Outcomes following free gracilis muscle transfer have steadily improved during the past decade. Areas for continued improvement include re-creating natural smile vectors, improving midface symmetry, minimizing scarring, improving spontaneity, and increasing reliability using various neural sources. Outcome standardization, pooled data collection, and remote data acquisition methods will facilitate comparative effectiveness research and continued surgical advancements.
Key points
- •
Masseteric-driven free gracilis muscle transfer (FGMT) reliably produces a voluntary smile.
- •
Cross-face nerve graft (CFNG)-driven FGMT can produce a spontaneous smile, but with a higher risk of failure.
- •
Dual-innervation FGMT attempts to combine the reliability of masseteric-driven procedures with the spontaneity of CFNG; some element of spontaneity is achieved in approximately 50% of cases.
- •
Current innovations revolve around re-creating natural smile vectors, improving midface symmetry by performing simultaneous static techniques, avoiding bulk, minimizing scarring, improving spontaneity, and increasing reliability.
- •
Outcome standardization, pooled data collection, and remote data acquisition methods will be critical to continued improvement of FGMT outcomes.
Introduction
History
The smile is an evolutionarily adaptive facial expression that facilitates complex social interactions. People smile to convey sympathy, happiness, sociability, and positive intentions toward others. , Further, people perceive smiling faces as having increased happiness, intelligence, and social standing. Hence, smile restoration is a critical component of successful facial reanimation.
Functional muscle transfer has been central to smile restoration since first being described by Sir Harold Gillies. In 1976, Harii and colleagues described using the deep temporal nerve to innervate the first free gracilis muscle transfer (FGMT) for smile reanimation. In 1980, O’Brien and colleagues described cross-face nerve graft (CFNG)-driven FGMT, and since then, the hypoglossal, spinal accessory, and masseteric nerves have been used to innervate transferred gracilis muscle. Most recently, dual-innervation FGMT using the masseteric nerve and a CFNG have been used to produce a spontaneous and reliable smile.
A decade ago, FGMT had unclear success rates in producing a smile; limited data demonstrated highly variable outcomes. Trigeminally-driven smiles increased in popularity, and ultimately the combination of multiple neural sources emerged as a potential reanimation strategy. This review discusses recent advances in FGMT technique and outcomes reporting.
Key Definitions
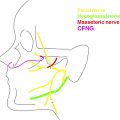
FGMT outcomes assessment must consider innervation and vector design. In masseteric-driven FGMT, the ipsilateral masseteric branch of cranial nerve V3 is coapted to the anterior branch of the obturator nerve, providing proximal neural input. An anterior branch of the masseteric nerve is preserved, when possible, to prevent asymmetry from muscle atrophy. In CFNG-driven FGMT, a healthy-side facial nerve branch that produces a pleasing smile innervates the gracilis, typically via a CFNG placed 6 to 9 months earlier. Direct neurotization without a graft is possible, although outcomes data are limited. Dual-innervation FGMT uses the ipsilateral masseteric nerve and a CFNG to power the gracilis. Dual-vector (aka multivector) FGMT uses 2 muscle paddles supplied by a common neurovascular pedicle, attempting to recreate natural smile vectors.
Historic obstacles to successful smile reanimation with the gracilis flap
Unreliability of Cross-Face Nerve Graft–Driven Free Gracilis Muscle Transfer
CFNG-driven FGMT is significantly more likely to produce a spontaneous smile compared with alternative neural sources. Unfortunately, these procedures have higher failure rates. O’Brien and colleagues 11 originally reported 51% of patients achieved a smile after CFNG-driven FGMT, and more recent data demonstrate CFNG-driven procedures account for 71% of FGMT failures. Further, CFNG-driven FGMT produces less oral commissure (OC) excursion compared with masseteric-driven procedures. , , ,
Lack of Spontaneity in Masseteric-Driven Free Gracilis Muscle Transfer
Masseteric-driven FGMT has excellent reliability; up to 95% of patients produce a smile after surgery. , Despite this excellent reliability, masseteric-driven procedures have low success rates at producing a spontaneous smile. , , Historically, lack of spontaneity in masseteric-driven FGMT, along with inconsistent outcomes in CFNG-driven procedures, forced patients and providers to choose between a procedure with high success rates versus a procedure that could produce a spontaneous smile, but with a greater chance of failure.
Cross-Face Nerve Graft–Driven Free Gracilis Muscle Transfer Triggered by Eye Closure
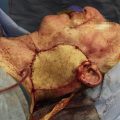
Rarely, CFNG-driven FGMT only produces a smile with eye closure, leading to suboptimal outcomes ( Fig. 1 ). This inappropriate smile trigger likely results from incorrect donor nerve selection during first-stage CFNG. Nerve selection can be difficult, as both nerve caliber and muscle movements elicited by nerve stimulation must be considered when selecting healthy-side donor facial nerve branch(es).
Dynamic Success but Poor Aesthetic Outcome
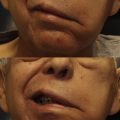
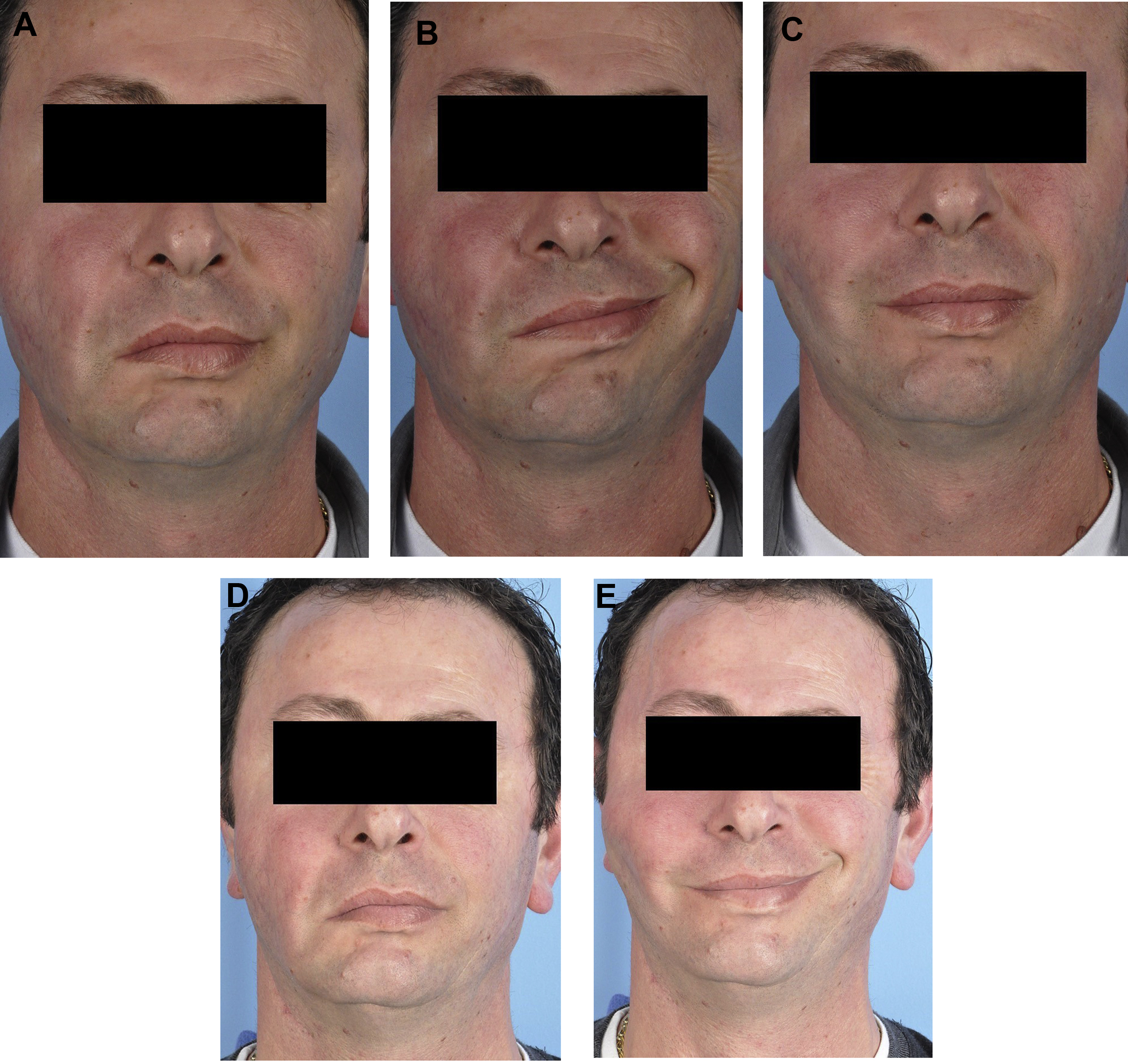
Data typically report changes in OC excursion after FGMT, despite lack of correlation between this measurement and joy conveyed by smiling. , , In Fig. 2 , excellent OC excursion is seen after FGMT, but the smile appears unnatural because of superior malpositioning of the lower lip ( Fig. 2 ). Historically, surgeons often focused on maximizing OC excursion rather than re-creating natural smile vectors.

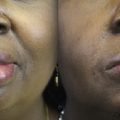
Suboptimal FGMT outcomes can also result from midfacial asymmetry. Midfacial bulk after FGMT can be disfiguring, and patients may avoid smiling to prevent dynamic asymmetry ( Fig. 3 ). In 1984, Manktelow and Zuker described transferring a “fascicular territory” of gracilis muscle to decrease bulk. Six years later, in situ muscle thinning was described as another strategy to decrease bulk while minimizing risk for facial hematoma. Despite these techniques, Terzis and Noah reported a 26% revision rate to debulk gracilis muscle in their 1997 case series.
Other factors can also contribute to facial asymmetry after FGMT. Although lymphedema is a widely accepted outcome associated with head and neck cancer therapies, it also likely contributes to midfacial asymmetry in FGMT as large facial and neck flaps are raised. Inappropriate facial contours and hollowing, secondary to fat atrophy and buccal fat prolapse, also can occur after FGMT.
Discussion
Meticulous data collection and outcomes assessment are critical to resolving pitfalls of FGMT. Comprehensive facial paralysis outcomes assessments, which allow comparison among innovations designed to address and resolve these pitfalls, include patient-reported outcome measures, automated analyses, clinician grading, layperson assessments, and spontaneity evaluation.
Addressing Lack of Spontaneity with Masseteric-Driven Free Gracilis Muscle Transfer and High Failure Rate with Cross-Face Nerve Graft–Driven Free Gracilis Muscle Transfer
Ideally, FGMT will combine reliability of masseteric-driven procedures with spontaneity of CFNG-driven FGMT. Dual-innervation FGMT, in which the gracilis is innervated by the masseteric nerve (end-to-end coaptation) and a CFNG (end-to-side coaptation), was described by Biglioli and colleagues to optimize reliability and spontaneity. Other neural coaptation patterns since described include Y-shaped and interfascicular split. Rigorous testing of dual-innervation FGMT demonstrates 33% of patients achieve a spontaneous smile, compared with 75% of patients who demonstrate spontaneity after CFNG-driven procedures. A recent review demonstrated higher rates of spontaneity after dual-innervation FGMT, but testing was not standardized. Reliability of dually innervated flaps is excellent. , , , Critical and standardized assessment of dual-innervation FGMT would clarify optimal coaptation pattern(s), and 1-stage versus 2-stage procedure outcomes.
An attractive strategy that may find clinical application in the near future is to provide patients with a reliable and spontaneous smile is a neuroprosthetic device, yoking the paretic hemiface to the healthy side. Implantable neuroprosthetic devices have been developed to treat hearing loss, obstructive sleep apnea, and improve control of prosthetic limbs. Multiple groups have demonstrated successful stimulation of paralyzed facial muscles using signals from the healthy hemiface in animal models. Devices also must prevent undesirable facial movements during activation to be clinically practical. Jowett and colleagues delivered high-frequency alternating current to the proximal facial nerve to inhibit this undesirable activity, without affecting distal nerve stimulation. Future work should continue to investigate long-term use of implantable neuroprosthetic devices to drive desirable facial movements.
Re-creating a Natural Smile
FGMT has benefited from improved understanding of characteristics that define a natural smile. Boahene and colleagues described multivector FGMT to recreate the zygomaticus and levator labii muscles. A multivector approach has also been described for smile reanimation using serratus anterior and sterno-omohyoid free muscle transfers. , Preliminary work to introduce multivector muscle slips suggests that improved smile morphology may be achieved using this surgical strategy.
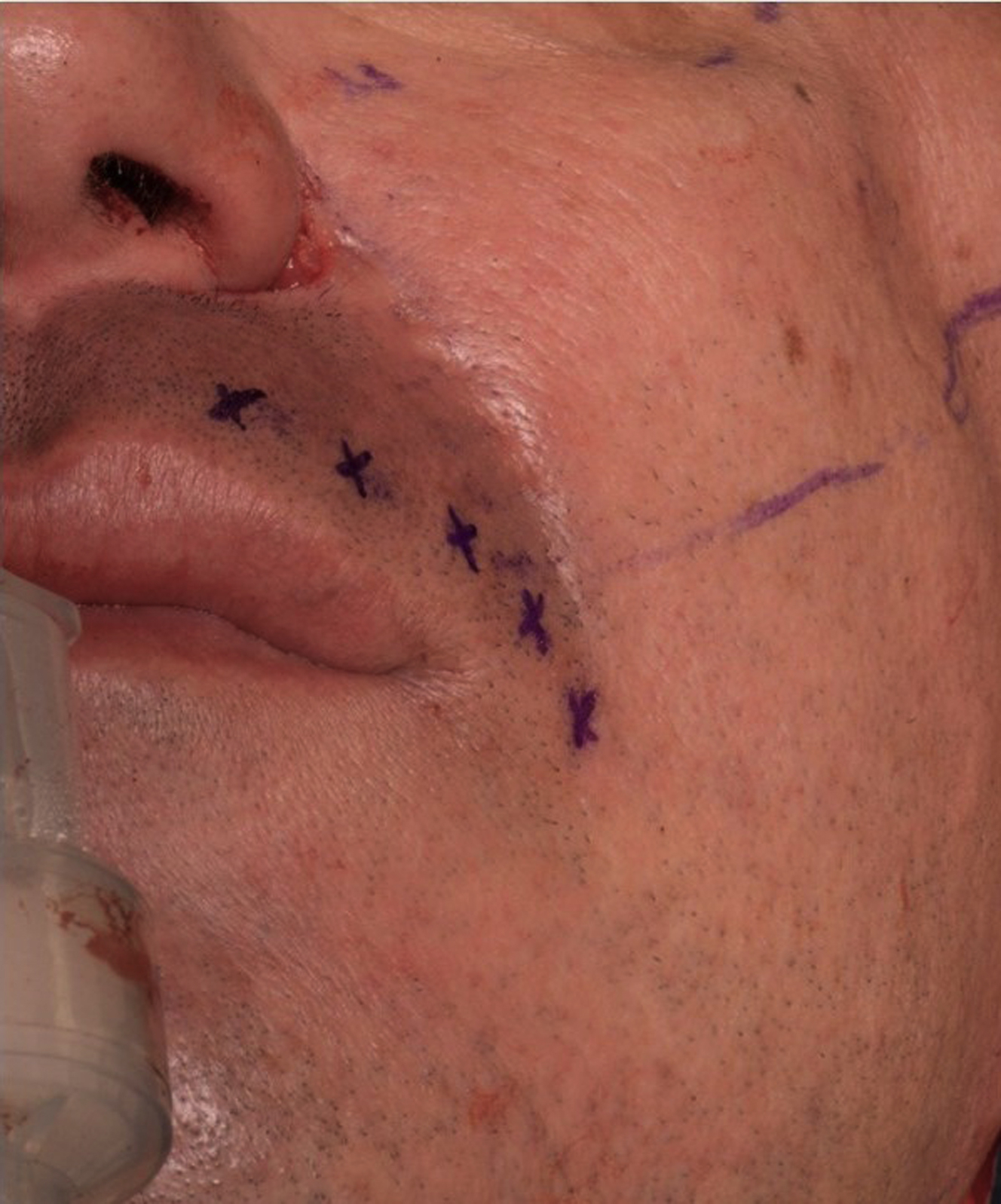
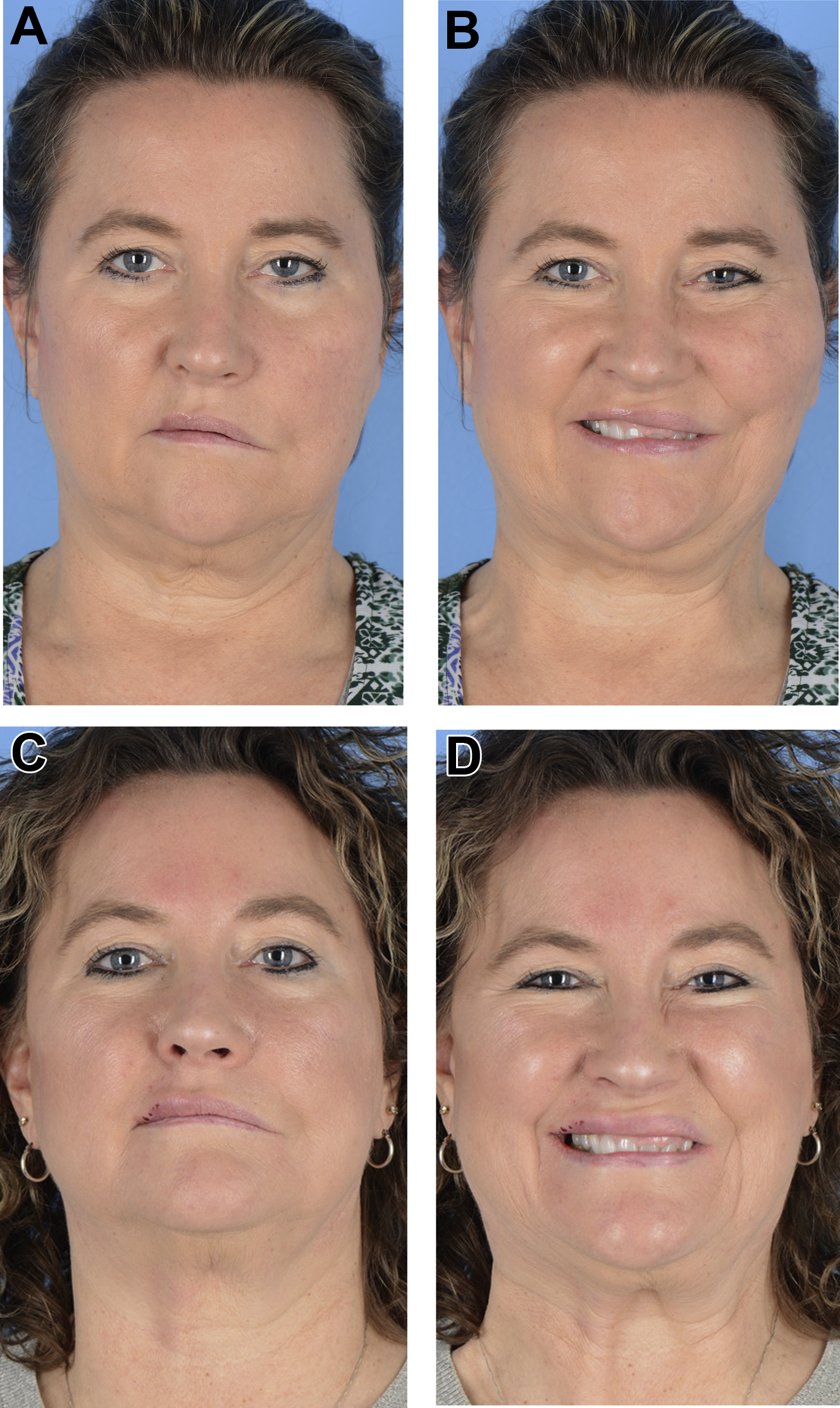
Historically, a commonly used FGMT inset technique involved securing the muscle primarily at the modiolus to animate the upper and lower lateral lip. However, this creates unnatural superior malpositioning of the lower lip with smile (see Fig. 2 ). A modified medial inset secures the muscle across the upper lip, from modiolus to philtrum, to improve dental show and create a more natural smile ( Figs. 4 and 5 ). Outcomes produced by these new inset and multivector techniques should be evaluated using prior outcomes as control groups.