorbital, infraorbital, and mental nerve block was achieved with 1 mL to 2 mL of 2% lidocaine when under sedation and locoregional anesthesia.
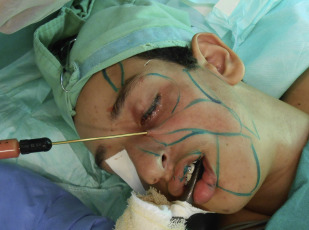
Aimed at carrying out the fat implantation, small scalp incisions were made with an 18G needle and the mechanically SVF-enriched fat was injected into the target sites. The fat was injected using a 2-mm to 3-mm blunt-tipped Coleman cannula connected to syringe, using a multichannel technique, retrogradely in each movement ( Fig. 3 ). The amount of fat to be implanted was assessed based on degree volume deficit, avoiding overcorrection and aiming at observing the degree of fat absorption. The parameter in the cases of Romberg syndrome and trauma sequelae was the contralateral hemiface that is healthy. Fat was homogeneously deposited in the subcutaneous level in multiple layers to achieve volume augmentation in these cases, always avoiding overcorrection ( Fig. 4 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree