Dual innervation in free muscle flap facial reanimation has been used to create a functional synergy between the powerful commissure excursion that can be achieved with the masseter nerve and the spontaneity that can be derived from a cross-face nerve graft. The gracilis has been the most frequently used muscle flap, and multiple combinations of neurorrhaphies have been described, including the masseter to the obturator (end-to-end) combined with a cross-face nerve graft to the obturator (end-to-side) and vice versa. Single and staged approaches have been reported. Minimally, dual innervation is as effective as using the motor nerve to masseter alone.
Key points
- •
Dual innervation can be used to create a marriage of power and spontaneity in free functional muscle flap smile restoration.
- •
Multiple patterns of dual innervation have been described using cross-face nerve grafts and nerve to masseter with no specific pattern, demonstrating a distinct advantage at this time.
- •
The presence of ephaptic nerve conduction may help explain the synergy achieved with dual innervation.
Introduction
Free functional muscle flaps are a highly effective and widely accepted method of smile restoration in cases of developmental and long-standing facial paralysis. The contralateral facial nerve via a cross-face nerve graft (CFNG) and ipsilateral nerve to masseter (NTM) are the most frequently used sources of innervation, with each donor site possessing a series of strengths and weaknesses. The most beneficial property of CFNGs is their ability to produce spontaneous, emotionally mediated muscle activation. Unfortunately, the commissure excursion produced by free muscle flaps innervated by CFNGs may be weaker than the uninjured side. The technique is well suited to children and young adults; however, the ability to achieve smile symmetry declines with age. No strict age cutoff for the technique has been established; however, many surgeons will transition to an ipsilateral nerve donor (CN V, XII, XI) after the third decade of life. In addition, larger muscle flaps are often used to accommodate for the lower axon densities yielded by CFNGs.
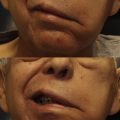
In contrast, the masseteric nerve has a dense population of myelinated axons that can reliably produce powerful commissure excursion when used to innervate free muscle flaps. The reconstruction can be achieved in a single surgery, and small, highly tailored muscle flaps can be used as a result of the high axonal load producing minimal cheek bulk. The Achilles heel of the masseter nerve donor site is its limited ability to produce both resting tone and spontaneous facial motion. There is little doubt that a subset of the population possesses communications between CN V and VII as evidenced by electromyographic (EMG) studies. , Regardless, the number of individuals that can achieve a truly spontaneous smile with a muscle flap innervated solely by the masseter nerve is limited. Clenching of the teeth is initially required for smile activation; however, with diligent practice over a 2-year period, most patients will not have to bite to elicit motion. In 1 study, 53% of patients thought they could produce a smile without conscious effort, which was accredited to the process of cerebral adaptation. In the authors’ experience, the development of an effortless, reflexive smile is common; however, true spontaneous motion arising from the masseteric nerve is rare.
The central question to be answered is whether dual innervation of a free muscle flap with a CFNG and the ipsilateral masseter nerve will produce a functional synergy, creating a marriage of power (CN V) and spontaneity (CN VII).
Discussion
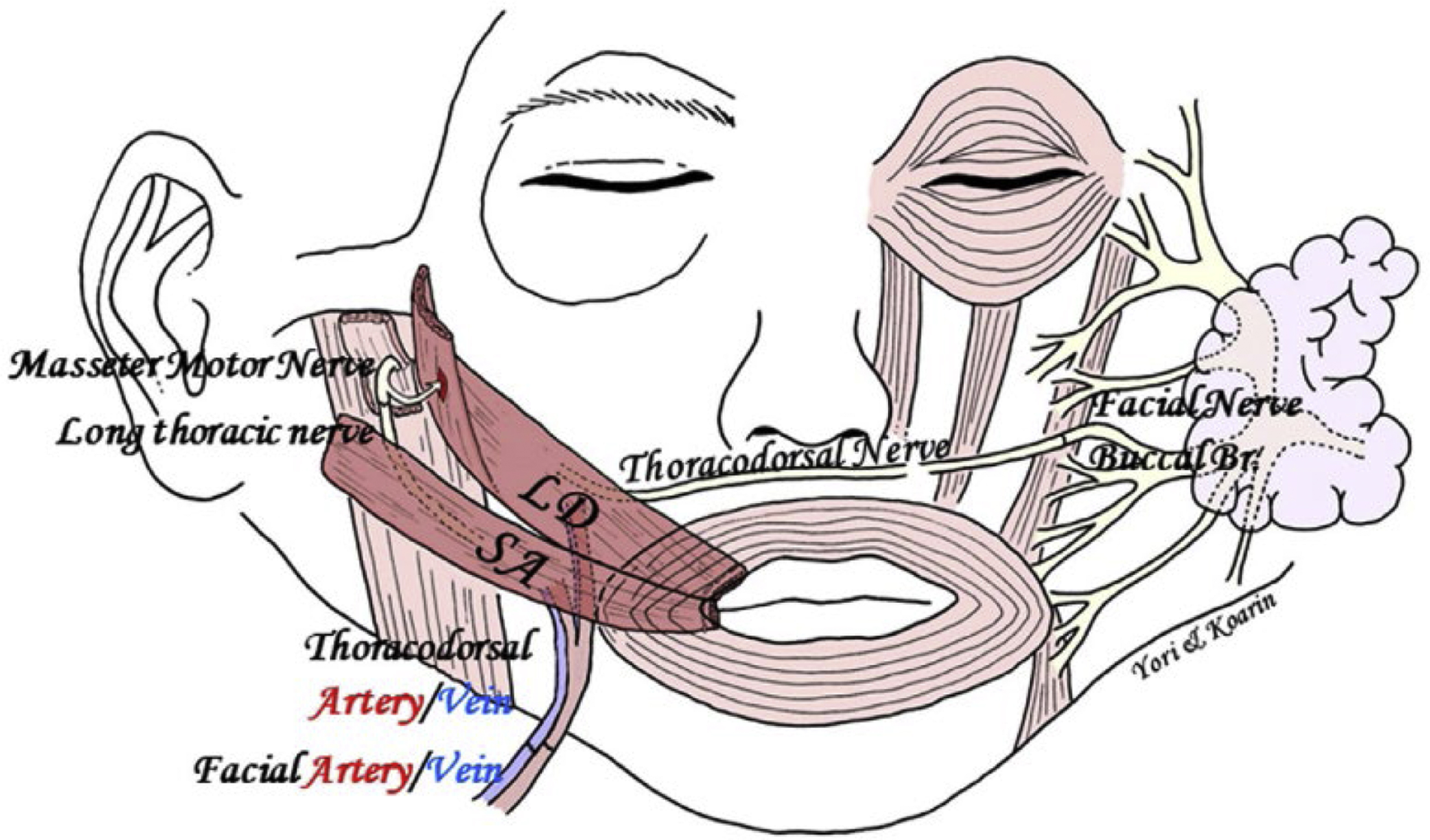
A series of different groups have explored this concept and have taken significantly different approaches in their effort to answer this question. Watanabe and colleagues are widely credited for first reporting dual innervation of free muscle flaps for facial reanimation. Their initial technique used a single-stage free latissimus dorsi flap with coaptation of the thoracodorsal nerve to the contralateral facial nerve. The masseter muscle was denuded and placed in direct contact with the latissimus dorsi muscle to encourage muscle-to-muscle neurotization. The technique has subsequently been modified to include a composite latissimus dorsi-serratus anterior muscle flap to create a dual vector. The thoracodorsal nerve is again anastomosed to a contralateral facial nerve branch. However, in this technique, a motor nerve branch to masseter is buried into the latissimus dorsi muscle to achieve direct intramuscular neurotization. The long thoracic nerve innervating the associated slip of the serratus anterior is coapted to a second “thin” motor branch to masseter, creating direct nerve-to-nerve neurotization ( Fig. 1 ). Small masseter branches and direct muscle neurotization are selected to disadvantage the masseter nerve and prevent it from overwhelming innervation arising from the contralateral facial nerve. Regardless of the style of nerve coaptation, using the masseter nerve in this fashion has the potential to limit muscle atrophy with the one-stage technique, where one must wait for axonal growth through a cross-face nerve segment. Innervation strategies like this could potentially permit the utilization of smaller muscle flaps that create less bulk, which has been a drawback of the one-stage latissimus flap technique.
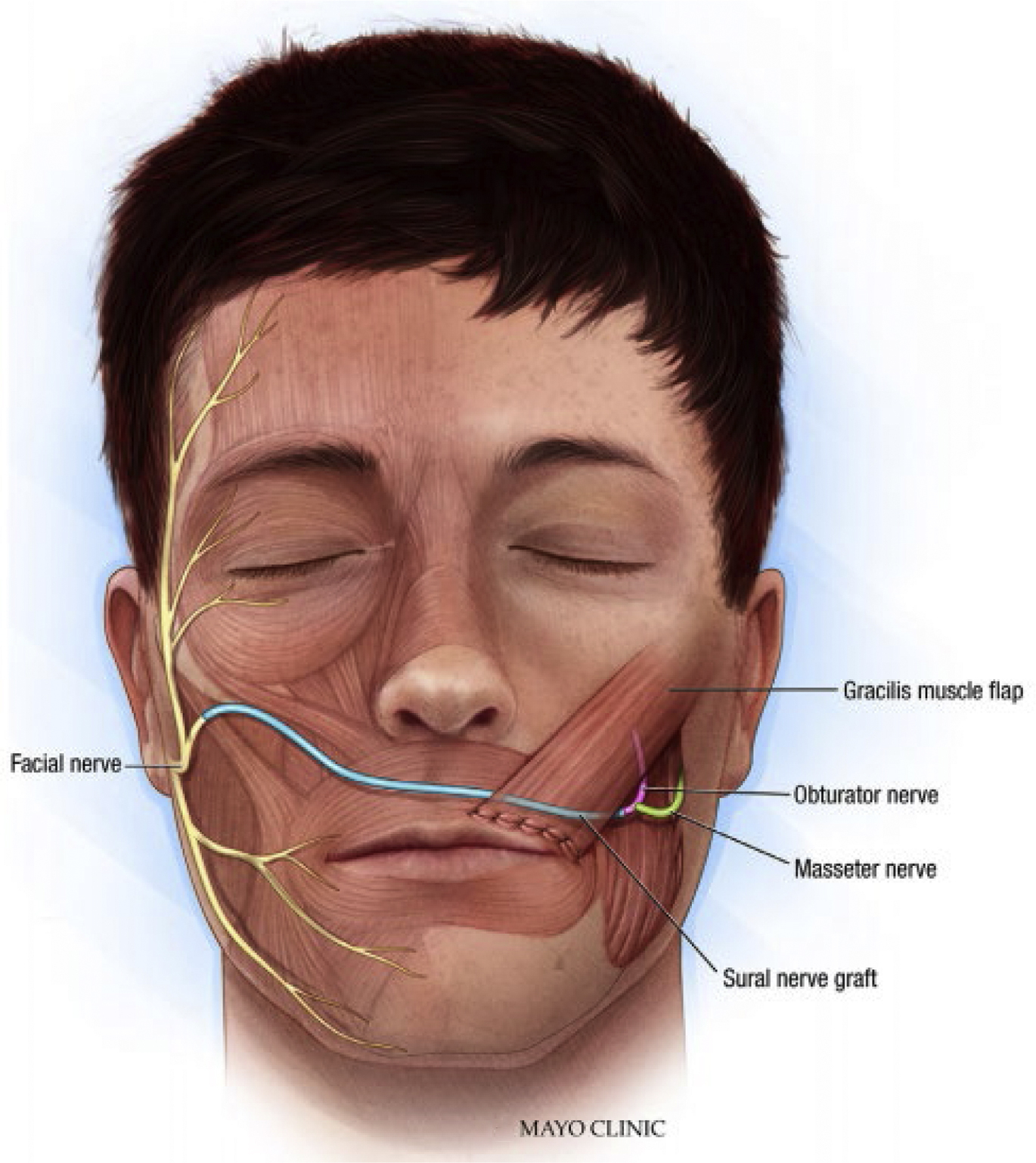
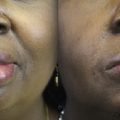
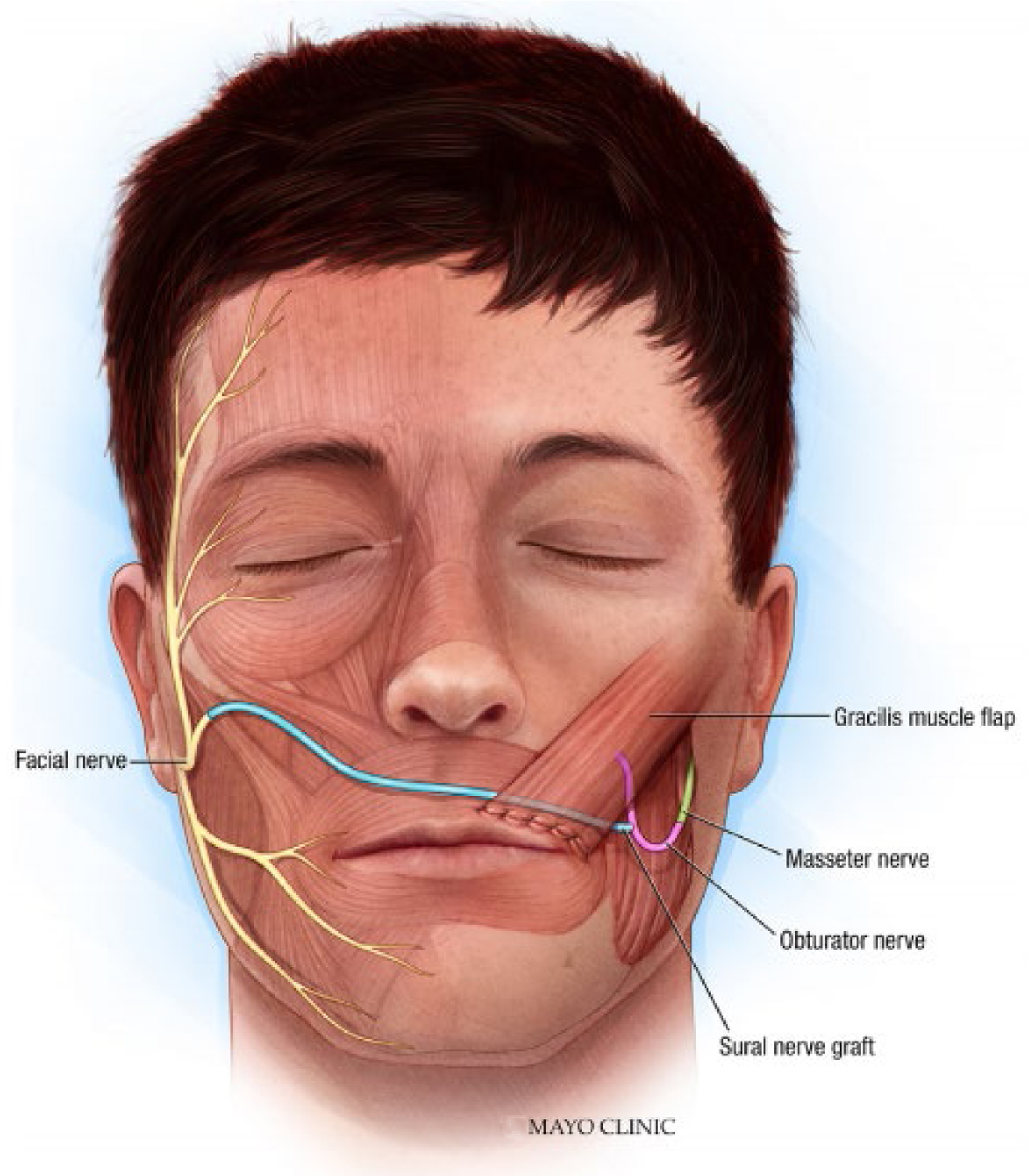
Biglioli and colleagues have also investigated the potential value of dual innervation. Their technique uses a single-stage free gracilis muscle flap innervated by the masseter and contralateral facial nerve. The obturator nerve is anastomosed end to end to the masseter nerve, and the distal CFNG is coapted to the obturator nerve end to side ( Fig. 2 ). Innervating in this fashion clearly provides a distinct advantage to the masseter nerve with its larger axon load, close proximity, and more efficient anastomosis (end to end). There is a potential concern with this pattern of connections that the masseter nerve will win the race to the motor endplates, crowding out the late-arriving axons from the CFNG. Regardless, this method of dual innervation is most frequently cited in the literature, and this study reports 2 excellent and 2 good results according to the Terzis classification with EMG evidence of gracilis muscle activity with stimulation of the contralateral facial nerve. The investigators postulate that the strength of spontaneous muscle contraction may represent axons from the CFNG costimulating the motor nerve fibers from masseter in conjunction with the flaps limited denervation time. The idea of a weaker population of nerve fibers triggering a more robust population is very intriguing and will be explored later in this article.
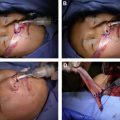
Cardenas-Mejia and associates have also described using these sources of innervation but in a distinctly different way. A dually innervated free gracilis muscle flap is performed in 2 stages. A CFNG with a proximal end-to-end anastomosis is performed during the first stage. Following successful nerve growth through the graft, the second stage is performed. Here, the CFNG is anastomosed end to end to the obturator nerve, and the masseter nerve is coapted to the obturator nerve end to side approximately 1 cm from the hilum ( Fig. 3 ). This configuration is used to optimize the opportunity of the axons from the CFNGs to occupy the flaps motor end plates before arrival of nerve fibers for the masseter nerve. The authors report 9 cases, with Terzis grade 3 achieved in 1 cases, grade 4 achieved in 4 cases, and grade 5 achieved in 4 cases.