Abstract
Cutaneous lesions may develop as a result of a variety of environmental insults from heat or cold exposure to water immersion and electricity. Injuries to the skin can also be due to exposure to chemicals (including heavy metals) as well as friction and trauma. Dermatoses related to sports activities and musical instruments will be reviewed as well as the cutaneous complications seen with limb amputation.
Keywords
heat injury, cold injury, temperature-related skin diseases, immersion foot, electrical burns, frictional injury, traumatic injuries of the skin, sports-related dermatoses, amputee-related dermatoses, musical instrument-related dermatoses, frost-nip, frostbite
The skin is our primary interface with the environment. Amongst Earth’s creatures, humans are unique in their ability to manipulate the environment by manufacturing clothing, shelter, and heating or cooling devices to allow them to live in a variety of environmental extremes. Despite technologic advances that have allowed humans to live, work and play in most of the environments on this planet, deliberate or inadvertent exposure to those environments may subject individuals to injury. The following discussion will focus upon chemical, frictional, traumatic and water immersion skin injuries, temperature-related skin diseases, electrical burns, cutaneous diseases due to heavy metals, sports-related dermatoses, and skin issues in amputees and instrumental musicians.
Injury Due to Heat Exposure
Heat-Related Illnesses and Thermal Burns
▪ Heat-related illnesses: heat cramps, heat syncope, heat edema, heat exhaustion, heat stroke ▪ Thermal burns: burns
- ▪
Heat-related illnesses occur when the body’s thermoregulatory mechanisms fail; correction of fluid and electrolyte balance is critical in their treatment
- ▪
Management of thermal burns requires assessment of burn depth (i.e. first, second or third degree) and body surface area involved
- ▪
Burn treatment includes prevention of infection, serial excisions, and skin substitutes
Introduction
Humans, like all other mammals and all birds, are homeothermic organisms that maintain their core body temperature within a narrow range by varying cutaneous and visceral blood flow. Normal core body temperatures vary between 36°C and 37.5°C (96.8°F and 99.5°F). At normal core temperature, cutaneous blood flow is about 4% of cardiac output or 250 ml/min. Modulation of cutaneous blood flow is performed by the hypothalamus via a delicate balancing act between heat retention and loss. Cutaneous, muscle, and spinal cord temperature sensors send signals to the preoptic area of the anterior hypothalamus. Communication with the posterior hypothalamus sets up the subconscious thermoregulatory mechanisms of vasomotor control, sweating, and shivering.
When prolonged exposure to a hot environment leads to an increase in core temperature, the subsequent decrease in sympathetic output results in peripheral vasodilation, allowing an increase in cutaneous blood flow of up to 6–8 L/min. Cholinergic output stimulates sweating. These changes allow radiant, conductive, convective, and evaporative heat loss. In contrast, cold temperature exposure leads to increased sympathetic output with peripheral vasoconstriction. Countercurrent heat exchange between parallel arteries and veins further reduces heat loss to the skin, thereby conserving core heat. The behavioral response of shivering generates heat through muscular activity .
Pathologic conditions arise when the normal hypothalamic regulatory set point is overcome by extremes of temperature ( Table 88.1 ). The following discussion will explore heat-related illnesses and thermal burns. Heat-induced urticaria is covered in Chapter 18 .
| HEAT-RELATED ILLNESSES | ||||
|---|---|---|---|---|
| Condition | Core temperature | Clinical features | Treatment | Pathogenesis |
| Heat cramps | Normal | Painful spasm in large muscle groups, weakness, fatigue, N/V, marked sweating , tachycardia, ↑BP | Fluid and salt replacement | Dilutional hyponatremia due to free water (but not electrolytes) being replaced |
| Heat syncope | Normal | Nausea, sighing, yawning, restlessness, fainting (brief) | Avoid standing still in heat, flex legs, lie down if prodrome, cool off, replace fluid | Decreased cerebral blood flow due to volume depletion, peripheral vasodilation, and decreased vasomotor tone in poorly acclimatized or elderly individuals |
| Heat edema | Normal | Dependent edema from vasodilatory pooling | None needed (self-limited) | Vasodilatory pooling |
| Heat exhaustion | Normal to 39°C (102.2°F) | Weakness, dizziness, headache, N/V, irritability, dyspnea/hyperventilation, malaise, myalgias/muscle cramps, tachycardia, orthostatic hypotension, profuse sweating, piloerection , impaired judgment, syncope Hypernatremic form: thirst, rare cramps Hyponatremic form: fatigue, cramps, history of excessive free water intake, altered mental status | Intravenous fluids and electrolytes, sprinkle water and fan or use ice packs, rest, cool environment | Volume and/or salt depletion |
| Heat stroke | >40°C (104°F) | Anhidrosis to profuse sweating , ↓BP, N/V, diarrhea, altered mental status (profound), seizures, coma, cardiac arrhythmias, ↑transaminases, rhabdomyolysis, acute kidney injury, DIC, respiratory alkalosis Poor prognostic indicators: T >42.2°C (108°F); coma >2 h; AST >1000 (day 1) | Rapid lowering of core temperature to 38°C (100.4°F), intravenous hydration, intensive care unit | End result of thermoregulatory failure; volume depletion leads to peripheral vasoconstriction and reduced transfer of heat peripherally; resultant core heating leads to organ failure |
History
It has been speculated that King Edward and his armored crusaders lost the final battle of the Holy Land because the Arab horsemen were adapted and dressed appropriately for the severe heat.
Epidemiology
The frequency of heat-related illnesses is unknown, since milder forms resolve with simple measures. Heat-related illness accounted for more than 8000 deaths in the US between 1979 and 1999, and is responsible for ~7% of wilderness deaths . The European heat wave of 2003 contributed to the deaths of about 14 800 individuals in France. The highest recorded incidence of heat-related deaths during sports participation occurred in the US between 2005 and 2009 . Risk factors are listed in Table 88.2 .
| RISK FACTORS FOR HEAT-RELATED ILLNESS | |
|---|---|
|
|
|
|
|
|
| |
| Medical conditions | |
| |
Burn injuries are far more commonly reported, occurring in 2 million people per year in the US alone, and resulting in 60 000 hospitalizations and 6000 deaths, half of which occur in children . The male to female ratio is 2 : 1. The main sources of burns in children are scald, flame, and electrical. Abuse or neglect may account for up to 20% of pediatric burns. Mortality rates have declined in recent years due to improved resuscitative and surgical management. In the 1940s, burns in children involving 50% body surface area (BSA) had a 50% survival rate, whereas today, 50% of children with 90% BSA burns survive .
A unique form of burn injury is that due to fireworks. In the US alone, there were at least 25 000 emergency department visits for injuries due to fireworks between 2006 and 2010. Half of the visits were by individuals <20 years of age, with a predominance of male patients (~75%). The most common injuries were distal extremity burns, followed by ocular injury, open wounds of distal extremities, and ocular burns .
Pathogenesis
Athletic activity can generate 800 to 1000 kcal/h of heat for prolonged periods. This results in a 1°C rise in core body temperature every 5 to 8 minutes. The normal thermoregulatory mechanisms dissipate this heat as it is generated. When these regulatory mechanisms fail, heat injury occurs. Heat illnesses are not defined by a specific temperature, but by the abnormal response to heat. Table 88.1 outlines the pathogenesis of heat-related disorders.
Thermal burns occur when skin is exposed to infrared radiation (800 to 170 000 nm) from an external heat source with a temperature exceeding 44°C (111°F). Time and temperature curves have been developed to determine maximum threshold thermal exposure times. Necrosis of the epidermis occurs in about 45 minutes at 47°C (117°F) but in only 1 second at 70°C (158°F). Denaturation and coagulation of cellular proteins occur in thermal injury. Interstitial edema develops from altered osmotic pressure and capillary permeability. Several chemical mediators with vasoactive and tissue-destructive properties are released, including prostaglandins, bradykinin, serotonin, histamine, lipid peroxides, and oxygen radicals .
Clinical features
Although heat-related illnesses are not primary skin diseases, they can present with cutaneous findings. Their clinical features are summarized in Table 88.1 .
Thermal burns of the skin differ in their clinical presentation, depending upon the depth of injury ( Table 88.3 ) . Conventional nomenclature categorizes cutaneous burn wounds as first degree (superficial; limited to epidermis), second degree (partial-thickness), and third degree (full-thickness). Second-degree burns are further subdivided into superficial ( Fig. 88.1 ) and deep variants. Burns involving deeper structures such as muscle are sometimes referred to as fourth-degree burns. However, a definitive determination of wound depth may not be possible for the first 24 to 72 hours because of vascular occlusive changes.
| CLINICAL AND PATHOLOGIC FEATURES OF THERMAL BURNS | |||
|---|---|---|---|
| Type | Depth | Clinical features | Pathology |
| First degree | Epidermis only | Pain, tenderness, erythema No blistering Heals without scar | Upper epidermal necrosis |
| Second degree – superficial | Epidermis and superficial dermis | Severe pain, tenderness, serous or hemorrhagic bullae, deep rubor, erosion, and exudation Heals in 10–21 days with mild but variable scarring | More extensive epidermal necrosis with vertical elongation of keratinocytes Necrotic areas may have serous crust with neutrophils, fibrin, and cellular debris Subepidermal bullae possible |
| Second degree – deep | Epidermis and most of dermis destroyed, including deep follicular structures | Intense pain but reduced sensation, deep red to pale and speckled in color Serosanguineous bullae and erosions May appear devitalized initially Prolonged healing time Hypertrophic scars and marked wound contracture | Destruction of entire epidermis, dermal collagen, and most of adnexal structures Collagen bundles may be fused, with eosinophilic appearance Thrombosis of deep vessels common Granulation tissue present at junction of normal and injured tissue |
| Third degree | Full-thickness epidermal and dermal destruction | Dry, hard, charred, non-blanching, insensitive areas of coagulation necrosis Small lesions heal with significant scarring Most require surgical correction | Necrosis of entire epidermis and dermis, with extension into subcutis Inflammatory infiltrate at interface between burned and normal skin If scar forms, it exhibits hyalinized collagen, decreased elastic tissue, loss of arrector pili muscles |
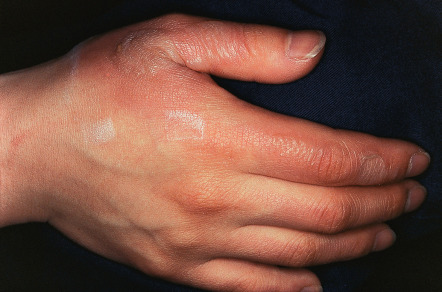
At sites where the dermis is thinner (ears, volar forearms, medial thighs, perineum), burns may be deeper than the initial presentation might suggest; this is also the case in the comparatively thinner skin of children and the elderly. Wounds that appear dull red from entrapment of clot (denatured hemoglobin) imply full-thickness injury .
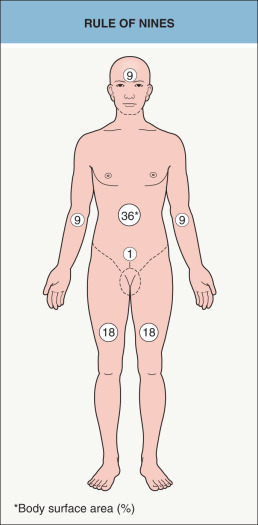
The severity of burn injuries is based upon depth and BSA involvement. BSA is estimated in adults by the “rule of nines” ( Fig. 88.2 ). This formula cannot be applied to children, since the head accounts for ~19% BSA in a 2-year-old, 15% in a 7-year-old, and 13% in a 12-year-old. Lund & Browder charts are useful for more accurate assessment of BSA involvement . Initial evaluation should address airway and circulatory status; associated inhalation injury is seen in up to 25% of burn patients. Cardiovascular evaluation must address the issue of hypovolemic shock, regardless of burn severity. Urine output must be monitored because of the significant fluid loss and rhabdomyolysis that may compromise renal function .
Pathology
Pathologic features of thermal burns are summarized in Table 88.3 .
Differential diagnosis
The differential diagnosis of heat injury is listed in Table 88.1 . Although there is considerable overlap in clinical features, an important distinction is the degree of neurologic compromise. The most important diagnostic issue with thermal burns is the depth of the injury (see Table 88.3 ).
Treatment
Management of heat-related injuries involves removal from the hot environment, rest, rehydration, restoration of electrolyte balance, and evaluation of involved systems ( Table 88.1 ; see Atha for more details).
Initial management of burn victims includes assessment of cardiopulmonary status as well as the extent and depth of the burn. Information regarding resuscitation of burn victims is beyond the scope of this text, but can be found in references , and . For first- and second-degree burns, the burn wound itself should initially be rinsed with cold running water for 20 minutes in order to ease pain, reduce heat, and reduce burn depth . Then the wound should be gently cleansed to remove any foreign material. The next step is prevention of infection, followed by creation of a proper healing environment. Topical antimicrobial agents shown to be effective in burn wound care include silver sulfadiazine, mafenide acetate, and silver nitrate. Silver sulfadiazine has gained wide acceptance for both pediatric and adult burn treatment, but it is known to be cytotoxic and percutaneous absorption can lead to leukopenia. Silver sulfadiazine also produces a pseudoeschar that may interfere with burn depth assessment and prolonged application may lead to localized argyria. Some experts therefore prefer mafenide acetate.
While superficial wounds may require minimal additional therapy , deeper burn wounds need more aggressive therapy, with the standard approach being serial excision and autografting (if there are sufficient donor sites). Third-degree burns are excised early, with indeterminate and deep second-degree wounds delayed until maximum depth and extent are known. Biologic dressings (e.g. pig skin, human allograft) were popular for several years, but have been largely displaced due to higher infection risk and poorer healing. Skin substitutes such as acellular human dermal matrix (Alloderm ® ), bilaminar bovine collagen–shark chondroitin sulfate (Integra ® ), and cultured epithelial autografts are increasingly being utilized (see Ch. 145 ).
Erythema Ab Igne
▪ Toasted skin syndrome ▪ Fire stains
- ▪
Localized areas of reticulated erythema and hyperpigmentation
- ▪
Chronic exposure to heat below the threshold for a thermal burn
- ▪
Commonly in lumbosacral region (heat applied to relieve pain) and shins (in regions without central heating in homes)
- ▪
Squamous atypia histologically (in later lesions)
- ▪
Risk of cutaneous malignancy, particularly squamous cell carcinoma
Introduction
Chronic exposure to low levels of infrared heat can lead to the characteristic reticulated cutaneous pattern of erythema ab igne (EAI), with subsequent risk of malignant degeneration.
History
EAI, once a very common entity, was first described in the UK as a result of standing near stoves fired with peat. In the southern US, the custom of sitting around pot-bellied stoves to keep warm while socializing also led to its development. Although the advent of central heating in much of the industrialized world has led to a dramatic decline in incidence, creative cultural and therapeutic applications of various heating devices continue to induce EAI .
Epidemiology
In the past, EAI was seen up to 10 times more frequently in women than in men. The vast majority of those affected were middle-aged or older. Risk factors include lack of central heating, occupations with close heat exposure, and medical conditions with symptoms relieved by heating or associated with decreased sensation. More recently, reports of EAI due to laptop computers have appeared, including in teens engaged in prolonged gaming .
Pathogenesis
Long-term exposure to heat below the threshold for thermal burn is the primary etiologic factor in EAI. The precise pathophysiology is unknown, but the clinical pattern corresponds with the dermal venous plexus. Repeated exposure to heat below 45°C (113°F) produces reticulated erythema followed by hyperpigmentation in the same pattern . Heat sources reported to cause EAI are found in Table 88.4 .
| HEAT SOURCES REPORTED TO CAUSE ERYTHEMA AB IGNE | |
|---|---|
|
|
Clinical features
The initial presentation is transient macular erythema in a broad reticulated pattern that easily blanches. The overall size and shape often approximates that of the heat source. With recurrent heat exposure, the erythema evolves into a dusky hyperpigmentation; lesions become fixed and are no longer blanchable ( Fig. 88.3 ). Epidermal atrophy may overlie the reticulated pigmentation. Later-stage lesions can become somewhat keratotic and bullae may appear. Lesions are characteristically asymptomatic, although a slight burning sensation is sometimes noted .
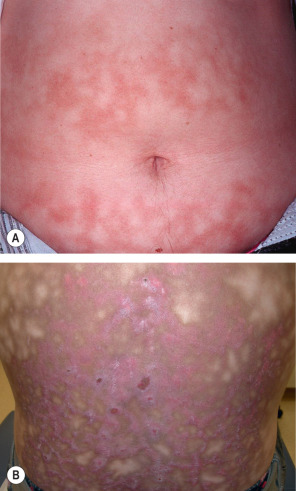
Once the heat source is identified, it is important to determine if it is being used to relieve pain and, if so, the cause of the pain. A lumbosacral location usually points to musculoskeletal disease or, less often, bone metastases. EAI of the abdomen, flank, or mid back may reflect an attempt to relieve pain from inflammation (e.g. pancreatitis, peptic ulcer disease) or malignancy (e.g. pancreatic, gastric). Symptoms of pain should prompt a thorough review of systems and consideration of a search for occult disease. An inquiry into occupation and hobbies is also important, as EAI can develop in exposed areas, e.g. the forearms of bakers, the face or arms of glass blowers and foundry workers, and the anterior thighs or abdomen in those with prolonged use of laptop computers .
The possible development of cutaneous squamous cell carcinoma (SCC) or Merkel cell carcinoma represents the major long-term risk. The latent period may be 30 years or more. Apparently, the risk of developing SCC is highest with hydrocarbon-fueled heat exposure, e.g. “peat fire cancers” on the shins of women, Japanese “Kairo cancers” and Tibetan “Kangri ulcers” due to coal-fired clothing warmers, and Chinese “Kang cancers” from sleeping on coal fire-heated bricks .
Pathology
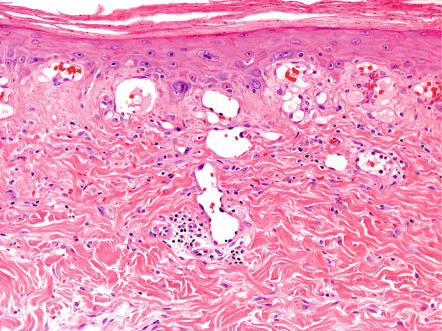
The earliest histopathologic changes include epidermal atrophy, vasodilation, and dermal pigmentation (both melanin and hemosiderin) ( Fig. 88.4 ). As lesions progress, the epidermal atrophy becomes more pronounced, with flattening of the rete ridges. Focal hyperkeratosis and dyskeratosis are noted along with squamous atypia. Basal cell vacuolization has been reported and the dermal–epidermal junction may exhibit interface dermatitis. The dermis appears thinned and sometimes edematous, with accumulation of abnormal elastic tissue and pigment as well as ectatic capillaries. Dermal capillaries may exhibit enlarged endothelial cells with hyperchromatic nuclei. Later lesions may also show basophilic degeneration of connective tissue. Hemosiderin deposition and prominent telangiectasias are seen most often in leg lesions. Ultrastructural evaluation also shows increased melanocyte dendritic processes, suggesting melanocyte activation .
Differential diagnosis
EAI needs to be distinguished from livedo reticularis, which is a temperature-sensitive vasculopathy that favors the extremities but only occasionally develops associated hyperpigmentation (see Ch. 106 ). Cutis marmorata and cutis marmorata telangiectatica congenita are less commonly considered in the differential diagnosis of EAI. The presence of telangiectasias along with atrophy and hyperpigmentation raises the possibility of poikiloderma and its various causes, e.g. cutaneous T-cell lymphoma (see Ch. 120 ), dermatomyositis (see Ch. 42 ), several genodermatoses (see Ch. 63 ).
Treatment
Management consists primarily of removal of the offending heat source. The epidermal atypia is comparable to that of actinic keratoses, and anecdotally it has been treated successfully with topical 5-fluorouracil. As mentioned above, the use of heat to treat symptoms of pain should prompt an evaluation to determine its etiology .
Burns Associated With Fluoroscopy and Magnetic Resonance Imaging (MRI)
- ▪
MRI may produce first-, second-, or third-degree burns due to metal or wire contact with skin, creating a closed-loop conduction system
- ▪
Fluoroscopy, particularly when repeatedly performed in patients with cardiovascular disease, may result in radiation-induced injury
Introduction
The radiologic literature is replete with reports of superficial to full-thickness thermal or electrical burns occurring during MRI. Risk factors for MRI burns are listed in Table 88.5 . In addition, there are reports in the radiologic and dermatologic literature of ulcerations on the back following repeated fluoroscopy .
| RISK FACTORS FOR BURNS DURING MAGNETIC RESONANCE IMAGING | |
|---|---|
|
|
Pathogenesis
Although the precise cause of MRI burns is unknown, the presence of both pulsed radiofrequency and pulsed magnetic gradient fields is thought to play a role. If an electrically conductive loop is formed between the patient and an electrode, patient and wiring, or even small areas of skin–skin contact, current can be produced by the changing magnetic flux, causing a thermal or electrical burn. Resistance at the skin surface may create a thermal injury, while contact with wiring in a capacitative coupling may create electrical injury. Alternatively, the burn may result from formation of a resonant conducting loop with an extended wire creating a resonant antenna, heating maximally at the antenna tip .
The mechanism behind fluoroscopy-induced radiodermatitis is related to the dose of radiation that is delivered. Repeated procedures are notorious for increasing the risk . Patients with diabetes mellitus, obesity, autoimmune connective tissue diseases (e.g. systemic sclerosis) or genetic defects involving repair of radiation-induced injury, as well as individuals receiving chemotherapy, appear to be more prone to developing this complication at lower doses of radiation .
Clinical features
Thermal burns appear soon after MRI, and may be partial- to full-thickness (see Table 88.3 ). The shape and size of the burns are determined by the conductor causing the injury (e.g. circular burns under ECG electrodes; figurate burns conforming to the shape of metallic wires or tattoo pigment deposition) .
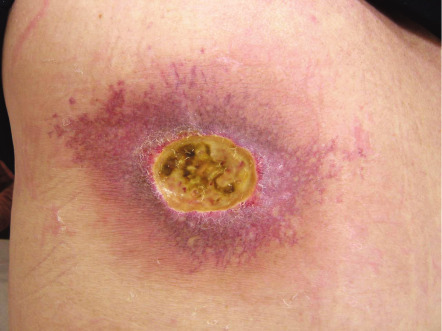
Fluoroscopy-induced radiation dermatitis may be acute, but, as time passes and the acute injury resolves, the patient may continue to develop further changes, including epilation of hair, desquamation, permanent erythema, and eventual ulceration ( Fig. 88.5 ) . The left upper back is a characteristic location for patients with cardiovascular disease who have had cardiac catheterizations with attempts at revascularization (e.g. angioplasty, stent placement) .
Differential diagnosis
Because of the proximity of the MRI event to the detection of a burn injury, the diagnosis is generally straightforward. However, in the case of a tattoo burn, allergic contact dermatitis or foreign body granuloma formation within the tattoo may be considered. Timing of the event plus the sensation of a burn rather than pruritus should allow a distinction.
The differential diagnosis of fluoroscopy-induced disease includes fixed drug eruptions, contact dermatitis, herpes virus infection, impetigo, and spider bites.
Treatment
Prevention of this injury by avoiding risk factors is ideal. However, when burns occur, they should be managed as any other thermal burn, or, in the case of fluoroscopy-induced injury, by limiting the dose delivered.
Airbag Burns
- ▪
Deployment of airbags causes numerous cutaneous injuries, including frictional and blunt trauma, thermal burns, and possibly chemical burns
- ▪
Friction due to contact with the bag, thermal burning from the bag and its released gases, and exposure to corrosive aerosol produce the injuries
Introduction
Automobile airbags provide additional crash protection, reducing driver and passenger fatalities by up to 30%. Although patented in 1953, US manufacturers only began installation of airbags in the mid-1970s. European airbag installation began in 1981. The US Federal Motor Vehicle Safety Standard 208 was amended in 1984, requiring that all vehicles made after 1989 have passive restraints. Further revisions in 1998 made dual front seat airbags mandatory. Despite the beneficial effect, numerous injuries have resulted from airbag deployment, including burns, abrasions, and lacerations .
Pathogenesis
Airbag activation consists of detection, inflation, and deflation phases. The sudden deceleration caused by impact triggers sensors in the front bumper to fire a sodium azide canister inside the nylon rubber bag. The sodium azide ignites with an explosive release of hot nitrogen gas which inflates the bag in about 10 milliseconds at speeds exceeding 160 km/h. The hot nitrogen gas and other by-products are then vented out the upper sides of the bag away from the occupant, thus deflating the bag. Vented aerosol contains nitrogen, carbon mono- and dioxides, sodium hydroxide, ammonia, nitric oxide, metallic oxides and other trace gases, creating a corrosive environment. Friction and pressure from the bag, heat from the bag and gases, and contact with the corrosive aerosol may produce injury .
Clinical features
The cutaneous injuries produced by airbag deployment are summarized in Table 88.6 . Chemical burns occur rarely, but appear to be a result of skin exposure to small amounts of the corrosive alkaline aerosol. The aerosolized particles need to dissolve in a water layer such as tears or sweat before creating a corrosive injury. Of note, alkali burns of the skin may continue to deepen and extend over time, unless all the corrosive chemical is washed away .
| AIRBAG INJURIES TO SKIN | ||
|---|---|---|
| Type | Frequency | Clinical features |
| Abrasions/friction burns | 65% | Patches of erythema or ecchymosis, typically with superficial erosion |
| Contusion | 40% | Ecchymosis |
| Lacerations | 20% | Variable severity |
| Thermal burns | 8% | First- and second-degree most common (see Table 88.3 ) |
| Localized full-thickness burn may result from metallic accessories or melted clothing | ||
| Face and forearms, followed by hands, chest | ||
| Chemical burns | ? | Painful, superficial, violaceous erythema and edema |
| Partial- to full-thickness, often in splash pattern | ||
| Irritant dermatitis | ? | Erythema and edema of upper chest, arms, face |
| Burning or stinging sensation | ||
| Desquamation and postinflammatory hyperpigmentation common | ||
Differential diagnosis
The pattern of injury and history of airbag deployment makes the diagnosis obvious. However, it is important to determine whether a burn injury is purely thermal or if it has a chemical component. Assessment of tissue pH and monitoring for continued extension of the burn help determine whether an alkali injury exists.
Treatment
Prevention is, of course, the most effective approach. Newer airbag designs include repositioning the exhaust vents in order to direct the escape of hot gases away from areas of potential skin contact. Seats should be positioned as far back from the airbag module as possible. Children too small to benefit from the adult three-point restraint should not be in the front passenger area. Children in any type of car seat or booster should never be put in the front seat. When a thermal or frictional injury occurs, local wound care and protection from secondary infection usually suffice.
Alkali burns require copious irrigation to dilute the corrosive substance. Acidic neutralization is contraindicated because of possible extension of injury by the resulting exothermic reaction. When the tissue pH is normal, local wound care is adequate.
Injury Due to Cold Exposure
Frostbite
▪ First-degree frostbite – frost-nip
- ▪
Frostbite can occur when the skin temperature drops below about −2°C (28°F)
- ▪
Erythema, edema, and numbness are followed by marked hyperemia and pain
- ▪
Tissue freezing, vasoconstriction, and inflammatory mediator release are central to the pathophysiology of frostbite
- ▪
Rapid rewarming in a warm water bath is the cornerstone of therapy
Introduction
Exposure to cold environments, whether during occupational or recreational activities, poses a risk of cold-related injury. Prolonged exposure may lead to hypothermia, frost-nip, or frostbite. Hypothermia, i.e. a decline in core temperature below 35°C (95°F), is a systemic process with minor cutaneous features and therefore will not be discussed in detail.
History
Cold injury predates recorded history – the quest for clothing, shelter, and fire as protection from the elements must have preoccupied prehistoric humans. Mass casualties from cold injury have historically been seen most often in military conflicts. However, as interest in outdoor activities and the number of homeless individuals have risen, there has been increased exposure to cold among the general population .
Epidemiology
A 10-year retrospective assessment (1986 to 1995) of cold injury in members of the British Antarctic Survey found an incidence of 65.6 per 1000 per year seeking medical attention. Ninety-five percent had frostbite, 3% hypothermia, and 2% cold water immersion foot. Superficial frostbite was the most common injury (74% of cases), with the face (nose) and ears being the most frequent sites of involvement. Seventy-eight percent of the victims were injured during recreational activities. Although temperature and wind chill had no influence on severity of injury, the prevalence of cold injury increased with falling temperature, with a maximum prevalence at −25°C to −30°C (−13°F to −22°F). A major risk factor was prior cold injury .
In addition to homelessness and previous cold injury, risk factors for frostbite include altered mental status, alcohol or illicit drug use, old age, circulatory impairment, smoking, dehydration, vehicular trauma, and high altitude .
Pathogenesis
The underlying pathophysiology of frostbite is a combination of freezing, vascular insufficiency (constriction and occlusion), and damage due to inflammatory mediators. As extremities cool, the “hunting response of Lewis” (alternating vasoconstriction and vasodilation) occurs, ending with vasoconstriction. This tends to conserve the core temperature at the expense of the extremity, which shifts toward ambient temperature. The head does not exhibit a vasoconstrictor response, except for the nose and ears. The trunk surface may cool slightly, but the core temperature is maintained. Frostbite is therefore unusual on the trunk or head (except the nose and ears), unless there is direct contact with ice or a refrigerant. The pathophysiologic stages of frostbite are summarized in Table 88.7 , along with key clinical features .
| FOUR STAGES OF FROSTBITE | ||
|---|---|---|
| Phase | Physiologic events | Clinical findings |
| I (cooling) |
| Cool extremity Blanched appearance Numbness Hard, woody texture |
| II (thawing and rewarming) |
| Erythematous to violaceous color Intense pain Blisters form |
| III (extension of injury) |
| Blisters rupture |
| IV (resolution) |
| Healing or amputation |
Clinical features
Frostbite has been divided into four categories of severity, analogous to burn injuries. These are only recognizable upon rewarming. Frost-nip (first-degree frostbite) presents with erythema, edema, cutaneous anesthesia, and transient pain ( Fig. 88.6A ). Full recovery is expected, with only mild desquamation. Second-degree frostbite is characterized by marked hyperemia, edema and blistering, with clear fluid in the bullae ( Fig. 88.6B ). Healing occurs, but many patients have long-term sensory neuropathy, often with significant cold sensitivity.
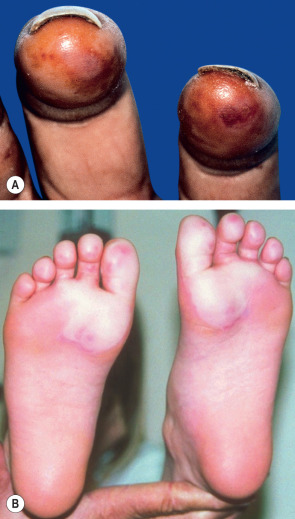
Third-degree frostbite consists of full-thickness dermal loss, with hemorrhagic bulla formation or development of waxy, dry, mummified skin. The latter features are poor prognostic indicators for tissue loss. Fourth-degree frostbite is a full-thickness loss of the entire part, with skin, muscle, tendon and bone damage. Injuries of this severity lead to amputation . Some authors advocate a simpler classification consisting of superficial or deep injury, citing better prognostic accuracy.
Pathology
Superficial dermal edema and subepidermal bulla formation are typical findings in frostbite. Vascular permeability leads to hemorrhage that is most pronounced in deep injury. The epidermis and involved dermis become necrotic, with an inflammatory infiltrate and granulation tissue at the junction with normal tissue .
Differential diagnosis
Frostbite injuries should be readily recognizable, given an exposure history and typical presentation. A greater diagnostic challenge is determining the depth of the injury (see above). One possible diagnosis in the differential is cold water immersion foot (see Table 88.9 ).
Treatment
Fundamental therapeutic goals in frostbite are rapid rewarming, prevention of further cold exposure, and restoration of circulation. Once the patient is in a setting in which re-freezing cannot occur, rapid water bath rewarming is indicated. The water bath temperature should be about 37–39°C (99–102°F). Rubbing the frozen extremity with ice, using dry heat, and slow rewarming are all contraindicated. In the hypothermic patient with frostbite injury, it is important to complete fluid resuscitation and core rewarming before limb rewarming, to prevent sudden hypotension and shock. Routine wound care, protection of the frostbitten part, and tetanus prophylaxis should then follow. Radiologic evaluation (MRI, bone scan, plain films) may help determine extent of injury and prognosis. Nowadays, triple-phase bone scans are considered the standard of care for assessing tissue viability during the initial days following injury .
Additional therapeutic suggestions have been based on animal studies, case reports, or small series. Because of increased blood viscosity and sludging, thrombolytic therapy (e.g. heparin, tissue plasminogen activator, streptokinase) ± a vasodilator (e.g. iloprost, limaprost) has been suggested. Superficial white (non-hemorrhagic) bullae may be debrided to avoid prolonged exposure to prostaglandins and thromboxanes in blister fluid. Aloe vera, a thromboxane inhibitor, has been shown to be useful as a topical agent in superficial frostbite. The problem of vasoconstriction has been addressed by sympathectomy, with the most impressive effect being prevention of re-injury with repeat exposure to cold conditions. Pentoxifylline has been found to be useful, as have anti-inflammatory agents such as methylprednisolone, methimazole, and aspirin .
Pernio
▪ Chilblains ▪ Perniosis ▪ Kibes
- ▪
Cold-sensitive inflammatory disorder in which erythrocyanotic discoloration of acral skin is accompanied by a sensation of itching, burning or pain
- ▪
Common trigger is exposure to cold, wet, non-freezing conditions
- ▪
Must be distinguished from chilblain lupus and cold-sensitive blood dyscrasias
- ▪
Nifedipine can be used to treat moderate to severe pernio
Introduction
Pernio is an abnormal inflammatory response to cold, damp, non-freezing conditions . It is most common in areas without central heating.
History
Over a hundred years ago, pernio/chilblains was apparently common enough in Europe and the UK that the condition was well recognized. In 1881, Piffard described it quite well:
“This affection, so common in cold climates, affects, by preference, the face, hands, nose, and ears. It is characterized by redness, usually with a purplish tinge, together with more or less pain of a burning character giving little or no trouble during the summer, but causing inconvenience and suffering as cold weather sets in.”
Epidemiology
Exposure to cold, wet conditions is the major risk factor for pernio. This condition is common in the UK and northwestern Europe, particularly in those whose homes lack central heating. Women, children, and the elderly are most commonly affected. Elderly patients may have a prolonged course, while younger patients improve spontaneously .
Pathogenesis
The precise pathogenesis of pernio is unknown, but the condition is thought to represent an abnormal inflammatory response to vascular ischemia caused by prolonged vasoconstriction in the setting of extended cold exposure . In children and adults, it may be associated with cryoglobulins or cold agglutinins .
Clinical features
Pernio presents with single or multiple, erythematous to blue–violet macules, papules or nodules ( Fig. 88.7A–C ). In severe cases, blistering and ulceration may be seen ( Fig. 88.7D ). Lesions are often symmetrically distributed on the distal toes and fingers, and less often on the heels, nose, and ears. Overlying scale can also develop (see Fig. 88.7B ). Deep pernio may be seen on the thighs, calves, and buttocks as blue-erythrocyanotic plaques. Patients describe itching, burning or pain. Lesions often resolve within 1 to 3 weeks, except among elderly patients with venous insufficiency, in whom lesions can become chronic .
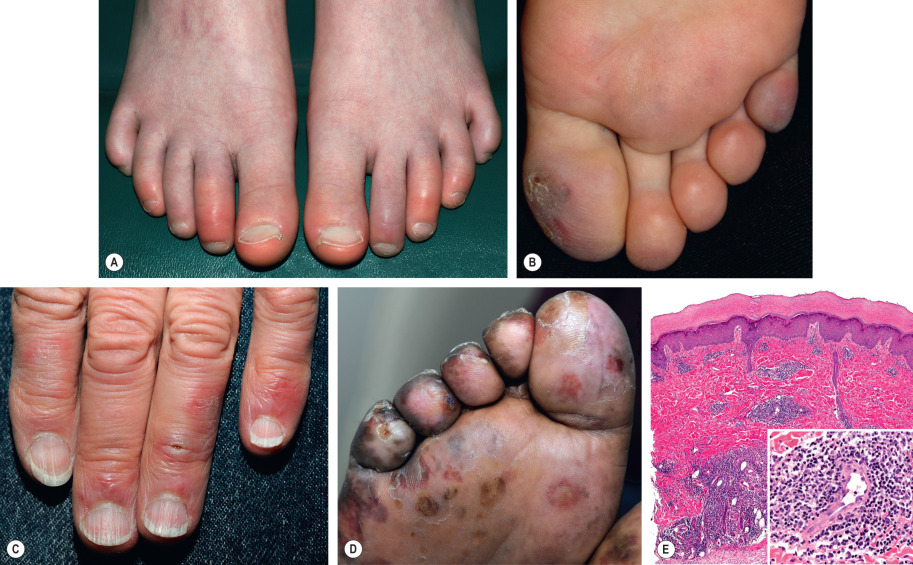
Pathology
Pernio has a nonspecific histology consisting of dermal edema plus a superficial and deep lymphohistiocytic infiltrate with peri-eccrine accentuation ( Fig. 88.7E ). The infiltrate is composed predominantly of T lymphocytes. Necrotic keratinocytes and lymphocytic vasculitis have been noted .
Differential diagnosis
Idiopathic pernio needs to be distinguished from several other cold-induced syndromes ( Table 88.8 ) . In addition to clinical features, e.g. retiform purpura, these entities can be further assessed by laboratory evaluation including: (1) complete blood count to exclude hemolytic anemia, myelogenous leukemia, or myelodysplastic syndrome; (2) cryoglobulin, cold agglutinin, and cryofibrinogen levels to eliminate cold-sensitive dysproteinemia; and (3) serum protein electrophoresis and immunofixation electrophoresis to exclude a monoclonal gammopathy. Chilblain lupus (Hutchinson), whose clinical features are similar to pernio, typically exhibits histologic features compatible with discoid lesions of lupus erythematosus and occurs in a patient with serologic evidence of autoimmunity (e.g. positive antinuclear antibody screen). Patients with type I interferonopathies, such as Aicardi–Goutières syndrome/familial chilblain lupus and proteosome-associated autoinflammatory syndrome/CANDLE syndrome, develop pernio-like lesions (see Table 45.7 ). Symmetric lividity of the palms and soles (pressure points) can involve the distal fingers and toes but is due to occlusive footwear, usually in association with hyperhidrosis. Diagnosis of the lupus pernio form of sarcoidosis, which favors the nose, is confirmed by the presence of the histologic features of sarcoidosis .
| DIFFERENTIAL DIAGNOSIS OF SKIN LESIONS INDUCED BY NON-FREEZING COLD EXPOSURE | |||
|---|---|---|---|
| Disorder | Features | Possible associated conditions | Chapter reference |
| Acrocyanosis | Red to purple discoloration Hands and feet Painless | Erythromelalgia | 106 |
| Cryoproteins | 23 | ||
| Anorexia nervosa | 51 | ||
| Pernio | Erythrocyanotic Symmetric distribution Painful | 88 | |
| Raynaud phenomenon | Clearly demarcated pallor, followed by blue then red discoloration Idiopathic form usually does not ulcerate | AI-CTD Blood dyscrasias Drugs Trauma | 43 |
| Livedo reticularis | Bluish, broad reticulated patches May be idiopathic | AI-CTD Hematologic disorders Vascular occlusive diseases Infections Medications | 106 |
| Cold panniculitis | Erythematous indurated plaques, most often on cheeks Children more commonly affected Upper lateral thighs in equestrians Can resolve in 2 weeks | 100 | |
| Cold urticaria | Cold-induced wheals May be idiopathic | Cryoproteins (see below) Infections Familial cold autoinflammatory syndrome | 18 |
| Chilblain lupus | Cold-induced acral lesions Histologic features of lupus erythematosus Coexistent lupus erythematosus | 41 | |
| “Pulling boat hands” | Rowing in cold, wet conditions Papules to vesicles Itching, burning, tenderness | ||
| Retiform purpura due to cryoproteins * | Favors acral sites | 23 | |
| Cryoglobulins | Cold serum protein precipitate | Plasma cell dyscrasias Lymphoproliferative disorders | |
| Cryofibrinogen † | Cold plasma protein precipitate | Infections Malignancies | |
| Cold agglutinins † | RBCs agglutinate in cold Occasional RBC lysis in cold | Infections (e.g. with Mycoplasma , EBV, CMV) Lymphoproliferative disorders | |
| Cold hemolysins † | RBC lysis in cold Paroxysmal hemoglobinuria | Infections (e.g. viral, syphilis) | |
* Acrocyanosis, Raynaud phenomenon, livedo reticularis, and cold urticaria can also be observed in association with cryoproteins.
Treatment
Adequate clothing and avoidance of cold, damp conditions are important preventive measures, as are keeping feet dry and avoidance of smoking. In a double-blind, placebo-controlled trial, nifedipine (20 to 60 mg daily) was found to be efficacious for the treatment of pernio. None of the nifedipine-treated patients relapsed, while all of the placebo-treated patients developed new lesions . It is effective in about 70% of patients with pernio. Other anecdotal remedies include topical corticosteroids, topical 0.2% nitroglycerin, oral nicotinamide, pentoxifylline, phenoxybenzamine, sympathectomy, and erythemogenic UVB phototherapy .
Injury Due to Water Exposure
Immersion Foot
▪ Trench foot ▪ Tropical jungle foot ▪ Shelter foot ▪ Paddy-field foot ▪ Swamp foot ▪ Jungle rot ▪ Foxhole foot ▪ Peripheral vasoneuropathy
- ▪
Injury occurs after continuous exposure of feet to moist, occluded conditions
- ▪
Cold water, warm water, and tropical variants exist
- ▪
The underlying pathologic process is overhydration of the stratum corneum
- ▪
Neuropathy may persist indefinitely
- ▪
Feet exposed to immersion injury are more sensitive to re-injury
Introduction
Immersion injury occurs in either cold or warm environments when the feet are exposed to continuous moisture with no opportunity for intermittent drying. Severe injury may produce permanent peripheral neuropathy and long-term morbidity.
History
Soldiers on the Western Front in World War I spent extended periods of time in the cold, wet, muddy conditions of trench warfare. The Second World War produced another variant, with shipwrecked troops spending days in partially submerged lifeboats. The Korean conflict and the Vietnam War led to more variations and tremendous numbers of casualties. The most recent military experience with immersion foot occurred during the Falklands War. Civilian immersion foot also occurs singly or in small groups, particularly among the homeless .
Epidemiology
Susceptibility to immersion foot is widely variable. Use of sandals or open shoes, constant activity, thinner plantar stratum corneum, and intermittent air drying seem somewhat protective. Risk factors for immersion foot include: prolonged occupational or recreational exposure to wet conditions, immobility, constrictive clothing, dehydration, poor nutritional status, smoking, altered mental status, peripheral vascular disease, uncontrolled diabetes, trauma, prolonged dependency of feet, and homelessness .
Pathogenesis
The underlying pathophysiology of all forms of immersion foot is overhydration of the stratum corneum. The permeability coefficient of plantar skin is ~10 times that of the dorsal foot skin, leading to increased water absorption. The plantar stratum corneum can absorb ~200% of its dry weight in water, with a proportionately higher absorption of fresh water than saline.
Warm water immersion foot (WWIF) results from severe hyperhydration of the stratum corneum, with subsequent maceration. Plantar skin temperature reaches body core temperature after about an hour in an impervious boot with 19°C (66°F) water. As the injury continues, inflammatory changes develop and may progress to a lymphocytic vasculitis with vascular compromise and erythrocyte extravasation ( tropical immersion foot [TIF]). Eventual involvement of the dorsal foot skin develops with striking inflammation. Secondary infection may develop in the compromised tissue after 3–4 days, further complicating management and worsening morbidity .
Cold water immersion foot (CWIF) is more severe due to the addition of cold-induced vasospasm. Because water conducts heat 23 times faster than air, the cold water immersion invokes “thermo-protective” peripheral vasoconstriction. This creates hypoxic injury to nerves (initially large myelinated axons, but eventually all types) and muscle, with later injury to subcutaneous fat and blood vessels.
Clinical features
The clinical syndromes of immersion foot are divided into the three variants previously discussed: WWIF, TIF, and CWIF ( Table 88.9 ) . Although TIF is sometimes viewed simply as a prolonged and more severe variant of WWIF, it is discussed separately because prolonged warm water exposure leads to different clinical features.
| IMMERSION FOOT: WARM WATER, TROPICAL, AND COLD WATER VARIANTS | |||||
|---|---|---|---|---|---|
| Variant | Exposure temperature | Exposure time | Clinical features | Complications | Duration |
| Warm water | 15.5–32°C (60–90°F) | 1–3 days | Thickening, softening, and exaggerated wrinkling of soles (thicker soles worse) Ambulation painful (“walking on rope”) | 3 days | |
| Tropical | 21–32°C (70–90°F) | 3–7 days | Prolonged warm water exposure In addition to plantar hyperhydration, dorsal foot becomes inflamed and edematous Pruritus, erythema, and scattered vesicles Later purpura, brawny edema, maceration, and scaling Fever, lymphadenopathy | >5 days | |
| Cold water | 15.5–21°C (60–70°F) | >7 days | Feet cold, heavy, stiff, and numb May appear swollen Waxy white to mildly cyanotic | Infection Muscle atrophy Chronic venous stasis Joint contractures Trophic ulcers Myonecrosis Permanent axonal damage Bony changes such as hammer toe | |
Three phases after rewarming:
| Hours to days | ||||
| Up to 10 weeks | ||||
| Years | ||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree








