![]() Visit Elsevier eBooks+ (
eBooks.Health.Elsevier.com
) for complete set of videos.
Visit Elsevier eBooks+ (
eBooks.Health.Elsevier.com
) for complete set of videos.
Introduction
Burn injury initiates a cascade of pathophysiologic responses that are driven by the patient’s stress and systemic inflammatory response. The systemic release of inflammatory markers is associated with capillary leak causing a fluid shift from the intravascular space into the interstitium. The global endothelial activation and glycocalyx damage in turn enhance the systemic inflammation. The primary injury is exacerbated by immune system hyperactivation, hypermetabolism, and adrenal hyperreactivity. Concomitantly, host defenses are compromised, resulting in significant exposure to microbial insult from commensal and pathologic organisms. The lack of host defenses, immune compromise, and hypermetabolism results in catabolism and often in multisystem organ failure (MOF). The severity of MOF involving three or more organs has been associated with 100% mortality as demonstrated by Kraft et al.
Several scoring systems are currently used to quantify MOF. The Denver 2 system is widely used in research and clinical care by scoring pulmonary, renal, hepatic, and cardiac function from 0 to 3 with a total of 12 points ( Table 24.1 ). There is significantly increased mortality when scores are greater than 3. The Sequential Organ Failure Assessment (SOFA) score is based on scores of six systems: pulmonary, coagulation, liver, cardiovascular, central nervous, and renal (each getting assigned a 0–4 score) ( Table 24.2 ). The SOFA score is incorporated in the Sepsis-3 definition to identify and diagnose sepsis and septic shock. Given that mortality increases with each additional failure of organ system, it becomes imperative to predict MOF and develop predictive markers. Currently, the best predictive model for burn mortality is proteomics combined with clinical covariates. The time course of organ failures and the patterns of multiorgan failures are demonstrated in Fig. 24.1 and Table 24.3 . There are two well-described phases of MOF in burn patients: early and late. The early clinical phase is characterized by challenges in resuscitation leading to hemodynamic failure encompassing renal failure, hepatic failure, gastrointestinal failure, pulmonary failure, and increased risk of sepsis. In the later phase, all organs can technically undergo organ failure, but more common are pulmonary failure, hemodynamic instability, renal failure, gastrointestinal failure, and hepatic failure.
Table 24.1
Denver 2 Criteria for Multisystem Organ Failure
| Component | Measurement | Score | ||||||
| 0 | 1 | 2 | 3 | |||||
| Pulmonary | PaO 2 /FiO 2 | ≥250 | 175–249 | 100–174 | <100 | |||
| Renal | Creatinine | ≤1.8 | >1.8–2.5 | >2.5–5.0 | >5.0 | |||
| Hepatic | Bilirubin | ≤2.0 | >2.0–4.0 | >4.0–8.0 | >8.0 | |||
| Cardiac | Inotropes | See definitions below | ||||||
| Cardiac Score : Scoring for cardiac component is a combination of number and dosage of inotropes administered. | ||||||||
| S = small dose, M = moderate dose, L = large dose | ||||||||
| Patient receives 0 agents : cardiac score = 0 | ||||||||
| Patient Receives 1 Agent: |
Patient Receives 2 Agents: |
|||||||
| Dose size | S | M | L | Dose size | (S, S) | (S, M) | M, M) | (L, anything) |
| Cardiac score | 1 | 2 | 3 | Cardiac score | 2 | 2 | 3 | 3 |
Patient receives 3 or more agents : cardiac score = 3
Table 24.2
Sequential Organ Failure Assessment Criteria
From Dubois MJ, Orellana-Jimenez C, Melot C, et al. Albumin administration improves organ function in critically ill hypoalbuminemic patients: a prospective, randomized, controlled, pilot study. Crit Care Med . 2006;34(10):2536-2540.
| SOFA Score | 0 | 1 | 2 | 3 | 4 |
|---|---|---|---|---|---|
|
WITH RESPIRATORY SUPPORT
|
|||||
| Respiration, PaO 2 /FlO 2 , mmHg | >400 | ≤400 | ≤399 | ≤200 | ≤100 |
| Coagulation, platelets × 10 3 /mm 3 | >150 | ≤150 | ≤100 | ≤50 | ≤20 |
| Liver, bilirubin, mg/dL (μmol/L) | <1.2 (<20) | 1.2–1.9 (20–32) | 2.0–5.9 (33–101) | 6.0–11.9 (102–204) | >12.0 |
| Cardiovascular, hypotension | No hypotension | MAP <79 mmHg | Dopamine ≤5 or dobutamine (any dose) | Dopamine >5 or epinephrine ≤0.1 or norepinephrine ≤0.1 | Dopamine >15 or epinephrine >0.1 or no epinephrine >0.1 |
| Central nervous system, Glasgow Coma Scale score | 15 | 13–14 | 10–12 | 6–9 | <6 |
| Renal, creatinine, mg/dL (μmol/L) | <1.2 (<110) | 1.2–1.9 (110–170) | 2.0–3.4 (171–299) | 3.5–4.9 (300–440) | >5.0 (>440) |
| Or urine output | Or <500 mL/day | Or <200 mL/day | |||
MAP , Mean arterial pressure; SOFA , sequential organ failure assessment.
The time course of organ failure over 60 days postburn.
A. The mortality associated with each individual organ failure. B. Denver 2 scores associated with each organ failure.
(From Kraft R, Herndon DN, Finnerty CC, et al. Occurrence of multiorgan dysfunction in pediatric burn patients: Incidence and clinical outcome. Ann Surg . 2014;259[2]:381-387.)
Table 24.3
Coincidence and Correlation Between Organ Failures
From Kraft R, Herndon DN, Finnerty CC, et al. Occurrence of multiorgan dysfunction in pediatric burn patients: incidence and clinical outcome. Ann Surg . 2014;259(2):381-387.
| Part A | Heart | Lung | Kidney | Liver |
|---|---|---|---|---|
| Heart (77) | NA | 73 | 10 | 16 |
| Lung (230) | 73 | NA | 16 | 22 |
| Kidney (16) | 10 | 16 | NA | 6 |
| Liver (23) | 16 | 22 | 6 | NA |
| Part B | 1 Organ | 2 Organs | 3 Organs | 4 Organs |
| Heart (77) | 4 | 51 | 18 | 4 |
| Lung (230) | 147 | 59 | 20 | 4 |
| Kidney (16) | 0 | 4 | 8 | 4 |
| Liver (23) | 1 | 4 | 14 | 4 |
| Failures |
Part A displays the coincidence of the single-organ failures. Logistic regression revealed a statistically significant relationship between liver failure accompanied by heart and renal failure. Part B depicts the incidence of single and combined organ failures in the patient population.
NA, Not applicable.
Although a patient can survive MOF, the evidence strongly supports that those with failure of greater than three organ systems have a mortality risk reaching 100%.
Porro et al. published that total body surface area (TBSA) greater than 60% in children and greater than 40% in adults was associated with mortality, sepsis, infection, and MOF. Furthermore, Kraft et al. looked to determine the incidence of MOF and its timeline. They determined that respiratory failure had the highest incidence in the first 5 days. Cardiac failure occurred throughout the hospital stay. Hepatic failure increased with length of hospital stay and is associated with high mortality in the later phase. Renal failure had an unexpectedly low incidence but was associated with high mortality in the first 3 weeks. Three or more organ failures were universally fatal in their cohorts. Overall mortality for patients with MOF was 41% compared to 2% without. The Helsinki Burn Center reported that 40% of their adult burn mortalities between 1999 and 2005 were caused by MOF and 40% due to untreatable burn injury. On average, four organ failures were noted in the deaths, with acute renal failure being the most common. Sepsis was associated with MOF in all of their deaths.
Having delineated the importance of MOF, this chapter focuses on the etiology and prevention of MOF, while management will be covered in the critical care chapter ( Chapter 25 ).
Etiology and cellular response
Attempts to define the etiology of MOF range from genomic to cellular, systemic, and epidemiologic studies.
One of the overarching hypotheses is that inadequate oxidative metabolism secondary to hypoperfusion leads to further organ failure and the release of humoral inflammatory mediators causing further cellular dysfunction. In ischemia-reperfusion models, oxygen radicals are generated, resulting in peroxidation of cell membrane lipids and accumulation of activated neutrophils, with progressive cellular and whole-organ dysfunction. Critically ill patients suffer from supply-dependent oxygen consumption because of defects in cellular oxygen extraction and utilization. , This results in inadequate aerobic metabolism. Grossly inadequate delivery of oxygen to cells dependent on aerobic metabolism can lead to cellular dysfunction, followed by organ failure.
Mitochondrial damage is one of the earliest responses to burn injury. There is tissue-specific damage to mitochondrial DNA (which is universally present in all organs and systems) associated with a time-dependent increase in oxidative stress and neutrophil infiltration. Porter et al. investigated mitochondrial dysfunction postburn and found significantly reduced mitochondrial coupling lasting for up to 2 years postburn.
Uncoupling protein 1 thermogenesis increases after burn trauma and is a mechanism of hypermetabolism in burn patients. This response is both adrenergic mediated and responsive to ambient temperature, linking thermal regulation to skeletal muscle metabolism. In their study, Jeschke et al. described the link between molecular changes to clinical outcomes and the pathophysiologic response to severe burn injury in 242 children with a mean burn size of 56%. All patients were found to be hypermetabolic with significant muscle protein loss, loss of bone mineral content, and profound alterations of serum proteome. Their cardiac function became compromised, developed insulin resistance in the first week, and were hyperinflammatory with marked changes in interleukin 8 (IL-8), monocyte chemotactic protein 1 (MCP-1), and IL-6.
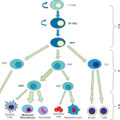
The Inflammation and the Host Response to Injury Large-Scale Collaborative Research Program had the overarching goal of identifying genomic and proteomic changes predicting outcomes in burn and trauma patients. One study looked at the leukocyte transcriptome after severe trauma and burn injury and found a similar genomic storm among different injuries, revealing a fundamental human response to severe inflammatory stress. The transcriptome of leukocytes following burn injury was linked to their immunologic response and related to outcomes. Zimmerman et al. in their study of 167 subjects over 28 days defined greater than twofold transcriptome changes in 80% of leukocyte genes (5136) compared to healthy controls. Within the first 12 hours, the gene expression favors innate immunity and inflammatory response, including NB1 (CD 177), metalloproteinase 8 (neutrophil collagenase), lactotransferrin, and haptoglobin, with a marked downregulation of T-cell function and antigen presentation ( Fig. 24.2 ). They found the genomic response was similar between isolated endotoxin challenge, minor trauma, and severe burn but differing in response time, magnitude, and persistence. These findings suggest that burn patients undergo a persistent inflammatory response and do not have a counterinflammatory response or an adaptive resolution. Key molecules identified in this process were danger-associated molecular pattern ( Fig. 24.3 ). These descriptions of genomic response to burn injury reflect a global response and do not account for complex interplays or subpopulations of different cell types within the total leukocyte populations, the microenvironmental effects within different compartments where the immune cells exert their effects, and proteomic or metabolomic effects. These data highlight the complexity of the immunologic response to severe injury.
A-D, genomic response of leukocytic genes and their association with MOF.
(From Xiao W, Mindrinos MN, Seok J, et al. A genomic storm in critically injured humans. J Exp Med . 2011;208[1]:2581-2590.)
Trajectories over time of immune responses in crtically injured humans.
(From Xiao W, Mindrinos MN, Seok J, et al. A genomic storm in critically injured humans. J Exp Med . 2011;208[1]:2581-2590.)
In addition, the group showed that systemic inflammatory response syndrome (SIRS) is an underlying response that can lead to MOF. While systemic inflammation seems to be involved in MOF, there are many different etiologies that can lead to MOF. Sepsis, for example, is the most common late initiator of SIRS. Of note, a single overwhelming infection is not necessarily required; and small repetitive infections may trigger this cascade. Endotoxins (released by Gram-negative bacteria) are mediators that signal via Toll-like receptor (TLR) pathways (mainly TLR4). In addition, Gram-positive bacteria can cause similar insults. Modern surgical and critical medicine intervention have reduced the incidence of severe infections, but multidrug-resistant organisms remain a source of worsening outcomes.
Intestinal barrier failure has been shown to be a source of endotoxemia and sepsis leading to MOF. Bacterial densities range from near 0 in the stomach to 10 4 to 10 5 in the distal small bowel, to 10 11 to 10 12 /g of stool in the normal colon. Although not seen immediately after trauma, serial insults result in increased translocation of bacteria and their products into the portal and lymphatic circulations. Hemorrhagic shock, endotoxin administration, burns, and burn wound sepsis have each been shown to result in increased translocation of bacteria from the gut. Using polyethylene glycol 3350 as a tracer, increasing burn wound size was demonstrated to increase gut permeability to macromolecules such as endotoxin. Smaller molecules, with lactulose as the tracer, passed more readily through the gastrointestinal membrane after injury. Both intra- and transcellular processes allow transloaction. , Consequences of loss of the gastrointestinal barrier are profound. Translocating whole bacteria can be a direct source of sepsis, can activate Kupffer cells, , and propagate an inflammatory response in conjunction with bacterial products such as endotoxin.
Humoral mediators
In critically ill burn patients, humoral mediators have been shown to be of crucial importance causing MOF, and studies are deciphering these humoral factors using modern molecular techniques.
Sood et al. used early leukocyte mRNA genomics to correlate transcriptome changes with outcomes in 324 severe burn patients. In many ways their mortality findings were as expected. Patients older than age 60 years had a relative risk of death (RR) of 4.53, burns greater than 40% TBSA carried an RR of 4.24, and inhalation carried an RR of 2.08, all independently associated with mortality. They found 39 gene signatures within the leukocyte transcriptome inherent in the genomic storm associated with platelet activation and degranulation, cellular proliferation, and downregulation of proinflammatory cytokines (see Fig. 24.1 ). Jeschke et al. differentiated burn survivors from nonsurvivors based on trajectories in inflammatory and hypermetabolic responses. Nonsurvivors had significantly higher IL-6, IL-8, granulocyte colony-stimulating factor, monocyte chemotactic protein 1, C-reactive protein, glucose, insulin, blood urea nitrogen, creatinine and bilirubin, and hypermetabolic response. IL-8 is a major mediator for inflammation and parallels %TBSA burnt and incidence of MOF. High levels of IL-8 were associated with sepsis, MOF, and mortality suggesting that IL-8 may provide a valid biomarker for monitoring sepsis, infections, and mortality in burn patients.
The humoral inflammatory mediators believed to underlie MOF in burn injury are the same factors and cascades governing the fundamental human responses to severe injury and have been discussed for decades: endotoxin, arachidonic acid (AA) metabolites, cytokines, platelet-activating factor (PAF), activated neutrophils and adherence molecules, nitric oxide, complement, and oxygen-free radicals.
Endotoxin, a lipopolysaccharide component of Gram-negative bacteria in outer cell walls, induces many of the symptoms associated with sepsis: fever, hypotension, release of acute-phase proteins, and production of multiple cytokines, including tumor necrosis factor (TNF) and IL-1 via interaction with TLRs. Endotoxin injection alone causes the same changes in the leukocyte transcriptome as severe burn injury. It also activates complement and the coagulation cascade and results in the release of PAF. Potential sources of endotoxin include Gram-negative bacteria in foci of infections as well as within the gut when the gut barrier fails.
AA makes up approximately 20% of cell membranes and is released from these membranes in response to a multitude of stimuli, which activate phospholipases A 2 and C, and is then metabolized by active mediators. The cyclooxygenase (COX) pathway yields prostaglandins and thromboxanes, whereas the lipoxygenase pathway results in the production of leukotrienes. Prostaglandins and leukotrienes interact with other mediators in a complex fashion and are later degraded. COX products, such as prostacyclin, inhibit platelet aggregation, thrombus formation, and gastric secretion, whereas other products, such as thromboxane A 2 (TXA 2 ), cause platelet aggregation, have profound vasoconstricting effects on both the splanchnic and pulmonary microvasculature, and induce bronchoconstriction and increased membrane permeability. Aspirin irreversibly inhibits COX, driving AA down the lipoxygenase pathway. The lipoxygenase pathway results in the formation of leukotrienes. There are two types based on their metabolism after the action of 5-lipoxygenase: leukotrienes C 4 , D 4 , and E 4 (the sulfidopeptide group) and leukotriene B 4 . Various cell types, including neutrophils, macrophages, and monocytes, can provide the stimuli to generate leukotrienes. In addition, vessel walls are capable of generating leukotrienes. Leukotrienes C 4 , D 4 , and E 4 have variable actions on vascular tone dependent on the presence or absence of other mediators, including COX products. In addition to their variable effects in redirecting blood flow, leukotrienes C 4 , D 4 , and E 4 increase vascular permeability and are elevated immediately prior to the development of pulmonary failure. The principal effect of leukotriene B 4 is enhancement of neutrophil chemotaxis. Thus leukotrienes as a group are involved in the formation of edema and pulmonary and systemic vascular changes seen in MOF.
Cytokines are regulatory proteins secreted by immune cells and have multiple paracrine and endocrine effects. There are six major classes: interleukins, TNF, interferons, colony-stimulating factors, chemotactic factors, and growth factors. Those most extensively characterized in sepsis are IL-1, IL-6, and TNF.
IL-1 and IL-6 are elevated in sepsis, and high levels are associated with fatal outcomes and predict systemic infection. IL-1β causes hypotension and decreased systemic vascular resistance, which may be synergistic with the effects of TNF. TNF causes hypotension, cardiac depression, and pulmonary dysfunction in animals. , When administered to humans, TNF causes fever, hypotension, decreased systemic vascular resistance, increased protein turnover, elevation of stress hormone levels, , and activation of the coagulation cascade.
PAF is a nonprotein phospholipid secreted by many cells, including platelets, endothelial cells, and inflammatory cells, and it is a major mediator of the effects of endotoxin seen in the patient’s pulmonary system. In addition, the effects of PAF on the patient’s hemodynamic state are characterized by vasodilation, cardiac depression, and enhancement of capillary leak. Its complex interactions with other mediators remain poorly understood.
Although tissue injury can occur in the absence of neutrophils, the inflammatory process results in local accumulation of activated inflammatory cells that release various local toxins (e.g., oxygen radicals, proteases, eicosanoids, PAF). When unregulated, such accumulations of activated cells can cause tissue injury. The initial attachment of neutrophils to the vascular endothelium at an inflammatory site is facilitated by the interaction of adherence molecules on the neutrophil and endothelial cell surfaces.
Induced by numerous stimuli, these neutrophil adherence receptors are, intriguingly, reduced after major thermal and nonthermal injury, perhaps explaining in part the increased incidence of infection. The importance of this adherence mechanism can be seen in patients deficient in one integrin class of neutrophil adherence receptors, CD18, who suffer from frequent bacterial infections. The biology of the transmembrane polypeptides governing these complex cell-to-cell interactions is an active area of research and holds promise for therapeutic interventions.
Oxygen radicals, such as hydrogen peroxide and superoxide anion, are released by activated neutrophils in response to a variety of stimuli and when xanthine oxidase is activated after reperfusion in ischemia-reperfusion models. These highly reactive products cause cell membrane dysfunction, increase vascular permeability, and release eicosanoids.
Nitric oxide, released when citrulline is formed from arginine, was identified as an endothelial product in the mid-1980s. Its half-life is merely a few seconds because it is quickly oxidized, but it has profound local microvascular effects. Nitric oxide synthesis is stimulated by various cytokines, endotoxins, thrombin, and injury to the vascular endothelium. It is a potent vasodilator, but its actions vary depending on the vascular bed and presence of other mediators. Nitric oxide is one of the major mediators leading to hypotension in septic shock. ,
Antigen-antibody complexes activate the complement cascade, and complement fragments thus generated interact with other cytokines to propagate the inflammatory response. Diminished levels of the natural inhibitor of C5a have been demonstrated in patients with acute respiratory distress syndrome (ARDS). In animal models of endotoxemia, the administration of anti-C5a antibody has been shown to diminish hypotension.
Organ-specific failure and prevention
MOF reversal is challenging, making prevention paramount. Genomic data have demonstrated that patients with MOF have an initial similar path as those who recover without complications. Prevention is based on the underlying mechanism(s) causing MOF: sepsis, gut barrier breakdown, the wound, and inadequate perfusion ( Table 24.4 ). However, the interplay between organ systems and engines of MOF in the burned patient are complex, making prevention even more challenging.
Table 24.4
Multiple Organ Failure Etiology and Established Preventive Measures
| Etiology | Prevention |
|---|---|
| Sepsis |
Early excision and biologic closure of deep wounds
Anticipation and early treatment of occult septic foci |
| Gut barrier failure |
Optimize whole-body hemodynamics
Early enteral feedings |
| Reduced organ perfusion |
Optimize whole-body hemodynamics
Enhanced oxygen delivery |
Hypermetabolism
Severe injury is associated with the hypermetabolic response, marked by increased oxygen consumption and carbon dioxide production, hyperdynamic circulation, increased minute ventilation, altered immune responses, and catabolism. Persistently higher metabolic rates are seen in nonsurvivors. An essential component of modern burn management is attenuating the hypermetabolic response. Reducing the patient’s metabolic demand via management of the external environment has been an indispensable component of burn care for decades. Presently, β-adrenergic blockade with propranolol is one of the most efficacious pharmacologic interventions. While the effects of growth hormone can be beneficial, its administration was associated with adverse outcomes. Insulin-like growth factor, intensive insulin therapy, and oxandrolone have also been studied.
Anabolic steroids, such as oxandrolone, have been used to reduce postburn catabolism and help shift patients to an anabolic state. Sousse et al. (2015) demonstrated that long-term administration of oxandrolone significantly reduced hypermetabolism and increased height percentile, bone mineralization, lean body mass, and strength. They showed improved pulmonary functions with greater maximum expired ventilation and higher maximum voluntary ventilation. Inhalation injury did not increase burn-induced hypermetabolic stress. Promising investigations continue into the use of anabolic agents to treat severe burn injury, and they are rapidly becoming a standard of care in centers around the world.
Cardiovascular
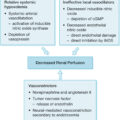
In the immediate 2 to 8 hours following burn injury, a shock state can develop due to decreased preload secondary to the shift of intravascular fluid to the interstitium leading to burn edema. Additionally, in the postburn ebb phase, cardiac function is depressed secondary to IL-1β and TNF-α, which can be blocked with a CD14 knockout or halted with nuclear factor κB blockers. By 48 hours following burn injury, the patient becomes hyperdynamic with observed tachycardia and increased cardiac output through β-adrenergy. The decrease in right ventricular ejection fraction seen after endotoxemia can be alleviated by blockade of thromboxane. Initially, systemic vascular resistance is increased; however, as systemic inflammation and sepsis emerge during the flow phase, vasoplegia ensues and exacerbates the distributive shock state that leads to MOF.
By 24 to 48 hours postinjury, the ebb phase ends, and the patient enters the flow phase, marked by elevated cardiac output. The pathophysiology of this phase of cardiac dysfunction has been thoroughly described. Two primary drivers are prolonged elevated levels of catecholamines and sepsis. Sepsis can continue to produce myocardial dysfunction in a cytokine-mediated process. Persistent β-adrenergic receptor activation causes uncoupling from G proteins, decreases cyclic adenosine monophosphate production and altered phosphorylation down the mitogen-activated protein kinase and Akt (protein kinase B) pathways away from the target of rapamycin (mTOR), and reduces sarcoplasmic reticulum calcium ATPase 2 (SERCA2) and ryanodine receptor (RyR). Collectively, these make the myocardium less responsive to catecholamines and perturb calcium homeostasis. Further, nitric oxide secretion from inflamed and burned tissue alters mitochondrial respiration and competes with calcium for binding sites on myosin, thus furthering calcium dysregulation. The prolonged leakage of calcium from the sarcoplasmic reticulum depletes calcium stores and depresses contractility leading to cardiomyocyte apoptosis.
Additionally, pulmonary hypertension and right ventricular failure occur, which are associated with high mortality. This occurs in burn patients due to sepsis-related myocardial effects or acute fluid overload secondary to overresuscitation and/or edema mobilization. Other common causes have been noted: left heart failure, acute pulmonary embolism, sepsis, acute lung injury, and perioperative state. Previously, Jeschke et al. reported nonsurvivors have a 30% higher cardiac index at 29 to 34 days and 91 to 180 days postburn than survivors, thus demonstrating the increased cardiac demand required in the persistent hyperdynamic state typifying MOF. The data suggest that these patients may need a supraphysiologic cardiac index to meet their hypermetabolic needs, although no definitive studies currently exist. Effective and expeditious management of cardiovascular failure is important to prevent or ameliorate MOF and is a critical component of burn center care.
There is increasing evidence that administration of propranolol can improve cardiac function after burn injury by reducing heart rate and cardiac work, thus dampening hypermetabolism and improving the hypercatabolic state.
Lungs
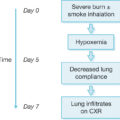
Pulmonary failure can be a result of direct injury from inhalation of toxins, fluid overload, heart failure, and pulmonary edema, resulting from resuscitation, injury from inflammatory and septic mediators, pneumonia, and iatrogenic ventilator injury. Prevention of the pulmonary component of MOF is based on infection prevention measures (early ventilator weaning/daily spontaneous breathing trials, suctioning, oral hygiene, chest physiotherapy, elevating head of bed) and limiting factors that exacerbate pulmonary injury. Current treatments, such as administration of nebulized heparin, albuterol, cortisol, and epinephrine and chest physiotherapy, as well as mucolysis, improve pulmonary ventilation, function, and outcomes. Cox et al. investigated the integrity of airway epithelium in autopsy specimens of 72 severely burned children, finding airway epithelial loss corresponded to inhalation injury and age. Mortality from inhalation injury was 16.4% and was associated with increased length of mechanical ventilation and length of stay.
Lopez and Enkhbaatar both demonstrated that nebulized epinephrine limited pulmonary vascular hyperpermeability to water and protein flux in an ovine burn model with smoke inhalation. This preserved dynamic compliance, mean airway pressure, and PaO 2 /FiO 2 ratio. In their ovine burn with inhalation injury model, Traber et al. administered γ-tocopherol, a reactive oxygen species scavenger. They found decreases in arginase and collagen, significantly improved diffusion capacity, decreased lung water, reduced pulmonary shunt fraction, and reduced peak pressures and bronchiolar obstruction, indicating that free-radical scavengers may reduce smoke-induced chronic pulmonary dysfunction.
In an ovine model of inhalation injury with Pseudomonas infection, there was severe deterioration in pulmonary gas exchange and increased lung lymph flow and protein content, lung water, nitrite/nitrate concentrations, tracheal blood flow, and vascular endothelial growth factor expression. In a different study with this model evaluating microcirculatory changes in response to activated protein C, significant reductions in heart rate and cardiac output were observed, and the changes in microvascular blood flow to the trachea, kidney, and brain were normalized.
For nearly 20 years, the ARDSNet doctrine of limited tidal volumes, pressure-limited ventilation, positive end-expiratory pressure (PEEP), and permissive hypercapnia have dominated the field of pulmonary critical care. Application of these principles to burn care has been limited due to increased CO 2 production resulting from the hyperdynamic state requiring higher minute ventilation, pulmonary edema from resuscitation increasing the alveolar-arterial gradient, and decreasing compliance and airway injury from inhaled toxins causing increased resistance and mucosal sloughing and plugging. For more than 28 years, Sousse et al. (2016) analyzed pulmonary outcomes in 932 burned pediatric patients with inhalation injury, stratifying for tidal volume. Their findings are staggeringly divergent from ARDSNet data; they determined high tidal volume (15 ± 3 mL/kg) corresponded to significantly decreased ventilator days and maximum PEEP and significantly increased maximum peak inspiratory pressure. ARDS was significantly decreased, but the pneumothorax rate was increased. They concluded that high tidal volumes may interrupt the events leading to lung injury following inhalation injury. Of note, this study was performed in the setting of early spontaneous breathing trials and aggressive discontinuation of mechanical ventilation. It remains unclear whether these data are an aberration or, moreover, point to a fundamental difference in the physiology of inhalation injury. In the setting of severe inhalation injury with hypoxia and hypercapnia, we have seen the remarkable ability of pediatric lungs to heal with eventual normalization of gas exchange several months postinjury; this phenomenon may be attributable to a still-active stem cell population.
Systemic factors beyond sepsis also affect pulmonary failure. For example, inflammation associated with burns leads to hyperglycemia. A threshold of greater than 150 mg/dL leads to overwhelming growth of bacteria in the bronchopulmonary system. The incidence of pneumonia requiring mechanical ventilation and ARDS was higher in patients with average daily glucose greater than 150 mg/dL. These patients also had significantly longer ventilation days and higher incidence of infection and sepsis.
Gastrointestinal dysfunction
Denver 2 and SOFA scores only address gut failure pertaining to the liver; however, other aspects of gastrointestinal dysfunction also cause significant morbidity and contribute to mortality. Lautenschlager et al. studied isolated rat small intestine perfused with PAF and demonstrated that intestinal circulatory disturbances, atony, ileus, edema, swelling, and loss of mucosal barrier function are engines of MOF. They induced mesenteric vasoconstriction, translocation of fluid and macromolecules from the vasculature to the lumen, edema, and the loss of motility seen in intestinal failure and observed that quinidine, a sodium channel blocker, inhibited this pathology whereas dexamethasone did not. Oliveira et al. studied the role of COX-2 inhibitors on gastric and small bowel ileus in prostaglandin-mediated etiology for postburn ileus. Gut mucosal integrity suffers when mesenteric flow is inadequate, and gut blood flow is decreased after burn injury, exacerbated by TXA 2 release. Support of splanchnic blood flow is an important aspect of MOF prevention, which can be accomplished as part of whole-body hemodynamic support.
In a chronic porcine model of burn injury, burned pigs were given endotoxin bolus, causing a marked decrease in systemic vascular resistance, blood pressure, cardiac index, and mesenteric blood flow. This state increased gut bacterial translocation into mesenteric lymph nodes, spleen, and burn wounds, possibly due to mesenteric ischemia and reperfusion injury.
Enteral feeding has beneficial effects on outcomes compared with parenteral feeding, possibly via an enhancement of gastrointestinal barrier integrity. Enterocytes are principally supported by intraluminal feeding. Intestinal mucosa deprived of intraluminal feedings develops mucosal atrophy. Early enteral feeding is tolerated in burn patients and attenuates their hypermetabolic response. The value of specific nutrients to support the enterocyte is murkier than that of providing adequate mesenteric blood flow and intraluminal nutrition. Glutamine is the preferred fuel of the small bowel enterocyte. Sepsis has been shown to decrease glutamine uptake by the small bowel enterocyte, which may result in barrier failure, and the addition of glutamine to the nutritional regimen is theorized to improve barrier function. Glutamine is not a component of commercial parenteral nutritional formulas because of its short shelf life, although the dipeptide is well tolerated and has a longer shelf life. While supplemental glutamine may improve protein balance in surgical patients and partially reverse mucosal atrophy, it has not been shown to improve intestinal barrier function when given parenterally. For some time, glutamine was thought to be of benefit for critically burned patients. Heyland et al. conducted a large trial to determine a definitive answer and clearly showed that glutamine is not beneficial for burn patients and should not be given in burns.
The large intestinal mucosa is trophic to butyrate, a fatty acid liberated by fiber fermentation. Enteral pectin may help support the colonic mucosa, but the value of such support in the hypermetabolic burn patient remains unclear.
Limited research suggests possible benefits of probiotics for gut barrier support. Probiotics may improve gastrointestinal barrier function, avoiding colonization with pathogenic microorganisms and immunomodulation. They reduce bacterial translocation in the gut of burned rats. Interestingly, they seem to increase diarrhea rates. Accumulating data suggest probiotics may reduce ventilator-associated pneumonia rates. There are some concerns in the current datasets that the control groups have too high an incidence of ventilator-acquired pneumonia, and there have been case reports of sepsis from probiotic organisms in high-risk patients. In a series of 20 severely burned children, probiotics were found to be safe and tended toward fewer surgeries and less time to achieve complete wound closure.
Decontamination of the gut lumen might diminish the impact of gastrointestinal barrier failure. Attempts have been made to assess the impact of selective decontamination of the gut by coating enteric bacteria to inhibit their ability to attach to the intestinal mucosa and translocate. While the rate of pneumonia may decrease by such mechanisms, there is no apparent impact on mortality.
Acute pancreatitis is also notable after severe burn injury, defined as a threefold increase in amylase or lipase with abdominal pain or feeding intolerance. In a pediatric cohort, Rivero et al. reported an incidence of 13/2699 or 0.05% and found pathologic evidence in 11/78 autopsies. This is in contrast to 40% of patients developing hyperamylasemia or hyperlipidemia without symptoms. They postulated that the etiology of these cases was from ischemic injury to the pancreas due to shock. They also noted increased mortality in this cohort.
In a severe burn injury, the liver plays a pivotal role in modulating inflammatory processes, immune functions, metabolic pathways, and the acute-phase response. Thermal injury causes hepatic damage by inducing hepatic edema, fatty infiltration, apoptosis, and the metabolic derangements associated with insulin resistance. Insulin administration decreases the rate of infections and the synthesis of proinflammatory cytokines and improves hepatic structure and function in severely burned and critically ill patients.
Burn injury promotes catabolism and lipolysis in burned children. The resulting free fatty acids are reesterified in the liver to triglycerides where they are not secreted as very low-density lipoprotein due to downregulation of transport proteins, thus leading to progressive fatty liver metamorphosis. Carbohydrates are utilized as a major energy source for critically burned patients. Lee et al. demonstrated that low-fat, high-carbohydrate feeds promote shorter ICU stays per %TBSA, a lower incidence of sepsis, and decreased hepatic steotosis.
Acute liver failure (ALF) is uncommon after burn injury but carries a high mortality. It presents with hepatic encephalopathy, jaundice, and coagulopathy in the absence of chronic liver disease and carries a mortality rate of 40% to 50%. The cause of death is cerebral herniation in 34%, refractory hypotension, and MOF. Henrionet al. studied 142 consecutive cases of ALF in the ICU and found that 70% were due to heart failure, 13% from respiratory failure, and 13% from toxins or sepsis. Most commonly, low flow states cause ALF. The liver receives most of its blood flow via the portal system, and the dependence on portal supply is increased in critical illness because the arterial supply is reduced while the central lobular oxygen requirements increase due to metabolic and synthetic demands. In the setting of heart failure, high central venous pressures associated with fluid overload, or the need for high preload, there is a decrease in the pressure gradient across the liver, which compromises portal flow (and oxygenation), leading to centrilobular necrosis. Thus most postburn ALF is hypoxemic liver failure, with high hepatic (central) venous pressure a major (unrecognized) cause of reduced hepatic oxygen delivery. Less common causes are toxins and viral infections. In addition, many of the medications used in the burn ICU can cause ALF (e.g., acetaminophen, amoxicillin, moxifloxacin, trimethoprim-sulfamethoxazole, fluconazole, voriconazole, amiodarone, metformin, isoflurane, opiates, phenytoin, ibuprofen, iron, anabolic steroids). Immunosuppression associated with burn injury predisposes the activation of latent hepatitis and viral infections (especially cytomegalovirus), which can lead to ALF. Loss of clotting factors causes coagulopathy, whereas loss of anticoagulants can cause thrombosis, collectively promoting disseminated intravascular coagulation. Burn patients with liver dysfunction become prone to hypoglycemia due to loss of glucose homeostasis. ALF leads to further MOF causing vasoplegia with diminished hepatic clearance of circulating vasoactive compounds, increased nitric oxide production, acute renal failure due to ischemia, and acute tubular necrosis, which may progress to hepatorenal syndromes.
Renal
To survive a burn injury, adequate renal clearance is essential; 24 hours following resuscitation, it becomes necessary to clear the excess volume administered. Renal function is also critical to clear the extensive metabolic wastes associated with injury and the hyperdynamic state. Concurrently, the kidney is subjected to prerenal insults, such as hypovolemia and shock, and to direct renal toxins, such as myoglobin and medications. To maintain renal function, there needs to be vigilant treatment of compartment syndrome to prevent rhabdomyolysis leading to myoglobinemia, adequate monitoring of abdominal compartment pressure to prevent compromise of renal blood flow, and avoidance of nephrotoxic medications. Mason et al. prospectively analyzed 330 resuscitations dividing the patients relative to the Parkland formula resuscitation. The groups receiving greater than Parkland resuscitation had a higher APACHE score. Patients resuscitated with less than Parkland resuscitation demonstrated greater probability of acute kidney injury (AKI) (odds ratio 3.25; 95% confidence interval 1.18–8.94) without difference in infectious complications. Maintenance of sufficient renal blood flow with appropriate volume resuscitation is vital to maintaining renal function. In a retrospective review of 41,179 burned Finnish patients, 86 had AKI related to the burn and 43 required renal replacement therapy. In the absence of diuretics, urine output of 0.5 mL/kg/hour generally indicates sufficient renal perfusion. In patients with increasing creatinine or decreased urine output, the finding of granular casts on centrifuged microscopic urinalysis is helpful in promptly differentiating organ damage (tubular necrosis) from appropriate renal response to decreased blood flow (prerenal state) and can promptly guide therapy.
Prevention of sepsis
Sepsis is a major driver in the formation of MOF, and MOF predisposes patients to sepsis—a lethal feed-forward loop. The postburn surge of proinflammatory mediators and resultant hypermetabolic response and protein wasting contribute to infection and sepsis. In multiple series, all mortalities with MOF were secondary to sepsis. MOF patients may have more major infections (non-MOF 2.1 vs MOF 3.3) and experience more episodes of sepsis (non-MOF 5% vs MOF 31%). Early burn excision and wound closure, regular wound surveillance and culture, and strict infection prevention and control remain principal adjuncts to control invasive infections in burn patients. As sepsis accounts for at least half of MOF cases, it is paramount to prevent wound sepsis and septic insults by early removal of devitalized tissue (manipulation of heavily colonized burn wounds is a frequent source of transient bacteremia). , Multiple episodes of occult bacteremia occurring during frequent manipulation of heavily colonized wounds contribute to the development of MOF by priming immune cells, making them react more intensively to each subsequent insult.
There is a clear benefit in use of perioperative and prophylactic antibiotics in minimizing bacteremia in patients with injuries greater than 60% TBSA and for any patient in whom the probability of bacteremia with wound manipulation is considered to be high. Culture-directed antibiotics are an important consideration because burn patients are prone to a large number of unusual and often occult infections. Rapid diagnosis and treatment are assisted by a high index of clinical suspicion.
Intravascular infections, such as suppurative thrombophlebitis and endocarditis, typically present in burn patients with fever and bacteremia without localizing signs. Burn patients with endocarditis rarely develop a murmur (just 9% of cases), with only 10% reported antemortem. Septic thrombophlebitis often presents without localizing signs, with 68% of cases having only fever and positive blood cultures. Diagnosis is made by thorough examination of all sites of prior cannulation, surgical exposure of suspicious sites, and complete excision of involved veins. Vigilant care, scheduled replacement, and early removal of intravascular devices will minimize the occurrence of catheter-related sepsis. Occult intracompartmental sepsis can also present with fever and bacteremia without localizing signs and is diagnosed only by careful examination and exploration of suspicious compartments.
Pneumonia, seen in approximately 35% of patients with inhalation injury, adds 20% to 60% to the expected mortality. Pneumonia should be vigilantly anticipated and aggressively treated with pulmonary care and systemic antibiotics. The incidence of nosocomial pneumonia increases with longer durations of intubation, emphasizing the importance of judicious use of mechanical ventilation and early extubation protocols utilizing daily spontaneous breathing trials.
Suppurative sinusitis is a rare cause of sepsis in the ICU traditionally described as resulting from transnasal tube placement in a case series published in 1986. Many burn centers routinely use nasotracheal or nasogastric tubes and do not report high rates of sinusitis. Subsequent studies have brought this etiology into question, and Deutschman et al. reported no episodes of sinusitis in 20 years. Furthermore, this is not a typical cause of septic shock even when it occurs. If suppurative sinusitis does occur, it is treated by removal of the tube, topical decongestants, and appropriate antibiotics; it rarely requires surgical drainage.
Acalculous cholecystitis presents with generalized sepsis without localizing signs, making diagnosis difficult. Percutaneous cholecystostomy tube drainage has become the management of choice in suspected cholecystitis in critically ill patients. This allows diagnosis and decompression in patients too unstable for immediate operation.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree