![]() Visit Elsevier eBooks+ (
eBooks.Health.Elsevier.com
) for complete set of videos.
Visit Elsevier eBooks+ (
eBooks.Health.Elsevier.com
) for complete set of videos.
Introduction
Approximately 4000 burn victims die each year from complications related to thermal injury. , Burn deaths generally occur in a bimodal distribution, either immediately after the injury or weeks later due to multisystem organ failure (MOF), a pattern covered in Chapter 24 . Reports had revealed a 50% decline in burn-related deaths and hospital admissions in the United States over the previous 20 years; that trend has flattened. In 1949, Bull and Fisher reported 50% mortality rates for children age 0 to 14 years with burns of 49% of total body surface area (TBSA), 46% TBSA for patients age 15 to 44 years, 27% TBSA for those age 45 to 64 years, and 10% TBSA for those age 65 years and older. These dismal statistics have improved, with the latest studies reporting a 50% mortality for greater than 95% TBSA burns in children 14 years and under, 75% TBSA burns in adults, and around 30% TBSA in the elderly. Therefore, a healthy young patient with almost any size burn should be expected to live, and the prospects for the older demographic are improving with modern wound treatment and critical care techniques.
Burn patients generally die from one of two causes: either early death resulting from burn shock and immolation or MOF leading to late death. With the advent of vigorous fluid resuscitation protocols in the severely burned, irreversible burn shock has been replaced by sepsis and the ensuing MOF as the leading cause of death associated with burns in those who do not die at the scene by a margin of 2 to 1. Those with a risk of mortality who do not die precipitously will be treated by critical care, a service performed in specialized units containing the equipment, supplies, and personnel to institute intensive monitoring and life-sustaining organ support to promote recovery.
Critical illness in burn patients is most commonly beset by sepsis. In a pediatric burn population with massive burns of greater than 80% TBSA, 17.5% of the children developed sepsis, defined as bacteremia with clinical signs of infection. Mortality in the whole group was 33%, most of whom succumbed to MOF. Some were bacteremic and septic, but the majority were not. These findings highlight the observation that the development of severe critical illness and MOF often associate with infection and sepsis and the definition of which has been further refined for the burn injured, but they are by no means required to develop this syndrome. What is requisite is an inflammatory focus, which is the massive skin injury in severe burns requiring inflammation to heal.
It is postulated that the progression of patients to MOF exists in a continuum with the systemic inflammatory response syndrome (SIRS). Nearly all burn patients meet the criteria for SIRS as defined by the consensus conference of the American College of Chest Physicians and the Society of Critical Care Medicine. It is therefore not surprising that severe critical illness and MOF are common in burn patients.
Patients who develop dysfunction of various organs, such as the cardiopulmonary system, renal system, and gastrointestinal system, can be supported to maintain homeostasis until the organs repair themselves or a chronic support system can be established. Critical care may be loosely defined as the process of high-frequency physiologic monitoring coupled with short response times for pharmacologic and procedural interventions. Entire textbooks and many of the preceding chapters in this book are dedicated to critical care. This chapter will focus on synthesizing a critical care system for burn injury, including the organization of specialized burn intensive care units (BICUs) and organ-specific management.
Burn intensive care unit organization
Physical plant
Optimally, a BICU should exist within a designated burn center, ideally verified by the American Burn Association (ABA), and in conjunction with a recognized trauma center, thus providing the capability to treat both thermal and nonthermal injuries. This unit, however, need not be physically located in the same space as that designated for nonburned trauma patients. In fact, the requirement for the care of wounds in burned patients necessitates additional equipment, such as shower tables and overhead warmers, so a separate space dedicated to the severely burned should be standard. This space may be located in a separate hospital with established guidelines for transfer or a specialized unit.
The optimal number of beds in the unit should be calculated by the incidence of moderate to severe burns in the referral area, which in the United States is approximately 20 per 100,000 people per year. The Committee on Trauma of the American College of Surgeons and the ABA recommend that 100 or more patients should be admitted to this facility yearly, with an average daily census of three or more patients to maintain sufficient experience and acceptable access to specialized care.
Most moderate to severe burns with hospital admission will require intensive monitoring for at least the day of admission during the resuscitative phase. Thereafter, approximately 20% will undergo prolonged cardiopulmonary monitoring for inhalation injury, burn shock, cardiopulmonary compromise, renal dysfunction, and the development of SIRS and MOF. In these severely burned patients, the average length of stay in the BICU is approximately 1 day per % TBSA burned. Using an average of 25 days admission for a severely burned patient (20% of the burns, 4/100,000 per capita) and 2 days for those not so severely injured (80%, 16/100,000 per capita), this suggests 132 BICU inpatient days per 100,000 persons in the catchment area. Thus a 10-bed BICU should serve a population of 3 million sufficiently when considered independently. Space provided should be at least 3000 ft 2 , including patient beds and support space for nursing/charting areas, office space, wound care areas, and storage.
Multidrug-resistant bacteria and fungi are commonly encountered in the BICU owing to the presence of open wounds. To prevent transmission of these organisms to other patients, isolation of burn patients from all other patients is recommended and should be considered when designing units for this purpose. Single rooms with negative-pressure ventilation are advisable. In addition, strict guidelines for contact precautions in wound care and interventions as well as handwashing are standard.
Personnel
A BICU functions best by using a team approach among surgeons/intensivists, nurses, laboratory support staff, respiratory therapists, occupational and physical therapists, mental health professionals, prosthetists, dietitians, and pharmacists ( Box 26.1 ). The unit should have a designated medical director, ideally a burn surgeon, to coordinate and supervise personnel, quality management, and resource utilization. The medical director will usually work with other qualified surgical staff to provide sufficient care for the patients. It is recommended that medical directors and each of their oxidation associates be well versed in critical care techniques and that each physician care for at least 50 patients per year to maintain skills. In teaching hospitals, three to four residents or other qualified medical providers should be assigned to the 10-bed unit described. A coverage schedule should be devised to provide 24-hour prompt responses to problems.
Box 26.1
Assigned Burn Unit Personnel
-
■
Experienced burn surgeons (burn unit director and qualified surgeons)
-
■
Dedicated nursing personnel
-
■
Physical and occupational therapists
-
■
Social workers
-
■
Dietitians
-
■
Pharmacists
-
■
Respiratory therapists
-
■
Psychiatrists and clinical psychologists
-
■
Prosthetists
Nursing personnel should consist of a nurse manager with at least 2 years of intensive care and acute burn care experience and 6 months of management responsibilities. The rest of the nursing staff in the BICU should have documented competencies specific to the care of burn patients, including critical care and wound care. Owing to the high intensity of burn intensive care, at least five full-time equivalent nursing providers are required per BICU bed to provide sufficient 24-hour care. Additional personnel are required for respiratory care, occupational and physical therapy, and other support. A dedicated respiratory therapist for the burn unit at all times is optimal.
Owing to the nature of critical illness in burned patients, complications may arise that are best treated by specialists not generally in the field of burn care ( Box 26.2 ). As such, these specialists should be available for consultation when the need arises. Given the regularity with which burn surgeons encounter subspecialty problems, such as corneal injuries, routine injuries are often managed directly by the burn surgeon without additional consultation.
Box 26.2
Consultants for the Burn Intensive Care Unit
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Equipment
The equipment needs of the BICU include those items common to all ICUs, but some of which are specialized ( Box 26.3 ). Each BICU bed must be equipped with monitors to measure heart rate, continuous electrocardiography, noninvasive blood pressure, invasive arterial and venous blood pressures, end-tidal carbon dioxide (EtCO 2 ) monitoring, and right heart cardiac output using dilution techniques or data derived from arterial pressure tracings. Arterial blood oxygen saturation measurement is also required, but continuous mixed venous saturation monitoring or the technical equivalent is optional. Equipment to measure weight and body temperature should be standard. Oxygen availability with at least two vacuum pumps must be present for each bed.
Box 26.3
Equipment for a Fully Equipped Burn Intensive Care Unit
Standard
-
■
Monitors (heart rate, electrocardiography, blood pressure, cardiac output, oxygen saturation, temperature)
-
■
Scales
-
■
Ventilators
-
■
Advanced cardiac life support cardiac cart
-
■
Laboratory support (blood gas analysis, hematology, chemistry, microbiology)
Specialty
-
■
Fiberoptic bronchoscopes
-
■
Fiberoptic gastroscopes/colonoscopes
-
■
Dialysis equipment (peritoneal dialysis and hemodialysis)
-
■
Portable plain radiography
-
■
Computed tomography/fluoroscopy/angiography
-
■
Indirect calorimeters
Ventilator equipment must also be available for all beds. The availability of a number of types of ventilators is optimal, including conventional ventilators with the capability to deliver both volume- and pressure-targeted modes, as well as high-frequency ventilators that are oscillatory and/or percussive in design. An emergency cardiac cart containing advanced cardiac life support medications and a battery-powered electrocardiograph/defibrillator must be present in the unit. Infusion pumps to deliver continuous medications and intravenous/intraarterial fluids must also be readily available. A laboratory providing blood gas analysis, hematology, and blood chemistry should be located onsite. Point-of-care blood analysis for glucose, arterial blood gas, and basic chemistries is strongly advised. , Microbiologic support to complete frequent, routine bacterial and fungal cultures and sensitivities must also be present, as well as virology.
Available specialty equipment should include various sizes of fiberoptic bronchoscopes for the diagnosis and treatment of pulmonary disorders, as well as personnel competent with these techniques. Fiberoptic gastroscopes and colonoscopes for gastrointestinal complications are also necessary for diagnosis, bleeding control, decompression, and difficult feeding access. For renal support, equipment to provide intermittent and/or continuous renal replacement should be present. Portable radiographic equipment for standard chest/abdominal/extremity radiographs must be immediately available. Equipment for computed tomography, fluoroscopy, and angiography should be available. Indirect calorimeters to measure metabolic rate are strongly advised. Overhead warmers and central heating with individualized ambient temperature controls must be available for each room as a specialized requirement.
Hemodynamic monitoring in the burn intensive care unit
Most burn patients follow an anticipated course of recovery, which is monitored in the BICU by measuring physiologic parameters. Experienced clinicians assess these physiologic measures in a repeated and sequential fashion to discern when potential interventions may be initiated to improve outcomes. Often no intervention will be necessary from the unit’s standard care protocol because the patient is following the anticipated course. At other times, this is not the case, and procedural or pharmacologic intervention is beneficial. Physiologic monitoring is then used further to determine the adequacy of the interventions. The following is a survey of monitoring techniques used in the BICU.
Cardiovascular monitoring
Arterial lines
Hemodynamic monitoring is directed at assessing the results of resuscitation and maintaining organ and tissue perfusion. Currently used measures are only estimates of tissue perfusion because the measurement of oxygen and nutrient transfer to cells cannot be made directly at the bedside. Instead, global physiologic measures of central pressures still serve as the principal guides.
Measurement of arterial blood pressure is the mainstay for the assessment of tissue perfusion. In critical illness, this measurement can be made using cuff sphygmomanometers; however, in practice this technique is not useful because the measurement is episodic, and placement of these cuffs on burned extremities is problematic. Diastolic pressures can also be artificially elevated in the elderly and obese. Instead, continuous monitoring for hemodynamic instability through the use of intraarterial catheters is generally preferable when the patient is in the BICU for a prolonged period. Lines are typically placed in either the radial or the femoral artery. The radial artery is the preferred site for critically ill patients because of safety, with the dual arterial supply to the hand as backup should a complication arise. However, it has been shown that radial artery catheters are inaccurate in the measurement of central blood pressure when vasopressors are used and are notoriously inaccurate in children because of greater vascular reactivity. Furthermore, femoral cannulation sites are often unburned due to the insulation provided by undergarments, and they do not preclude mobilization with physical therapy or rehabilitation goals. For these reasons, we recommend femoral arterial blood pressure measurement in most burn patients.
For arterial catheters, systolic, diastolic, and mean arterial pressures (MAPs) should be displayed continuously on the monitor screen. Either systolic or MAP can be used to determine adequacy of pressure, although a MAP greater than 70 mmHg is considered a more accurate descriptor of normal tissue perfusion on the whole. Reasons for this include the finding that, as the arterial pressure wave traverses proximally to distal, the systolic pressure gradually increases while the diastolic pressure decreases; the MAP determined by integrating areas under the curve, however, remains constant. The adequacy of the waveform must also be determined, with a diminished waveform indicative of catheter damping, which requires catheter replacement. Care must be taken to ensure that the diminished waveform is not true hypotension, which can be determined using a manual or cycling sphygmomanometer. Exaggerated waveforms with elevated systolic pressure and additional peaks in the waveform (generally only two are found) may be a phenomenon known as catheter whip, which is the result of excessive movement of the catheter within the artery. Typically, this problem is self-limited, but care must be taken not to interpret normal systolic blood pressure values with evidence of catheter whipping as unexceptional because the effect generally overestimates pressures. Again, use of MAP as the principal guideline for the assessment of blood pressure is optimal, as effects of catheter whip or other problems with intraarterial monitoring are then diminished.
Complications associated with arterial catheters include distal ischemia associated with vasospasm and thromboembolism, catheter infection, and arterial damage/pseudoaneurysm during insertion and removal. Although these complications are uncommon, the results can be devastating. Physical evidence of ischemia in the distal hand or foot should prompt immediate removal of the catheter and elevation of the extremity. If improvement in ischemic symptoms is not seen promptly (within an hour), angiography and intervention must be considered. Should thromboembolism be found, the clot can be removed with operative embolectomy or clot lysis at the discretion of the treating physician. If, during angiography, extensive arterial damage is found with ischemia, operative repair may be indicated. Consideration for anticoagulation must be made while balancing the risk of hemorrhage from open wounds vs. the benefit of tissue salvage.
Evidence of catheter infection hallmarked by purulence and surrounding erythema should instigate removal of the catheter, which often will suffice. With continued evidence of infection, antibiotics and incision and drainage of the site should be entertained. Great caution must be exercised to avoid arterial bleeding if an incision is made over the catheter site. If a pseudoaneurysm is encountered after arterial catheterization and removal without signs of distal ischemia, injection of thrombin or compression with a vascular ultrasound device until no further flow is seen in the pseudoaneurysm will often alleviate the problem without operative intervention.
Cardiac output measurement
Pulmonary artery catheters placed percutaneously through a central vein (internal jugular, subclavian, or femoral) and floated into the pulmonary artery through the right heart have been used extensively in hemodynamic monitoring in BICUs. By measuring the back pressure through the distal catheter tip wedged into an end-pulmonary branch, an estimate of left atrial pressure can be measured. In addition, dyes or isotonic solutions injected into a proximal port can be used to determine cardiac output from the right heart. These data are used to estimate preload delivery to the heart, cardiac contractility, and afterload against which the heart must pump, which then directs therapy at restoration of hemodynamics. These catheters are used in BICUs under conditions of unexplained shock, hypoxemia, renal failure, and monitoring of high-risk patients.
The use of pulmonary artery catheters, however, has come under scrutiny from reports indicating no benefit from their use. A study of 5735 critically ill adults in medical and surgical ICUs showed an increase in mortality and use of resources when pulmonary artery catheters were used. Most of these patients had medical conditions. The authors of this report suggested that their results should prompt a critical evaluation of the use of pulmonary artery catheters under all conditions. This was followed by a clinical trial in the United Kingdom demonstrating no benefit from the use of pulmonary artery catheters in a general ICU setting. A more recent evaluation of the usefulness of these devices has demonstrated that, with proper training and in the appropriate setting, they can provide data not available through other modalities. , Over the past years, use of pulmonary artery catheters has significantly diminished except in special circumstances, such as unexpected response to treatment, as in volume replacement for oliguria. Even in this condition, new technology based on arterial waveform analysis gives an estimate of cardiac output and end-diastolic volume, which generally gives enough information to guide appropriate therapy. However, in the appropriate patients, pulmonary artery catheters may still play a valuable role.
Arterial waveform analysis
Multiple devices have been developed over the past decade using arterial waveform analysis to continuously measure cardiac output as well as to estimate preload. Stroke volume (SV) variation provides a good estimate of the fluid responsiveness of shock with only arterial access. The transpulmonary thermodilution technique provides an even more complete hemodynamic dataset without the use of a pulmonary artery catheter. Using only a central line and central arterial line, thermodilution allows monitoring of preload with global end-diastolic volume index, intrathoracic blood volume, continuous cardiac output, and extravascular lung water index. Numerous studies have shown that these volumetric indices represent preload more precisely than urine output or cardiac filling pressures. In a study involving 54 burned children, Herndon et al. determined pulse index continuous cardiac output to be the superior measurement for cardiac parameters to transthoracic echocardiography and an objective cardiovascular monitor to guide goal-directed fluid resuscitation.
Echocardiography
Transesophageal echocardiography has been used for a number of years as an intraoperative monitor in high-risk cardiovascular patients. It has not been used extensively in other critically ill patients because of the lack of available expertise and paucity of equipment. Since this device can be used as a diagnostic tool for the evaluation of hemodynamic function, it stands to reason that it could be used to monitor critically ill, severely burned patients. A report documented the use of transesophageal Doppler measurements of cardiac output in a series of severely burned patients and showed that intravascular volume and cardiac contractility are significantly diminished the first day after burn in spite of high-volume resuscitation.
Echocardiography has also been studied as a means to supplement urine output monitoring. Investigators in China examined whether esophageal Doppler monitoring of heart function might be an improvement by studying 21 patients with massive burns (79 ± 8% TBSA burned) who were resuscitated with a goal of 1 mL/kg/hour. They found that cardiac output was predictably low after injury and increased linearly with time by increases in preload and contractility and decreased afterload. However, changes in cardiac output were most closely associated with increased cardiac contractility and decreased afterload rather than increases in preload. Additionally, urine output was not closely associated with cardiac output. Held et al. evaluated 11 adult burn patients with a mean TBSA of 37% and found that changes in volume status on echocardiography preceded changes in urine output and vital signs, and they were able to titrate inotropes and vasopressors in elderly patients.
These results call into question the validity of urine output as the primary measure of the adequacy of resuscitation. A similar study by investigators in Sweden probing the role of cardiac function, as measured by echocardiography, and myocyte damage, as measured by troponin abundance in the serum, showed that half of their patients had myocardial damage during resuscitation universally associated with some temporary cardiac wall motion abnormality. However, systolic function was not adversely affected. Bedside echocardiographic equipment and skills are increasingly common in BICUs and are an increasingly common means of hemodynamic monitoring in critical care. However, the intermittent nature of this procedural assessment allows it to only serve as a useful adjunct to add clarity to a difficult clinical scenario and prevents echocardiography from supplanting continuous monitoring modalities, such as thermodilution or waveform analysis. We look forward to further work regarding the optimal method of assessment of resuscitation; for the present, however, urine output remains the standard, and other measures are useful adjuncts.
Laboratory estimates of perfusion
Mixed venous saturation is the gold standard for the measurement of total tissue perfusion but has fallen out of favor because it requires a pulmonary artery catheter. As such, peripheral surrogates, such as base deficit and serum lactate, have become the standard values followed to monitor shock. These can be measured in minutes using point-of-care techniques and rapid guide interventions.
The base deficit is a value calculated using the Henderson-Hasselbalch equation based on the relationship between pH, pCO 2 , and serum bicarbonate:
It is the stoichiometric equivalent of base required to return the pH to 7.4. Base deficit is routinely calculated on blood gas analysis and provides a reasonable estimate of the degree of tissue anoxia and shock at the whole-body level, particularly in hemorrhagic shock. A rising base deficit indicates increasing metabolic acidosis and may stratify risk of mortality in patients after major trauma. The same can be said for the use of base deficit in resuscitation of burn patients. , These studies showed a correlation between higher base deficit and increased mortality, and some have suggested that this value is a better monitor of resuscitation than the time-honored monitors of urine output and arterial blood pressure. Recent studies of burn patients showed the base deficit was higher in nonsurvivors during resuscitation, although the authors could not identify a specific boundary for the effect. , Despite its utility as an indicator of shock, base deficit remains a nonspecific indicator of metabolic acidosis and may be elevated with many confounding conditions other than shock, including hyperchloremia, uremia, and alcohol, cocaine, and methamphetamine use. Interpretation can be difficult under these circumstances.
Lactate is another common measure used to determine the adequacy of tissue perfusion. Under acute low-flow conditions, cells transition from primarily aerobic metabolism to anaerobic metabolism for energy production (i.e., adenosine triphosphate). A byproduct of anaerobic metabolism is lactic acid. Under ischemic conditions, plasma lactate concentration will increase, leading to a decrease in pH. Measurement of lactate is commonly performed to determine the adequacy of generalized perfusion; increases suggest ischemia. Investigators showed that lactate does increase, along with base deficit, in burn patients during resuscitation, and higher levels are associated with poorer outcomes. Later in the course, however, lactate concentrations must be used with some caution because elevated levels do not necessarily indicate ischemia. Under hypermetabolic conditions common in the severely burned, pyruvate dehydrogenase activity is sufficiently inefficient that lactate levels might be elevated without ischemia. Isolated elevations of lactate should then be interpreted with caution, and confirmation of ischemia or shock by physical or other laboratory findings sought. There are, however, recent indications that an early, even minor increase in lactate during the resuscitation period may affect outcome.
Multisystem organ failure
MOF is largely a creation of our success in critical care enabling previously moribund patients to survive long enough for organ failure to develop. Often particular organ systems are allowed to fail to maintain overall patient survival (e.g., performing an excision and grafting procedure that leads to renal failure to remove a septic burn, which would otherwise be lethal). The topic of MOF is more thoroughly covered in Chapter 24 , but we will briefly summarize it here.
Humoral mediators
Humoral inflammatory factors elaborated from the burn wound and the resultant immune, adrenal, and sympathetic activation mediate the development of SIRS.
A number of theories have been developed to explain the progression to MOF ( Box 26.4 ). In the infection theory, as organisms proliferate out of control, endotoxins and exotoxins are released that cause the initiation of a cascade of inflammatory mediators through activation of pathogen-activated molecular pathway (PAMP) receptors, such as Toll-like receptors 2, 4, and 9, as well as the recruitment of inflammatory cells. These pathways can result in organ damage and progression toward MOF if unchecked.
Box 26.4
Theories for the Development of Multiorgan Failure
-
■
Infectious causes
-
■
Macrophage theory
-
■
Microcirculatory hypothesis
-
■
Endothelial–leukocyte interactions
-
■
Gut hypothesis
-
■
Two-hit theory
MOF can also be initiated by inflammation from the presence of necrotic tissue, and open wounds can incite a similar inflammatory mediator response to that seen with endotoxins. Evidence suggests that this response is due to activation of the cytokine cascade through damage-associated molecular pathways (DAMPs), which might be antigens associated with liberated mitochondria from our own cells. Four of these cytokines, tumor necrosis factor α, interleukin 1β (IL-1β), IL-6, and IL-8, are most strongly associated with sepsis and MOF in burns. The primary support of this theory is that many patients, including those burned, can develop MOF without identified infection. Regardless, it is known that a cascade of systemic events is set in motion, either by invasive organisms or from open wounds, that initiates SIRS and may progress to MOF, thus supporting early burn excision and grafting.
Another theory implicates prolonged tissue hypoxia and the subsequent generation of toxic free radicals during reperfusion as the primary mediator of end-organ damage. As discussed in Chapter 7 on burn edema, this free radical damage can be ameliorated with high-dose intravenous vitamin C during resuscitation. From in vitro models and in vivo animal models, we know tissues that were in shock initially and subsequently reperfused produce oxygen free radicals known to damage a number of cellular metabolism processes. It was found that free radical scavengers, such as superoxide dismutase, improve survival in animal models, but these results have not yet been established in humans. Endogenous natural antioxidants, such as vitamins C and E, are low in burn patients, suggesting that therapeutic interventions may be beneficial.
The final two theories revolve around the role of the gut in the generation of organ failure and the two-hit theory of MOF. For years, investigators have implicated the gut as the so-called engine of organ failure, which is associated with loss of gut barrier function and translocation of enteric bacteria and/or their toxic metabolites. Bacterial translocation has been shown to occur after burn in patients. No studies have clearly shown whether bacterial translocation is the cause of SIRS/MOF probably because investigators have as yet been unable to control bacterial translocation effectively during shock in humans, thus a cause-and-effect relationship cannot be established. The two-hit theory ascribes a summation of insults to the development of MOF. Each of the insults alone is inadequate to cause the response, but one or more can prime the inflammatory response system described, such that another normally insignificant injury causes the release of toxic mediators ending in MOF.
It is likely that some part of all these theories is a cause for MOF in burn patients; probably the relative contribution is unique in each patient. Therefore a single solution is unlikely, and this should be kept in mind when devising strategies to improve care and outcomes.
Course of organ failure
Generally, MOF will begin in the renal and/or pulmonary systems and progress in a systematic fashion through the liver, gut, hematologic system, and central nervous system. The development of MOF does not inevitably lead to mortality, however. Efforts to support failed organs until they recover are justified.
Critical care interventions
Critical care of a burn patient in the modern era is predicated upon seven key factors:
-
■
Sufficient goal-directed fluid resuscitation
-
■
Early burn excision and grafting
-
■
Aggressive antimicrobial and source control of sepsis
-
■
Aggressive and sufficient nutritional support
-
■
Active warming
-
■
Aggressive physical, occupational, and respiratory therapy
-
■
Aggressive and continuous support of organ failures
Sufficient fluid resuscitation of an acute burn wound is thoroughly covered in Chapter 8 on fluid resuscitation. Various formulas to predict fluid requirement, balances between crystalloid vs. colloid, and resuscitation end points have been advocated. Early in resuscitation, it is critical to provide sufficient volume to maintain preload and perfusion in the setting of fluid losses into burn edema and distributive shock while avoiding overresuscitation, with the resultant costs such as heart failure, liver failure, and compartment syndromes. ,
Early burn excision and grafting are discussed thoroughly in Chapter 11 on operative management. The overriding principle is to remove inflammatory and diseased burned tissue to break the hyperinflammatory state underlying burn shock. Early grafting reduces the inflammatory load on the patient, fluid loss, heat loss, the area susceptible to infection, and the total length of critical care. Collectively, it reduces the exposure time available for MOF to occur.
Furthermore, the blood loss associated with large-scale early excision often results in a functional plasma exchange. Plasma exchange has been shown to be effective in reducing burn resuscitation ostensibly by removing the inflammatory and oxidative humoral mediators underlying burn shock. Klein et al. reviewed 44 plasma exchanges in patients reaching twice Parkland with albumin or fresh frozen plasma and found a 40% reduction in fluid resuscitation. In a plasma exchange protocol triggered at 1.2 times Parkland, Neff et al. found a 24% increase in MAP, 400% increase in urine output, and 25% reduction in resuscitation rate, with a reduction in lactic acid as well. In 37 patients undergoing plasma exchange with a mean TBSA of 48.6%, hourly fluid, base deficit, lactate, and hematocrit all improved and were associated with decreased resuscitation volume and increased urine output. , Collectively, these data support the notion that plasma exchange improves burn shock; however, there are thus far no studies directly linking plasma exchange resulting from intraoperative blood loss with improvements in burn outcome demonstrated in early burn excision.
C hapter 10 on infection control well-defines the critical nature of early surgical source control and appropriate antimicrobials, but generally emphasizes that meticulous aseptic technique, excision of infected or devitalized tissue, coverage with viable grafts, topical antimicrobials, culture surveillance, and systemic antimicrobials when appropriate are critical components. Similarly, Chapters 22 and 23 on nutrition support and hypermetabolism, respectively, effectively discuss the critical need for enteral feeding and nutritional support, physical therapy, and the requirement to keep patients warm. The remainder of this chapter is directed toward organ-specific critical care support.
Toxicologic burn critical care
Many toxins can affect the burn-injured patient, particularly with occupational injuries. Specific therapies for all toxins are beyond the scope of this chapter and require appropriate decontamination, antidotes, and consultation with material data safety sheets and poison control centers for known exposures if beyond the burn physician’s comfort. However, the most common toxins are cyanide and carbon monoxide. , Cyanide can be elaborated from the combustion of various plastics, and significant exposure can result from smoke inhalation. Mounting evidence indicates that cyanide toxicity is clinically significant in inhalation injuries, being found at clinically significant levels in up to 76% of inhalation injury patients. Few clinical labs return blood cyanide results in a clinically useful time scale, so surrogate markers of less than 15% TBSA with smoke inhalation, Glasgow Coma Scale under 14, abnormal hemodynamics, and/or a lactate level greater than 10 mmol/L are known sensitive indicators of cyanide toxicity of greater than 1 mg/L. In these cases, empiric therapy is recommended with hydroxycobalamin. It is the first-line antidote for cyanide toxicity and has a very mild side effect profile of transient hypertension, bradycardia, and urine discoloration. Hydroxycobalamin is also a nitric oxide scavenger and effectively reduces the hypotension often seen in burn shock. ,
As a common product of combustion, carbon monoxide should be considered in any inhalation injury, enclosed fire, or patient with altered mental status. Patients with carboxyhemoglobin levels above 25% should be mechanically ventilated on 100% fraction of inspired oxygen (F io 2 ), which reduces the half-life from 4 hours to 1 hour. There are rare indications for hyperbaric oxygen (HBO) because it can reduce the carbon monoxide half-life to 15 minutes, particularly in the setting of pregnancy or seizures. HBO is only indicated in the burn setting if it can be immediately employed in conjunction with definitive burn care in specialized centers. For cases where instituting HBO unnecessarily delays burn care, mechanical ventilation can bring carboxyhemoglobin to safe levels prior to HBO’s institution, and appropriate burn care should take precedence. ,
Neurologic burn critical care
The main aspects of neurologic management of burn patients are pain control, sedation, delirium management, and management of acute stress disorder or posttraumatic stress disorder (PTSD). Furthermore, there can be a need for seizure treatment or prophylaxis, management of the traumatic brain injury, or drug and alcohol withdraw protocols, which can occur concomitantly with the burn injury and are beyond the purview of this chapter. An important component of neurologic care is early mobilization; physical/occupational therapy should be performed unless firmly contraindicated.
Pain control is the most common neurologic intervention in the burn patient. Basal pain management with narcotics, such as morphine, is administered as needed to maintain comfort, while taking care to avoid oversedation that can prevent achieving physical therapeutic goals. Methadone can help by providing basal pain coverage and weaning of narcotics. Interventions required for the patient to heal, such as physical therapy or wound care, will create a certain amount of pain that cannot be fully alleviated without preventing the patient’s progress. The burn team must assess the patient’s pain management and use their experience to carefully balance short-term analgesia with long-term recovery and function.
Additional analgesics of short duration should be used for painful procedures, such as extensive wound care or staple removal. Often additional narcotics, such as fentanyl or additional morphine, are sufficient. For more extensive procedures, ketamine is a safe, effective, and recommended agent. Several large series support ketamine use in nurse-driven protocols. In a series of 522 painful procedures in pediatric burn patients, only 2.9% required intervention, such as airway repositioning, zero intubations in lengths from 1 to 105 minutes, and weights from 2 to 111 kg. A meta-analysis of 8282 ketamine sedations from 32 emergency department studies demonstrated no intubations. There was a 0.8% rate of transient apnea and 0.3% rate of laryngospasm; however, all were resolved with positioning and/or bag valve masking. In these studies, trained nurses typically performed the sedation. Ketamine has a favorable safety profile relative to benzodiazepine and narcotics due to its lack of respiratory depression and its cardiovascular stimulating effects. Other alternatives include the use of intravenous lidocaine. Care should be taken to prevent emergence delirium in adult patients, which has a high frequency in critical care in burns, especially among older adult patients.
Sedation in the BICU is more complicated, requiring the balance of short-term sedative goals and apparent patient comfort with the intermediate costs of delirium and decreased therapy participation, as well as with long-term neuropsychiatric costs. The first goal is to minimize sedation administered, which is accomplished best with the use of a sedation scale and avoiding continuous benzodiazepine infusions. Use of the Richmond Agitation-Sedation Scale has been shown to decrease mean duration of ventilator and sedation consumption.
Benzodiazepines have reduced utility due to increased delirium and length of mechanical ventilation. Propofol, ketamine, and remifentanil have been increasingly used as alternatives to the formerly standard benzodiazepine-based therapies. When compared directly to midazolam, patients sedated with the α-adrenergic agonist dexmedetomidine have required less sedation and have less hypotension. This sedative can be considered more effective as well as being less of a risk in terms of hypotension. Benzodiazepines are associated with an increased risk of agitation and delirium, and benzodiazepine and propofol are associated with more ventilator-associated events than dexmedetomidine. Dexmedetomidine is associated with less time to extubation and higher rates of bradycardia, but this is generally well tolerated in burn patients due to their hyperdynamic state and tachycardia. The less-expensive oral equivalent, clonidine, is a common adjuvant to sedation working on a similar α-adrenergic mechanism as dexmedetomidine; it is currently under meta-analysis for that role. Ketamine infusion has been shown to be safe and effective in continuous BICU sedation as well.
Psychiatric and psychological care is a critical component of BICU care. Standard psychological therapies and interventions are important factors of burn care. The critical care team can help prevent delirium and PTSD by avoiding benzodiazepine, minimizing sedation, treating pain first, limiting sleep disturbances, encouraging mobility, reorientating, and avoiding prolonged infusion of sedatives. Despite our best efforts, many patients in the BICU experience delirium and agitation. Haloperidol remains in use in critical agitation and delirium settings and has documented safety in pediatric and adult burn populations. Atypical antipsychotics have gained a substantial role in basal coverage of delirium and agitation and are safe and effective. The BICU team must remember to discontinue these medications when patients are no longer delirious; 84.2% of patients started in the BICU continued use after discharge from BICU, and 28.6% continued following discharge from the hospital.
Cardiovascular burn critical care
Treatment of cardiovascular responses after burn requires an understanding of cardiovascular physiology and the effects of treatment. One of the hallmarks of serious illness is the direct link between cardiac performance and patient performance. Cardiac compensation to burn injury is hyperdynamic to meet hypermetabolic needs, maintain perfusion of injured vascular beds, and compensate for the vasoplegia associated with burn shock. As such, patients often require supraphysiologic cardiac output to compensate for their systemic pathology. The four determinants of cardiac function and hence tissue perfusion are:
-
■
Ventricular preload or end-diastolic muscle fiber length
-
■
Myocardial contractility or strength of the heart muscle
-
■
Ventricular afterload, or the degree of resistance against which the heart must pump
-
■
Heart rate and rhythm
A thorough understanding of the effects of each of these components on heart function is necessary to initiate effective treatments for burned patients with cardiovascular abnormalities.
Preload
Preload is the force that stretches the cardiac muscle prior to contraction. This force is composed of the volume that fills the heart from venous return. Due to the molecular arrangement of actin and myosin in muscle, the more the incoming venous volume stretches the muscle, the further it will contract. This is best demonstrated on a Frank-Starling curve ( Fig. 26.1 ), first described by Otto Frank in a frog heart preparation in 1884; Ernest Starling extended this observation to the mammalian heart in 1914. The relationship demonstrated in the Frank-Starling curve justifies the use of preload augmentation by volume resuscitation to increase cardiac performance. However, when the end-diastolic volume becomes excessive, cardiac function can decrease; probably the muscle fibers overstretch and pull the contractile fibers past each other, thereby reducing the contact required for contractile force. The preload necessary to reduce cardiac function in experimental settings is in excess of 60 mmHg, which is rarely encountered in patients.
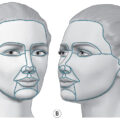
Frank-Starling curve. The solid line depicts the pressure–volume relationship of the heart, showing that as pressure to the heart (preload) increases, the volume pumped by the heart increases. Immediately after burn, contractility diminishes, shifting the curve downward (dashed line) . It must be noted that, with this change, the volume pumped by the heart still increases with increased pressure (preload), validating the use of increased atrial pressure as a means of increasing cardiac output after severe burn.
Preload is estimated clinically by central venous pressure (CVP), pulmonary artery wedge pressure, echocardiography, or transpulmonary thermodilution. These measures can be used to optimize preload, balancing vascular volume loading and cardiac performance against interstitial and pulmonary edema.
Cardiac contractility
The force with which the heart contracts is referred to as cardiac contractility. It is directly related to the number of fibers contracting, their preload, and afterload. Contractility is diminished in patients with low preload or high afterload, in coronary artery disease with loss of myocardium from infarction and ischemia, in burn patients during acute resuscitation due to myocardial depressant factor, in septic shock with Takotsobu cardiomyopathy, or in patients who are severely malnourished. Calculating the left ventricular stroke work (LVSW) from pulmonary artery catheter-derived values provides the best estimate of cardiac contractility and can be determined with the following formula:
where SV is cardiac index divided by heart rate, and PCWP is pulmonary capillary wedge pressure. In current practice, contractility is usually tracked with arterial waveform continuous cardiac output monitoring or echocardiography.
Afterload
Afterload is the force impeding or opposing ventricular contraction and, in conjunction with cardiac output, creates blood pressure. This force is equivalent to the tension developed across the wall of the ventricle during systole. Clinically, afterload is measured by arterial resistance as an estimate of arterial compliance. Arterial resistance is measured as the difference between inflow pressure (mean arterial) and outflow pressure (venous) divided by the flow rate (cardiac output [CO]):
where SVR is systemic vascular resistance. Pulmonary artery catheters, arterial waveform analysis, or echocardiography calculate this value.
Heart rate and rhythm
For the heart to function properly, the electrical conduction system must be intact to provide rhythmic efficient contractions to develop sufficient force to propel blood through the circulatory system. For example, if the heart rate approaches 200 beats/minute, the heart will have insufficient time to fill completely, thereby reducing myocardial fiber stretch and heart function. Also, if frequent premature ventricular contractions are present, the heart will not perform optimally, for similar reasons. Heart rate and rhythm are monitored continuously as routine in every critically ill patient using electrocardiography. A combination of sympathetic and adrenergic tone, combined with high atrial stretch, can predispose burn patients to atrial arrhythmias, such as atrial fibrillation. Judicious fluid and electrolyte management, β-blockade, rate control, and the appropriate use of antiarrhythmics are cornerstones of treatment. An important note is that mortality is higher in BICU patients after developing atrial fibrillation.
Effects of burn on cardiac performance
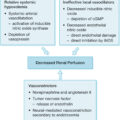
Severe burns affect cardiac performance in a plethora of ways. The first is to reduce preload to the heart through volume loss into burned and nonburned tissues. It is for this reason that volumes predicted by resuscitation formulas must be used to maintain blood pressure and hemodynamics. In addition, severe burn induces myocardial depression characterized by a decrease in tension development and velocities of contraction and relaxation. Cardiac output is then reduced. These effects are most evident early in the course of injury during resuscitation; however, they are followed shortly thereafter by a hyperdynamic phase of increased cardiac output primarily caused by a decrease in afterload through vasodilation and an increase in heart rate.
Hemodynamic therapy: Preload augmentation.
When hypotension or other signs of inadequate cardiac function (i.e., decreased urine output) are encountered, the usual response is to augment preload by increasing intravascular volume. This is a sound physiologic approach based on the Frank-Starling principle and should be the first therapy for any patient in shock. Intravascular volume can be increased using either crystalloids or colloids to increase the CVP and PCWP to a value between 10 and 20 mmHg. Preload goals can also be SV variation below 12% or global end-diastolic volume index between 680 and 800 mL/m 2 . The adequacy of this therapy can be monitored by the restoration of arterial blood pressure, a decrease in tachycardia, and a urine output of greater than 0.5 mL/kg/hour.
Some caution must be exercised when augmenting preload for hemodynamic benefit in burned patients. Excessive volume administration may lead to significant interstitial edema and volume overload, with the development of peripheral and pulmonary edema. These changes can lead to conversion of partial-thickness burns to full-thickness injuries in the periphery and cause significant respiratory problems, liver failure, and compartment syndrome. Once hemodynamics are restored, fluid overload can be treated with spontaneous diuresis, pharmacologic diuresis, dialysis, or even therapeutic phlebotomy if required.
Hemodynamic therapy: Inotropes and vasopressors.
If preload optimization is insufficient to improve hemodynamics, then patients may require inotropes to increase cardiac output and/or vasopressors to increase afterload. Inotrope classes include phosphodiesterase inhibitors, digoxin, and adrenergic agonists. Phosphodiesterase inhibitors, such as milrinone, increase contractility and decrease afterload without increasing myocardial oxygen demand by raising intracellular cyclic adenosine monophosphate (cAMP) levels to increase myocyte calcium (Ca 2+ ) levels. Digoxin increases contractility and decreases heart rate without increasing myocardial oxygen demand by inhibiting the sodium/potassium pump and increasing intracellular Ca 2+ . Dobutamine is a commonly used inotrope with effects limited to β-adrenergic stimulation, thereby increasing cardiac output and causing vasodilation. Dobutamine can be associated with increased heart rate, and it does increase myocardial oxygen demand. The associated vasodilation may be useful in perfusing peripheral vascular beds, as in threatened skin. The data support dobutamine reducing extravascular lung water and decreasing SVR index with increasing cardiac index and urine output, despite the global end-diastolic volume index being unchanged.
Catecholamines, the most commonly used medication to augment blood pressure, are considered inoconstrictors because they have both inotropic and vasoconstrictive properties. One caveat regarding the use of these inoconstrictors is that myocardial oxygen consumption increases, which may affect the ischemic areas of the heart. However, the hypotension being treated by the catecholamines will also compromise myocardial oxygen delivery substantially, so this consideration should not preclude their use by an experienced critical care burn physician. Epinephrine is the catecholamine of choice because it provides a greater proportion of its increase in blood pressure from inotropy rather than vasoconstriction. However, norepinephrine is the preferred pressor in septic shock ; its greater reliance on vasoconstriction vs. inotropy can compromise dermal vascular beds critical to wound healing and survival in a burn patient, particularly in the setting of inadequate preload. Dopamine has generally fallen out of favor due to tachyphylaxis and tachycardias. Therefore we consider epinephrine the preferred catecholamine.
Pure vasoconstrictors have a very limited role in burn care because concomitant inotropic support is advisable due to the patient’s hyperdynamic state. Agents with primary effects on the α-adrenergic receptor can be used to induce vasoconstriction and increase blood pressure. Norepinephrine is often considered in this group, although 40% of its increase in blood pressure is due to α-mediated vasoconstriction, whereas 60% is from increased cardiac output. , Phenylephrine is a pure α-agonist, and can decrease cardiac output and perfusion. , These agents are effective in septic shock or neurogenic shock to increase vascular tone. However, in burn patients, it is believed that these agents will cause vasoconstriction of the skin and splanchnic circulation, thereby redistributing blood flow. This can cause grafts to fail and conversion of partial-thickness skin injuries to full thickness, as well as resulting in ischemic injury to the gut. Additionally, norepinephrine in both physiologic and pharmacologic doses suppressed wound macrophage efficiency in a cAMP-mediated manner via the adrenergic receptor. This may be part of the benefit provided by β-blockers. The physiology of catecholamines is discussed in Chapter 19 on hormonal, adrenal, and sympathetic responses to burn injury.
Another pure vasoconstrictive agent used with great popularity is vasopressin. A very potent vasoconstrictor, it is mediated through its own receptor independent of the adrenergic receptors. Levels of vasopressin have been shown to be low in septic shock, and physiologic replacement at 0.02 to 0.044 U/kg/minute without titration is used in some burn units to increase MAP to good effect. Some investigators found that the use of vasopressin in this setting increased blood pressure, decreased heart rate, and spared norepinephrine dosing when used concomitantly. In these physiologic replacement doses, there is believed to be minimal effect on splanchnic or dermal flow. Although there was a trend toward reduced renal failure with the administration of vasopressin, the VANISH trial failed to conclusively demonstrate reduced renal failure rates in septic patients treated with vasopressin compared to norepinephrine. In an animal model, Li et al. found that adding a small physiologic dose of vasopressin to norepinephrine improved mitochondrial function and all measures of hemodynamics, as well as tissue and splanchnic perfusion. In a series studying 30 septic burn patients treated with vasopressin plus norepinephrine, reduction of total norepinephrine dosing was shown; however, one patient died from upper gastrointestinal necrosis, presenting with increased peripheral ischemia and donor site conversion, as well as skin graft failure. These data again support concerns regarding the use of pure vasoconstrictive agents when treating burn patients.
Very rarely, methylene blue, as a pure vasoconstrictor, may prove beneficial in treating refractory vasoplegia. It is a potent nitric oxide synthase inhibitor and, in case reports, has been shown to successfully reverse refractory vasoplegia following severe burn and thus should be in the armamentarium. ,
Use of vasopressors and inotropes in burn patients is an indispensable component of burn care. Balancing risks and benefit profiles of various medications in an individual patient’s physiology is the purview of an experienced, trained burn critical care surgeon.
Effects of β-blockade on cardiac performance after severe burn
One response to severe burn is a dramatic increase in catecholamine production; this has been linked to a number of metabolic abnormalities, including increased resting energy expenditure, muscle catabolism, and altered thermoregulation. A nonspecific β-blocker, propranolol, has been used to reduce heart rate and myocardial work in severe burns. Propranolol administration also reduces peripheral lipolysis and muscle catabolism, which are additional benefits. Wurzer et al. demonstrated that propranolol reduced cardiogenic stress by reducing cardiac index and MAP in severely burned, injured children without reducing peripheral oxygen delivery or increasing lactic acidosis events or organ dysfunction. Further trials are in progress to define the benefits of β-blockade as well as its interaction with other anabolic medications, such as oxandrolone. Regardless, β-blockade is becoming an increasingly standard component of burn care.
Pulmonary burn critical care
Lungs can be injured from inhalation injury, infection, inflammatory mediators, heart failure, or a sequela of critical care interventions, such as fluid overload or ventilator injury. Minor pathology can be treated with supplemental oxygen, diuresis, bronchodilators, or mucolytics. However, mechanical ventilation is an essential treatment to manage pulmonary failure.
Mechanical ventilation in the severely burned generally occurs for three reasons: airway control during the resuscitative phase, airway management for smoke inhalation, and support during acute respiratory distress syndrome (ARDS). The first indication is for airway control early in the course, with the development of massive whole-body edema associated with the great resuscitative volumes required to maintain euvolemia. In this situation, the need for mechanical ventilation is not due to lung failure per se, but rather to maintain the airway until the whole-body edema is resolved. Once this occurs, usually 2 to 3 days into the course, extubation can be accomplished with minimal sequelae. Ventilator management during this phase is routine. The second indication is airway management early in the course of smoke inhalation, which is a direct toxic injury to the airways and alveoli resulting in mucosal sloughing, loss of mucociliary escalator function, airway narrowing and edema, loss of surfactant, weakening of cartilaginous support of the airways, and fibrinous exudation into the airways. , Chapter 15 on inhalation injury comprehensively covers this issue. The third indication is the development of hypoxemia or hypercarbia due to a high alveolar-arterial (A-a) gradient, shunting, ventilation/perfusion (V/Q) mismatching, poor compliance, or high resistance. Severe burns are known to be associated with hypoxemia and the development of ARDS. The clinical manifestations are dyspnea, severe hypoxemia, and decreased lung compliance, with radiographic evidence of diffuse bilateral pulmonary infiltrates.
Indications for intubation
Intubation entails passing an endotracheal tube (ETT) from either the nose or the mouth through the pharynx and into the trachea. This tube is subsequently connected to a mechanical ventilator to induce inspiration and passive exhalation. In burn patients, indications for intubation are, in general, to improve oxygenation and ventilation or to maintain a compromised airway, such as in a severe inhalation injury or obtunded patient ( Table 26.1 ).
Table 26.1
Clinical Indications for Intubation
| Criteria | Value |
|---|---|
| Pao 2 (mmHg) | <60 |
| Paco 2 (mmHg) | >50 (acutely) |
| P/F ratio | <200 |
| Respiratory rate (breaths/minute) | >40 |
| Respiratory/ventilatory failure | Impending |
| Upper airway edema | Severe |
P/F , Partial-pressure arterial oxygen/fraction of inspired oxygen.
It is important to intubate appropriate patients prior to respiratory arrest. It should be considered, however, that in a large study series more than 33% of burn patients were extubated within 1 day of intubation for transfer to a burn center without reintubation. These patients were subjected to risk without reward, and loss of an ETT in a heavily sedated or paralyzed patient is potentially fatal. ,
Securing an ETT to a burned and/or edematous face can be challenging. In a large survey of burn centers, ETTs were reported secured with linen nonadhesive tape in 59% of cases, manufactured devices in 48%, and orthodontically in 24%. Our center has used nasal intubation with a septal tie for 20 years without accidental extubation or septic sinusitis.
Tracheotomy can provide a long-term durable airway with less patient discomfort. In a survey of US burn centers, the average tracheostomy was performed at 2 weeks; however, there is a consensus that there are indications for earlier tracheostomy. Interestingly, however, in a study of 600 adult medical ICU patients, Terragni et al. found that early tracheotomy did not result in significant improvement in ventilator-associated pneumonia, although the duration of ventilator-assisted respiration was reduced, as was ICU time.
Pulmonary physiology
There has been a proliferation of management strategies for pulmonary failure with an alphabet soup of ventilator modes; however, the physiology of human lungs remains constant regardless of the proliferation of devices. Maintaining a broad physiologic view allows optimal matching of the ventilator’s actions to the patient’s needs. Lungs have three main functions:
-
■
Ventilation
-
■
Oxygenation
-
■
Expectoration
Ventilation.
Ventilation allows the elimination of carbon dioxide (CO 2 ), as measured on arterial blood gas by the Pa co 2 . The ventilator accomplishes this with minute ventilation (V min ) minus dead space ventilation. In general, Pa co 2 varies inversely with V min , so this value must be considered when making ventilator adjustments to alter Pa co 2 . V min is equal to tidal volume multiplied by respiratory rate. Therefore Pa co 2 can be adjusted downward by increasing either tidal volume or respiratory rate. In general, the respiratory rate should be set between 10 and 20 breaths/minute and tidal volume at 6 mL/kg ideal body weight initially. In a normal patient, V min is 100 mL/kg/minute, but with the high CO 2 production seen in burn patients, the needed minute ventilation can increase two- to fourfold. Adjustments can then be made in minute ventilation to optimize Pa co 2 , which is usually 40 mmHg but can be higher in patients with preexisting chronic obstructive pulmonary disease or smoking habits. When making these adjustments, it should be noted that the respiratory rate cannot be increased above 40 breaths/minute in those who are not neonates, and tidal volume should be minimized to avert ventilator-induced lung injury (VILI).
As respiratory rates increase in an attempt to increase minute ventilation while complying with low tidal volumes (LTVs), the fraction of dead space can also increase, further impeding ventilator function. In these cases, decreasing the minute ventilation will increase CO 2 elimination. Dead space V/Q abnormalities can be monitored with volumetric capnography performed bedside to measure physiologic and alveolar dead space. This can be a useful adjunct for ventilator management. Volumetric capnography accounts for total CO 2 exhaled, unlike late expiratory EtCO 2 monitoring, which assumes an A-a gradient of only 2 to 3 mmHg, inappropriate in many critical care patients. EtCO 2 monitoring has utility in monitoring trends or in the setting of a low gradient, as seen in traumatic head injuries. However, in critically ill burn patients, the A-a gradient can be in a state of flux, calling into question values received from the EtCO 2 monitor and making volumetric capnography more appealing. Factors affecting the A-a gradient include cardiac output, airway dead space, airway resistance, and metabolic rate; each of these may change in a severely burned patient, particularly those with inhalation injury. For these reasons, EtCO 2 monitoring is ill advised in burned patients for the estimation of Pa co 2 . Serial blood gas examination is a more reliable monitor.
The ARDSNet studies have well documented that ventilatory injuries to the lung can induce ARDS and have an appreciable effect on morbidity and mortality. Volutrauma occurs when an excess of volume overdistends the lungs, injuring compliant alveoli. Volumes are preferentially directed to compliant and uninjured alveoli because noncompliant alveoli have too long a time constant to accept the volume. As healthy alveoli are sequentially injured, positive feedback is established, and the injury continues to worsen. This is the principle underlying LTV ventilation. Use of positive end-expiratory pressure (PEEP) can facilitate maintaining more compliant alveoli and reduce atelectotrauma by reducing alveolar collapse as well as preserving the lung in a more compliant portion of the pressure volume loop. Furthermore, having more nitrogen in the ventilated gas maintains a stenting function because it is not absorbed out of the airways as is oxygen. When plateau airway pressures are greater than 30 mmHg, the ventilated lung is relatively noncompliant, indicative of ARDS or pulmonary edema, which subjects the lung to barotrauma similarly. In this situation, permissive hypercapnia is a strategy that can be used to reduce barotrauma. This strategy seeks to limit peak and plateau airway pressures by reducing tidal volumes to allow for respiratory acidosis (Pa co 2 >45 mmHg, arterial pH <7.3). This strategy was used to some extent in the trial investigating the efficacy of pressure-limited ventilation on improving outcomes in critically ill ventilated patients.
LTV ventilation has been shown in the ARDSNet trials to be protective against ARDS, but it can also compromise ventilatory function and CO 2 elimination. Burn patients represent unique challenges in mechanical ventilation and ARDS given the reduced compliance due to eschar, chest wall and pulmonary edema due to resuscitation, the unique physiology from inhalation injury, and the increased CO 2 production due to hypermetabolic response. Furthermore, the work of breathing progressively increases at lower tidal volumes. LTV protocols have been proved ineffective in the burn population as 33% of burn patients failed to meet oxygenation and ventilation requirements, increasing to approximately 67% in inhalation patients.
Sousse et al. analyzed pulmonary outcomes in 932 burned pediatric patients with inhalation injury over 28 years, stratifying for tidal volume. Their findings, starkly divergent from ARDSNet predictions, demonstrated high tidal volume (15 ± 3 mL/kg) was associated with significantly decreased ventilator days, maximum PEEP, and significantly increased maximum peak inspiratory pressure; ARDS was significantly decreased, but pneumothorax increased. They concluded that high tidal volumes may interrupt the sequences of events leading to lung injury following inhalation injury.
Another school of thought holds that surrogate markers, such as airway pressures, should be used to avoid ARDS. By this logic, when tidal volumes were lowered, patients with more compliant lungs did poorly, whereas patients with less compliant lungs did well. Regardless, lung-protective strategies are notoriously difficult to institute in the burn population, and some lung injuries often must be accepted to assure overall patient survival.
Oxygenation.
Like the adequacy of ventilation, oxygenation has been classically determined using the Pa o 2 in arterial blood. Arterial oxygenation is calculated using three factors: mean alveolar pressure of oxygen (MAP-O 2 ), the A-a gradient, and V/Q mismatching. MAP-O 2 , as determined by the ventilator, is the area beneath the pressure-time curve multiplied by the F io 2 . Increasing the F io 2 has limited ability to improve oxygenation because in excess of 60% it is considered pulmonary toxic for prolonged time courses. However, increasing MAP-O 2 profoundly improves oxygenation, often quite safely. This can be accomplished by increasing the PEEP or the inspiratory time during which the airways are maintained at inspiratory pressure for longer, thereby enlarging the area under the curve. However, as more time is maintained in inspiration, less is available for exhalation and ventilation. Thus hypercarbia and acidosis can limit the maintenance of high MAP-O 2 . These implications will be discussed in further detail in the ventilator mode section.
The A-a gradient is a function of the diffusion membrane separating air from blood. This is affected by pulmonary edema. A-a gradient issues are best managed by reducing pulmonary edema with diuresis, improving cardiac performance using inotropes, and allowing time for the lungs to heal their diffusion membrane by repopulating type I pneumocytes.
V/Q mismatching occurs as deoxygenated blood is shunted through poorly ventilated lungs and hypoxic vasoconstriction becomes dysregulated in the pulmonary vasculature. This is treated first by improving the aeration of the lung using pulmonary toileting, recruitment maneuvers, and open lung techniques to aerate long time-constant aveoli. Furthermore, inhaled pulmonary vasodilators, such as nitric oxide or prostaglandins, can vasodilate aerated beds and improve V/Q matching. Finally, prone positioning can also improve V/Q matching as well as posterior aeration. In a survey of US burn centers, ARDS was managed through fluid restriction/diureses and enteral nutrition as well as by neuromuscular blockade. In severe ARDS, prone positioning was used in 33% of centers and extracorporeal membrane oxygenation (ECMO) in 18%.
Oxygen is principally delivered to tissues on hemoglobin, so, in general, a Pa o 2 value of 60 mmHg is considered sufficient because it equates to a saturation of approximately 92%. Pulse oximetry effectively measures oxygenation and can be used to guide ventilator management. Falsely measuring methemoglobin and carboxyhemoglobin as oxygen-saturated hemoglobin, which is common initially in patients with smoke inhalation injury, highlights the shortcoming of this technique. Otherwise, this is a very accurate method to determine the oxygen content in arterial blood because 97% of oxygen is carried to the tissues via hemoglobin. This assertion has been corroborated by in vitro studies showing the accuracy of pulse oximetry to within 2% to 3% of oxyhemoglobin levels. The major limitations of this technique lie in its insensitivity to changes in pulmonary gas exchange. Because of the shape of the oxyhemoglobin dissociation curve, when the Sa o 2 exceeds 90% and the Pa o 2 is greater than 60 mmHg, the curve is flat, and changes in Pa o 2 can move considerably with little variation in Sa o 2 . Regardless, it is presumed that an Sa o 2 value greater than 92% is indicative of adequate oxygenation. That the oxygenation saturation measurement is a continuous direct measure immediately available whereas blood gas measurement of Pa o 2 is intermittent is an advantage that should not be overlooked.
A commonly used parameter to assess the adequacy of oxygenation is the ratio of Pa o 2 to F io 2 (P/F ratio). It is an easily calculated surrogate for the A-a gradient. As such, the P/F ratio is one of the criteria utilized to diagnose ARDS in the Berlin definition ( Table 26.2 ). ARDS is defined as bilateral opacities not explained by effusion, collapse of nodules occurring within 1 week of a clinical insult not fully explained by cardiac failure, or fluid overload associated with a P/F ratio less than 300. ARDS is stratified to mild, with a P/F between 200 and 300; moderate, between 100 and 200; and severe, below 100.
Table 26.2
Berlin Definition: Acute Respiratory Distress Syndrome
| Onset within 1 week of onset of injury or illness | |
| Bilateral lung opacities not explained by effusion, atelectasis, or nodules | |
| Respiratory failure without cardiac failure or fluid overload | |
| Diminished oxygenation | |
| Mild | 200 < P/F < 300 |
| Moderate | 100 < P/F < 200 |
| Severe | P/F < 100 |
P/F , Partial-pressure arterial oxygen/fraction of inspired oxygen.
However, not incorporating MAP (mmH 2 O), a key determinant in oxygenation, is a major flaw when utilizing the P/F ratio. Thus two patients on two different levels of ventilator support (one on minimal PEEP, the other on maximal) are indistinguishable based on this parameter, although they are clearly disparate when using the A-a gradient. To account for this variable, the oxygenation index (OI) may be used :
This parameter is particularly helpful in determining a patient’s oxygenation status in relation to the level of ventilator support: The higher the number, the worse the level of oxygenation. Generally, an OI greater than 20% should be a cause for concern.
Expectoration.
The final aspect of pulmonary physiology necessary for consideration in pulmonary critical care is expectoration. The injured lung must clear its secretions, damaged mucosa, pathogens, and aspirated material. In the case of an inhalation injury, there is the transudate of fibrinous material and sloughing of injured mucosa that must be cleared in the setting of a compromised mucociliary escalator. Chest physiotherapy, mucolytics, suctioning, and particular ventilator modes aid in the expectoration of the lungs. ,
A commonly used technique to treat inhalation injury is the combination of nebulized heparin and N-acetylcysteine with albuterol and pulmonary toileting. The goal is that the heparin will prevent coagulation of the transudated plasma, which comes through the injured pulmonary capillaries. The N-acetylcysteine serves as a mucolytic agent and allows expectoration, along with the sloughed mucosa, during physiotherapy and suctioning.
In a meta-analysis of five studies comprising 286 patients, inhaled heparin was found to reduce ventilator days and resulted in more patients alive at day 28 with lower lung injury scores, although methodologic issues were noted. However, in a follow-up analysis of those studies, individual patient data provided no convincing evidence regarding any benefit of heparin nebulization in intubated and ventilated ICU patients, although none of these patients had inhalation burn injuries. In a review of inhalation-injured patients, Kashefi found a nebulized heparin and N-acetylcysteine/albuterol protocol did not reduce mortality or duration of mechanical ventilation but did increase pneumonia rates. Conversely, Sood and Waldroth more recently published a case control study of 72 inhalation-injured patients and found that a 7-day course of nebulized heparin with N-acetylcysteine and albuterol decreased mean ventilator days from 14 to 7 and increased ventilator-free days. There was no change in mortality, pneumonia rates, or bleeding in their study. In this series of inhalation-injured patients, nebulized heparin was found to be safe and effective. This efficacy is consistent with our own clinical experience.
Mechanical ventilation
Mechanical ventilation is an essential component of burn pulmonary critical care. Mechanical ventilatory support allows life to be sustained with lungs that would otherwise inexorably lead to death. It is important that the use of mechanical ventilation provides minimal interruption to burn care, including both wound care and physical therapy. The safety of mobilization of mechanically ventilated patients is well documented, including out-of-bed mobilization of intubated patients ( Fig. 26.2 ).
Respiratory considerations for in-bed and out-of-bed exercises. Green circle indicates low risk for adverse event. Yellow triangle indicates potential risk and consequences of an adverse event but may be outweighed by the potential benefits of mobilization. Red octagon indicates a significant potential risk of an adverse event, and mobilization should only be carried out with specific consideration by senior staff nurse or physical therapist. H FOV , High-frequency oscillation ventilation; PEEP , positive end-expiratory pressure.
(From Hodgson CL, Stiller K, Needham DM, et al. Expert consensus and recommendations on safety criteria for active mobilization of mechanically ventilated critically ill adults. Crit Care. 2014;18[6]:658.)
The principal clinical difference between mechanical ventilation and spontaneous ventilation is the effect of positive pressure, as opposed to normal physiologic negative pressure. The use of positive pressure improves ventilation by recruiting alveoli and increasing functional residual capacity (i.e., the number and volume of open alveoli at the end of expiration), thus improving V/Q mismatch and reducing shunting of blood past nonventilated lung areas ( Fig. 26.3 ). Positive-pressure ventilation (PPV) also allows maintenance of higher MAP-O 2 to overcome high A-a gradients. Adverse effects of PPV lie in its propensity to produce trauma to the airways (barotrauma) and its effects on intrathoracic pressure, which can impede venous return to the heart, thus reducing cardiac output.
A schematic of intrapulmonary shunting. Blood comes into the lung and then divides into capillaries and passes close to alveoli, where gas exchange occurs. In the case of alveolar collapse, the passing blood does not undergo gas exchange and thus returns to the pulmonary vein unchanged (low oxygen concentration, blue color ). This then mixes with oxygenated blood from the aerated alveoli (high oxygen concentration, red color ) to return to the heart and the periphery. In effect, more collapsed lung equates to less oxygenation through this process.
In a survey of US burn centers, pressure-support ventilation (PSV) and volume assist control were the most common starting ventilator modes for all burn patients; however, 53% of centers report using an open lung technique, such as high-frequency percussive ventilation (HFPV), high-frequency oscillation ventilation (HFOV), or airway pressure release ventilation (APRV) in the setting of inhalation injury.
When oxygenation begins to decline, initial maneuvers are to increase the F io 2 to greater than 40%, possibly to 100%. Concentrations of oxygen greater than 60% are considered toxic to airway epithelium over the course of hours; other means to increase oxygenation should be employed. As discussed earlier, treating hypoxia is first accomplished by increasing the MAP-O 2 by raising the level of PEEP incrementally until the desired level of oxygenation is reached while keeping the F io 2 to less than 60%. Once a level of 15 to 20 mmHg of PEEP is reached, other means of increasing MAP-O 2 will need to be employed. These consist of ventilator modes, including inverse ratio ventilation (IRV), HFOV, HFPV, and APRV.
After MAP-O 2 is maximized, therapies are directed at improving V/Q mismatching with inhaled nitric oxide or prone positioning. ECMO has been used with success in burn patients when the lungs are too injured to perform life-sustaining gas exchange. This must be done with caution given the need for anticoagulation and the limitation imposed in the ability to mobilize the patient, both of which can compromise other critical aspects of burn care.
Ventilator modes.
Regardless of the mode ( Fig. 26.4 ), the basic pulmonary physiology principles balancing ventilation, oxygenation, and expectoration against VILI are the purview of a skilled burn surgeon and respiratory therapist.
Different ventilators commonly used in burn units in the United States. A, The most commonly used is the servo type ventilator, which can be set to give volume- or pressure-regulated breaths with or without pressure support or positive end-expiratory pressure. The inspiratory to expiratory ratios can also be reversed. B, The volumetric diffusive respiration ventilator delivers high-frequency percussive ventilation or as continuous positive airway pressure. C, The airway pressure release ventilation (APRV) ventilator can be set to all the servo settings as well as APRV.
Volume-control mode delivers a preset tidal volume regardless of patient effort and is typically used only under general anesthesia due to ventilator–patient dyssynchrony; it takes all respiratory control away from the patient ( Fig. 26.5 ). Assist-control mode differs by allowing the patient to demand additional breaths with a ventilator-preset minimum rate. All breaths are delivered at a prescribed volume, irrespective of any patient attempts for a larger or smaller breath. Some patients find this mode uncomfortable due to the inability to control their volume and require greater sedation than they would under other modes.
Airway pressure curves illustrating the three mechanical functions: IN , initiation of the cycle; LIM , the preset limit (i.e., pressure or volume) imposed on the positive pressure cycle; CYC , the function (i.e., time or volume) ending the cycle. Each mechanical function is governed by one of four physical factors: volume, pressure, flow, and time. A, A time-initiated, volume-limited mode. B, A pressure-initiated (subbaseline pressure produced by the patient’s effort to breathe), volume-limited mode. C, A time-initiated, volume-limited, time-cycled mode that extends inspiration beyond the time that the volume is delivered. A plateau is reached after flow has stopped but before the ventilator cycles into exhalation.
(From Shapiro BA, Lacmak RM, Care RD, et al. Clinical Application of Respiratory Care . St. Louis, MO: Mosby Year Book; 1991.)
Volume-cycled ventilation delivers a set volume of air regardless of pressure required. In the case of poor lung compliance, such as ARDS, this mode could lead to excessive ventilator pressures and significant airway injury. For this reason, time-cycled pressure control ventilation was developed; this delivers inspiration at a given flow rate to a preset pressure. The breath is terminated at a set cycle time, not on the basis of volume of flow, as with volume-controlled ventilation. Therefore pressure control has the advantage of limiting inspiratory pressure despite changes in compliance. It has the disadvantage of a variable tidal volume during dynamic changes in lung compliance, which can lead to inadequate or excessive minute ventilation if compliance respectively worsens or improves.
Intermittent mandatory ventilation (IMV) allows spontaneous ventilation interspersed with volume-cycled or time-cycled pressure control mechanical ventilation. The addition of synchronization, in synchronized IMV (SIMV), avoids placing a mechanical breath on top of a spontaneous patient breath, greatly improving this mode. It was hoped that maintaining some patient work in breathing would preserve respiratory strength while mechanical ventilation was required and that this mode would support weaning to progressively increasing patient effort while reducing mechanical support, although outcome data did not support a role in weaning ( Fig. 26.6 ).