Abstract
The skin is one of the most common targets for adverse drug reactions. Approximately 2% of all drug-induced skin reactions are considered “serious” based upon the World Health Organization (WHO)’s definition, including acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms/drug-induced hypersensitivity syndrome (DRESS/DIHS), Stevens–Johnson syndrome, and toxic epidermal necrolysis. Over the past decade, the introduction of new targeted therapies and immunotherapies has led to a considerable increase in the incidence of cutaneous adverse reactions (CARs).
The diversity of drug-induced skin reactions is reflected in the various underlying pathomechanisms, and there has been significant progress in understanding cutaneous adverse reactions, including genetic predispositions. In general, the drugs potentially responsible for a particular CAR can be placed into low-, medium-, or high-probability categories. Although the two most common skin reactions are an exanthematous (or morbilliform) eruption and urticaria, recognition of the many less common CARs is an important aspect of the field of dermatology and they are reviewed in this chapter. Prompt discontinuation of the offending agent represents the initial, and often only, intervention.
Keywords
drug reaction, cutaneous drug reaction, cutaneous adverse reaction, serious cutaneous adverse reaction, SCAR, exanthematous drug reaction, morbilliform drug reaction, fixed drug eruption, acute generalized exanthematous pustulosis, AGEP, drug reaction with eosinophilia and systemic symptoms, drug-induced hypersensitivity syndrome, DRESS, DIHS, photoxicity, urticaria, Stevens–Johnson syndrome, SJS, toxic epidermal necrolysis, TEN, injection site reactions, anaphylactoid reactions
- ▪
The skin is one of the most common targets for adverse drug reactions
- ▪
To determine the cause of the eruption, a logical approach based on clinical characteristics, chronologic factors, and a literature search is required
- ▪
Exanthematous eruptions and urticaria are the two most common forms of cutaneous drug reactions
- ▪
When an exanthematous eruption is associated with fever, lymphadenopathy, and/or edema of the face, drug reaction with eosinophilia and systemic symptoms (DRESS)/drug-induced hypersensitivity syndrome (DIHS) must be considered and an evaluation for systemic involvement conducted
- ▪
Less common drug reactions include fixed, lichenoid, pustular, granulomatous, pseudolymphomatous, acneiform, bullous, photosensitivity and vasculitic reactions, as well as Stevens–Johnson syndrome and toxic epidermal necrolysis
- ▪
Early withdrawal of all potentially responsible drugs is essential, particularly in the case of severe cutaneous adverse reactions (SCARs)
Introduction
The skin is one of the most common sites for adverse drug reactions . Although eruptions are observed in 0.1% to 1% of patients enrolled in pre-marketing trials of most systemic drugs, higher incidences have been reported with many of the newer targeted therapies. Up to 5% of patients treated with antibiotics and aromatic anticonvulsants may develop a cutaneous eruption. Approximately 2% of all drug-induced skin reactions are considered “serious” according to the following World Health Organization (WHO) definition: “if it results in death, requires hospitalization or prolongation of existing hospital stay, results in persistent or significant disability/incapacity, or is life-threatening”. Toxic epidermal necrolysis (TEN) and drug reaction with eosinophilia and systemic symptoms (DRESS; also referred to as drug-induced hypersensitivity syndrome [DIHS]) are examples of such “serious reactions”. Roujeau and Stern estimated that 1 of every 1000 hospitalized patients has a serious cutaneous drug reaction. Prompt identification of severe cutaneous adverse reactions (SCARs) is an important goal, followed by discontinuation of the most likely offending drug(s) and thereby decreasing morbidity ( Table 21.1 ) .
| SEVERE CUTANEOUS ADVERSE REACTIONS (SCARs) |
|
It may be tempting to consider most eruptive cutaneous drug reactions, especially the severe ones, as various presentations of a “hypersensitivity syndrome”. However, in order to begin to understand these entities, it is more useful to examine the specificity of the clinical, pathologic and biologic patterns, thus eventually allowing classification of each type of adverse cutaneous reaction as a single entity with a well-defined and specific mechanism .
This chapter focuses on adverse cutaneous reactions due to systemically administered medications. In addition, several specific types of drug reactions are discussed in other chapters ( Table 21.2 ).
| ADDITIONAL REVIEWS OF SPECIFIC TYPES OF DRUG REACTIONS | |
|---|---|
| Psoriasiform | Chapters 8 & 128 |
| Erythroderma | Table 10.3 |
| Lichenoid | Table 11.2 |
| Urticaria | Chapter 18 |
| Stevens–Johnson syndrome and toxic epidermal necrolysis | Table 20.5 |
| Warfarin- and heparin-induced necrosis | Chapter 23 |
| Vasculitis | Table 24.4 |
| Pemphigus and bullous pemphigoid | Chapters 29 & 30 |
| Linear IgA bullous dermatosis | Table 31.5 |
| Acneiform/folliculitis | Table 36.1 |
| Hyper- and hypohidrosis | Tables 39.2 & 39.9 |
| Lupus erythematosus (systemic and cutaneous) | Chapter 41 |
| Pseudoporphyria | Table 49.5 |
| Hypopigmentation (skin and hair) | Table 66.9 |
| Hyperpigmentation and dyschromatosis | Tables 67.4 & 67.10 |
| Nail abnormalities | Table 71.8 |
| Gingival enlargement | Table 72.1 |
| Phototoxic and photoallergic | Table 87.5 |
| Granulomatous (interstitial) | Table 93.7 |
| Cutaneous lymphoid hyperplasia (pseudolymphoma) | Table 121.1 |
Epidemiology
Cutaneous reactions to drugs occur in up to 8% of hospitalized patients . In departments of dermatology, they represent 2% of consultations and ~5% of the admissions to an inpatient dermatology department. The incidence of fatalities due to drug reactions (systemic and cutaneous) among inpatients is between 0.1% and 0.3% . There are multiple risk factors for developing an adverse drug reaction including female gender, immunosuppression (e.g. HIV-infected individuals have a 10- to 50-fold greater risk of developing an exanthematous eruption to sulfamethoxazole), dermatomyositis (specifically for hydroxychloroquine), and specific HLA alleles .
The epidemiology of common adverse cutaneous drug reactions is still evolving. As a result, the incidence of drug reactions is difficult to establish precisely. Pre-marketing clinical trials, conducted before a new drug is licensed, include a limited number of patients, thus preventing a clear estimate of the true incidence. It is usually after several months or years of greater utilization that a more precise profile of the side effects of a newly released drug is established. Moreover, available information must be interpreted with caution, depending upon the method of data collection .
Reaction rates to commonly administered drugs have been confirmed by several prospective studies; however, the latter have been limited to inpatients . In one series, adverse skin reactions occurred in 2.7% of 48 000 patients hospitalized over a 20-year period on a general internal medicine service; maculopapular exanthems (91.2%), urticaria (5.9%), and vasculitis (1.4%) were the reactions most commonly observed. The primary responsible drugs were penicillins, sulfonamides, and nonsteroidal anti-inflammatory drugs (NSAIDs).
Except for studies in hospitalized populations and those focusing on the most severe reactions, data regarding the incidence of cutaneous drug reactions in the general population are minimal, in part due to the absence of comprehensive post-marketing surveillance programs. In a retrospective cohort study from the Netherlands of 13 679 patients from general practices, the most frequently reported skin reactions to antimicrobials were due to trimethoprim–sulfamethoxazole (2.1% of users), fluoroquinolones (1.6%), and penicillins (1.1%).
Pathogenesis
Drug-induced skin reactions are mediated by either an immunologic or a non-immunologic mechanism ( Table 21.3 ). In the former, drugs or their metabolites act as haptens, inducing a specific cell-mediated or humoral response. In the latter, some of the reactions are predictable given their relationship to the dose and/or the pharmacologic properties of the drug. However, the responsible pathophysiology may be difficult to identify simply on the basis of clinical appearance as the skin responds to a wide variety of stimuli through a limited number of morphologic reaction patterns. This may explain, at least in part, why the underlying mechanism of many cutaneous drug eruptions is still unknown.
| MECHANISMS OF CUTANEOUS DRUG-INDUCED REACTIONS | |
|---|---|
| Immunologic mechanism (unpredictable) |
|
| Non-immunologic mechanisms (sometimes predictable) |
|
| Idiosyncratic with a possible immunologic mechanism (unpredictable) |
|
Immunologically Mediated Drug Reactions
- •
IgE-dependent drug reactions (formerly type I, Gell–Coombs classification): urticaria, angioedema, and anaphylaxis.
- •
Cytotoxic drug-induced reactions (antibody against a fixed antigen; formerly type II): petechiae secondary to drug-induced thrombocytopenia.
- •
Immune complex-dependent drug reactions (formerly type III): vasculitis, serum sickness, and certain types of urticaria.
- •
Delayed-type, cell-mediated drug reactions (activation of CD4 + and CD8 + T cells; formerly type IV) : exanthematous, fixed, and lichenoid drug eruptions, as well as Stevens–Johnson syndrome (SJS) and TEN. This group has been further subdivided into the following:
- •
Th1 immune reaction (IVa): monocytes are preferentially recruited and activated by IFN-γ, leading to CD8 + T-cell activation and a proinflammatory response (TNF, IL-12)
- •
Th2 immune reaction (IVb): eosinophils are preferentially recruited and activated in part by IL-4, -5, -13 and eotaxin, as in DRESS
- •
Cytotoxic immune reaction (IVc): involves CD4 + and CD8 + T cells with release of perforin and granzyme B and/or Fas–FasL interactions, as in SJS/TEN (see Fig. 20.6 )
- •
Neutrophil and T-cell-based immune reaction (IVd): mediated via chemokines (e.g. CXCL8) and cytokines (e.g. GM-CSF), as in acute generalized exanthematous pustulosis (AGEP)
- •
Genetic factors
Specific HLA alleles have emerged as important genetic risk factors for SCARs, especially SJS/TEN and DRESS ( Table 21.4 ) . Given the strong associations between HLA-B*1502 and carbamazepine-triggered SJS/TEN in Asians and between HLA-B*5701 and abacavir-triggered DRESS, pretreatment genetic testing is now recommended.
| SPECIFIC HLA ALLELES THAT INCREASE THE RISK OF CUTANEOUS DRUG REACTIONS | |||
|---|---|---|---|
| Drug | Higher risk population(s) | HLA allele | Type of drug reaction |
| Abacavir | B*5701 | DRESS | |
| Allopurinol | Asians (Han Chinese) > Europeans | B*5801 | SJS/TEN |
| Asians (Taiwanese) > Europeans | B*5801 | DRESS | |
| Carbamazepine | Han Chinese and other Asian populations | B*1502 | SJS/TEN |
| Northern Europeans, Japanese, Chinese, Korean | A*3101 | Hypersensitivity reactions | |
| Dapsone | Chinese | B*1301 | DRESS |
| Feprazone (NSAID) | Scandinavians | B22 | FDE |
| Lamotrigine | Taiwanese | B*1502 | SJS/TEN |
| Nevirapine | French | DRB1*01:01 | DRESS |
| Phenytoin | Southeast Asians | B*1502 | SJS/TEN |
| Trimethoprim–sulfamethoxazole | Turkish | B55 | FDE |
Non-immunologic Mechanisms (see Table 21.3 )
Overdose
The clinical manifestations of a drug overdose are predictable and represent an exaggeration of the medication’s pharmacologic actions. It may occur as a consequence of a prescribing error, deliberate excess by the patient, or altered absorption, metabolism or excretion. An example of the latter is methotrexate toxicity in elderly patients with reduced renal function ( Fig. 21.1 ).
Pharmacologic side effects
These reactions include undesirable or toxic effects that cannot be separated from the desired pharmacologic actions of the drug. An example would be alopecia and mucositis due to chemotherapeutic agents that target more rapidly dividing cells.
Cumulative toxicity
Prolonged exposure to a medication or its metabolites may lead to cumulative toxicity. For example, methotrexate can lead to hepatic fibrosis and accumulation of minocycline or amiodarone within the skin can lead to cutaneous discoloration.
Delayed toxicity
This corresponds to a toxic, dose-dependent effect that occurs months to years after the discontinuation of a medication. Examples include squamous cell carcinomas and palmoplantar keratoses following exposure to arsenic and acute leukemia due to alkylating agents.
Drug–drug interactions
Interactions between two or more drugs administered simultaneously may occur at several different steps: (1) intestinal drug interactions; (2) displacement from binding proteins or receptor sites; (3) enzyme stimulation or inhibition; and (4) altered drug excretion (see Ch. 131 ). Examples of each include the interactions between tetracycline and calcium, methotrexate and sulfonamides, cyclosporine and azoles, and methotrexate and probenecid.
Alterations in metabolism
Drugs may induce cutaneous changes by their effects on the nutritional or metabolic status of the patient. Bexarotene may induce severe hypertriglyceridemia and eruptive xanthomas, while isoniazid may be associated with pellagra-like changes.
Exacerbation of disease
A variety of drugs can exacerbate pre-existing dermatologic diseases, such as androgens in patients with acne vulgaris or lithium and interferon in patients with psoriasis.
Idiosyncratic With a Possible Immunologic Mechanism (see Table 21.3 )
The pathophysiology of drug-induced skin reactions such as exanthematous drug eruptions, DRESS, AGEP and TEN, as well as the increased susceptibility of HIV-infected patients, may be partially explained by an interplay between immune mechanisms and genetic predisposition (e.g. slow versus rapid acetylators).
Diagnostic Features
Drug eruptions, both suspected and unsuspected, frequently lead to a dermatologic consultation, and it is often (although not always) possible to categorize a drug as having a high, medium or low probability of being the culprit. A logical approach begins with an accurate description of the skin lesions and their distribution, in addition to associated signs and symptoms ( Table 21.5 ). Data regarding all the drugs taken by the patient, including prescription, non-prescription/over-the-counter and complementary or alternative treatments, as well as the dates of administration and doses need to be collected. The chronology of drug administration is of paramount importance ( Table 21.6 ). The time between initiation of the drug and the onset of the skin eruption is a key element in identifying the offending drug, as most immunologically mediated reactions occur 8 to 21 days after initiation of a new medication.
| LOGICAL APPROACH TO DETERMINE THE CAUSE OF A DRUG ERUPTION | |
|---|---|
| Drug responsibility assessment | |
| Clinical characteristics |
|
| Chronological factors |
|
| Search of databases |
|
| CHARACTERISTICS OF MAJOR DRUG-INDUCED ERUPTIONS | ||||
|---|---|---|---|---|
| Clinical presentation | Percentage that are drug-induced (%) | Time interval | Mortality (%) | Selected responsible drugs |
| Exanthematous eruption | Child: 10–20 Adult: 50–70 | 4–14 days | 0 | Aminopenicillins Sulfonamides Cephalosporins Anticonvulsants (aromatic) Allopurinol Abacavir Nevirapine |
| Urticaria | <10 | Minutes to hours | 0 | Penicillins Cephalosporins NSAIDs Monoclonal antibodies Radiocontrast media † |
| Anaphylaxis | 30 | Minutes to hours | 5 | |
| Fixed drug eruption | 100 | First exposure: 1–2 weeks Re-exposure: <48 hours, usually within 24 hours | 0 | TMP-SMX NSAIDs Tetracyclines Pseudoephedrine * |
| Acute generalized exanthematous pustulosis (AGEP) | 70–90 | < 4 days | 1–2 | β-Lactam antibiotics Macrolides Calcium channel blockers |
| Drug reaction with eosinophilia and systemic symptoms (DRESS)/drug-induced hypersensitivity syndrome (DIHS) | 70–90 | 15–40 days | 5–10 | Anticonvulsants (aromatic) Lamotrigine (especially in combination with valproate) Sulfonamides Abacavir Allopurinol Dapsone Minocycline Nevirapine |
| Stevens–Johnson syndrome | 70–90 | 7–21 days | 5 | Sulfonamides Anticonvulsants (aromatic) Lamotrigine Allopurinol NSAIDs NNRTIs, e.g. nevirapine |
| Toxic epidermal necrolysis | 30 | |||
† Often anaphylactoid reaction.
Evolution after drug withdrawal may be helpful, as the cutaneous eruption usually clears when the suspected drug is discontinued. However, this assessment may prove difficult in the case of drugs with a long half-life or “persistent” drug reactions such as lichenoid and photoallergic eruptions or drug-induced pemphigus foliaceus and subacute cutaneous lupus erythematosus.
The suspect drug should be withdrawn as soon as possible. The usual practice is to discontinue all drugs that are non-essential. However, in some instances, it is necessary to weigh the risks versus the benefits of each drug and to determine if a similar-acting, but non-cross-reactive, drug is available as a substitute.
In the process of identifying the responsible drug, access to drug databases is very helpful . However, new or unusual drug reactions may not be identified. Moreover, the drug most frequently associated with adverse reactions may be innocent in a particular patient, and the physician dealing with a suspected drug reaction must remain open-minded.
With the exception of assays for IgE antibodies, diagnostic or confirmatory assays to establish the responsible drug are not available. A number of in vitro tests have been designed, including the histamine release test, migration inhibition factor test, lymphocyte toxicity assay, lymphocyte transformation test, and basophil degranulation test . However, their sensitivity and specificity have not been assessed in a reliable way with relevant controls. As a result, they are of limited value in the clinical setting.
Results of patch testing, in which drugs (usually with petrolatum or alcohol as the vehicle) are applied to the upper back for 48 hours, vary depending upon the responsible drug and the type of eruption ( Table 21.7 ). Prick and intradermal tests can be performed in patients with urticaria and angioedema, but are contraindicated in SJS/TEN due to the risk of relapse . Of note, delayed reading of prick, intradermal, and patch tests is particularly important in the case of amoxicillin-induced morbilliform eruptions. Unfortunately, investigations involving series of patients with several different types of drug reactions have shown heterogeneous results. In general, these tests, when positive, may be helpful in preventing readministration of the offending drug, but with the exception of prick and intradermal tests in patients with urticaria and perhaps patch tests in a few disorders (see Table 21.7 ), their specificity and/or sensitivity are low.
| CUTANEOUS DRUG ERUPTIONS – USE OF PATCH TESTING TO IDENTIFY THE RESPONSIBLE DRUG | ||
|---|---|---|
| Type of cutaneous drug reaction | % Patch test positivity * | Comments |
| Symmetrical drug-related intertriginous and flexural exanthema (SDRIFE) | 50–80 | Positive results most frequently observed with β-lactam antibiotics, clindamycin, erythromycin, pseudoephedrine, contrast agents > allopurinol, H1 blockers |
| Acute generalized exanthematous pustulosis (AGEP) | 50–60 | Positive results most frequently observed with β-lactam antibiotics and pristinamycin > carbimazole/metamizole, ciprofloxacin, clindamycin, diltiazem, metronidazole, morphine, NSAIDs, and pseudoephedrine |
| Fixed drug eruption | >40 | In situ patch test recommended, i.e. at the site of a previous lesion |
| Drug reaction with eosinophilia and systemic symptoms (DRESS) | 30–60 | Should be performed at least 6 months after clinical resolution Positive results primarily observed with abacavir, carbamazepine, proton pump inhibitors, and antituberculous drugs |
| Exanthematous (morbilliform) drug eruption | 10–40 | If patch test negative, prick tests can be discussed to increase positivity, depending upon disease severity |
| SJS/TEN | 10–25 | Reports of positive results with β-lactam antibiotics, carbamazepine, trimethoprim–sulfamethoxazole, and pseudoephedrine |
Rechallenge carries the risk of inducing a more severe reaction, thus limiting its use for both ethical and medico-legal reasons. Furthermore, the recurrence rate is not 100% with rechallenge (e.g. there are refractory periods) and a negative result may give an erroneous sense of security. Even with these limitations, in patients with fixed drug eruptions, topical provocation or rechallenge may prove helpful.
Clinical Features
Urticaria, Angioedema, and Anaphylaxis
In urticaria, there is vasodilation and transient edema within the dermis whereas in angioedema, the edema is present in deep dermal, subcutaneous, and submucosal tissues. Although several different mechanisms may be responsible, dramatic outbreaks of acute urticaria usually represent an immediate hypersensitivity reaction mediated by IgE antibodies, especially when the urticaria is associated with angioedema and/or anaphylaxis. Clinically, “anaphylactoid” reactions may mimic IgE-induced histamine release, but are secondary to a non-immunologic liberation of histamine and/or other mediators of inflammation.
Urticaria (see Ch. 18 for details)
Urticaria presents as transient, often pruritic, erythematous and edematous papules and plaques that may appear anywhere on the body, including the palms, soles, and scalp. Lesions can vary significantly in size and number and may assume a figurate configuration ( Fig. 21.2 ). The primary effector cell is the cutaneous mast cell which releases histamine and other inflammatory mediators (see Fig. 18.4 ) .
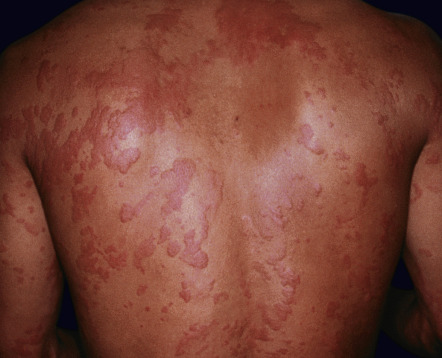
Although drugs are thought to be responsible for <10% of all cases of urticaria, they are more often associated with acute rather than chronic urticaria. That said, patients with chronic urticaria should avoid acetylsalicylic acid (ASA; aspirin), as well as other NSAIDs, as they can lead to an exacerbation.
In IgE-mediated urticaria, lesions typically appear within minutes to less than an hour after drug administration, especially when there has been prior sensitization. Both immunologic assays, such as radioallergosorbent tests (RAST) that detect specific IgE antibodies, and skin tests (prick tests) can prove useful in confirming the diagnosis. However, the number of drugs for which there are commercially available assays is limited, consisting primarily of penicillin, aminopenicillin, cephalosporin, and insulin. Of course, prick tests should be performed under appropriate medical supervision due to the risk of an anaphylactic reaction. It should also be noted that in some series, only 10–20% of patients who reported a history of penicillin allergy were truly allergic when assessed by skin testing.
Drugs that most frequently produce immunologically-based urticaria are antibiotics, especially penicillins and cephalosporins, and less often, sulfonamides and minocycline. As the use of monoclonal antibodies for neoplastic and inflammatory diseases increases, so will cases of urticaria (and vasculitis; see below) due to these exogenous proteins (see Ch. 128 ).
In anaphylactoid reactions, vasodilation results from the liberation of large amounts of histamine, bradykinin, and/or leukotrienes. Acetylsalicylic acid is the classic example of a drug that induces an anaphylactoid reaction and it does so via cyclooxygenase inhibition and subsequent accumulation of leukotrienes. The majority of urticarial reactions to radiocontrast media are also non-immunologic ( Table 21.8 ), as are many, but not all, reactions to NSAIDs (e.g. ibuprofen, naproxen). Allergic reactions to latex in gloves or medical devices can induce local or generalized urticaria, especially in the case of direct contact with mucosal surfaces (see Chs 16 & 18 ).
| ADVERSE REACTIONS TO RADIOCONTRAST MEDIA |
| Iodinated |
|
| Gadolinium |
| Urticaria, erythema (rare) Anaphylactoid reactions, anaphylaxis (rare) Nephrogenic systemic fibrosis (in the setting of renal insufficiency; see Ch. 43 ) |
The most important step in the treatment of drug-induced urticaria is withdrawal of the causative agent. Treatment consists primarily of H 1 antihistamines. Additional therapeutic options are discussed in Chapter 18 as is the differential diagnosis which includes the immune complex-mediated disorders urticarial vasculitis and serum sickness.
Angioedema (see Ch. 18 for details)
Angioedema is a reflection of transient edema of the deep dermal, subcutaneous and submucosal tissues. It is associated with urticaria in 50% of cases and may be complicated by life-threatening anaphylaxis. Angioedema occurs in 1 to 2 per 1000 new users of angiotensin-converting enzyme (ACE) inhibitors and is due to an accumulation of bradykinin (see Fig. 18.6 ). The most severe cases of angioedema may start within a few minutes after drug administration. However, in the case of ACE inhibitor-induced angioedema, lesions may appear from 1 day to several years after starting the drug; most appear within the first year. African-Americans and women are at increased risk for developing ACE inhibitor-induced angioedema .
The most common clinical presentation is an acute, asymmetric, pale or pink, subcutaneous swelling involving the face. Involvement of the oropharynx, larynx, and epiglottis can lead to impaired swallowing and stridor. Occasionally, in drug-induced angioedema, there is edema of the intestinal wall with abdominal pain, nausea, vomiting, and diarrhea.
The major drugs implicated in angioedema, besides penicillins and ACE inhibitors, are NSAIDs, radiographic contrast media and, more recently, monoclonal antibodies (see Ch. 128 ). Although angiotensin II receptor antagonists do not increase levels of bradykinin, they are also associated with angioedema, albeit less frequently. It is important to note that drug-induced angioedema may actually represent an unmasking of another cause for angioedema, e.g. acquired C1 inhibitor deficiency due to autoimmune or lymphoproliferative disorders.
Anaphylaxis
Anaphylaxis consists of an acute life-threatening reaction that occurs within minutes of drug administration, usually parenteral. It occurs in about 1 per 5000 exposures to penicillin and combines skin signs (urticaria and/or angioedema) with systemic manifestations such as hypotension and tachycardia. Occasionally, there is hypotension in the absence of cutaneous lesions. In severe cases, the patient becomes unconscious as a result of cardiovascular shock and may die. Prompt discontinuation of the offending drug is mandatory, as is strict avoidance of the drug in the future. Subcutaneous epinephrine (adrenaline) and systemic corticosteroids are the primary treatments for life-threatening angioedema and anaphylaxis, along with careful monitoring. Of note, patients taking β-blockers may have a limited response to epinephrine.
The most frequently incriminated drugs are antibiotics, in particular the penicillins/aminopenicillins but also cephalosporins and quinolones; in one series, additional causes were muscle relaxants (e.g suxamethonium), acetaminophen, and gadolinium-based contrast media . Anaphylaxis can also be seen following exposure to latex (see Ch. 16 ) while anaphylactoid reactions are usually seen with NSAIDs and radiocontrast media (see Table 21.8 ). Rarely, anaphylaxis occurs following cutaneous injections (e.g. local anesthetics) or topical applications of medications (e.g. bacitracin, chlorhexidine).
Exanthematous Drug Eruptions
▪ Morbilliform drug eruption ▪ Maculopapular drug eruption ▪ Urticarial drug eruption
The primary underlying pathomechanisms are most likely immunologic, complex, and cell-mediated. Several mechanisms have been proposed (see above) in which the drug or drug-peptide hapten presented by dendritic cells to T lymphocytes can either bind covalently or non-covalently to MHC molecules . CD4 + and CD8 + T cells that strongly express perforin and granzyme are then recruited and their cytotoxic activity leads to death of keratinocytes.
Overall, most drug classes can induce an exanthematous eruption in ~1% of treated patients. Higher risk medications (>3% of treated patients develop an eruption) include aminopenicillins, allopurinol, sulfonamides, cephalosporins, and aromatic anticonvulsants. Certain viral infections are also known to increase the incidence of drug reactions. Depending upon the series, the frequency of aminopenicillin-induced exanthematous eruptions in patients with infectious mononucleosis ranges from 33% to 100% (see Ch. 80 ). One theory is that reactive drug metabolites disturb the balance between cytotoxic and regulatory immune responses, leading to a cytotoxic reaction that targets virally infected keratinocytes.
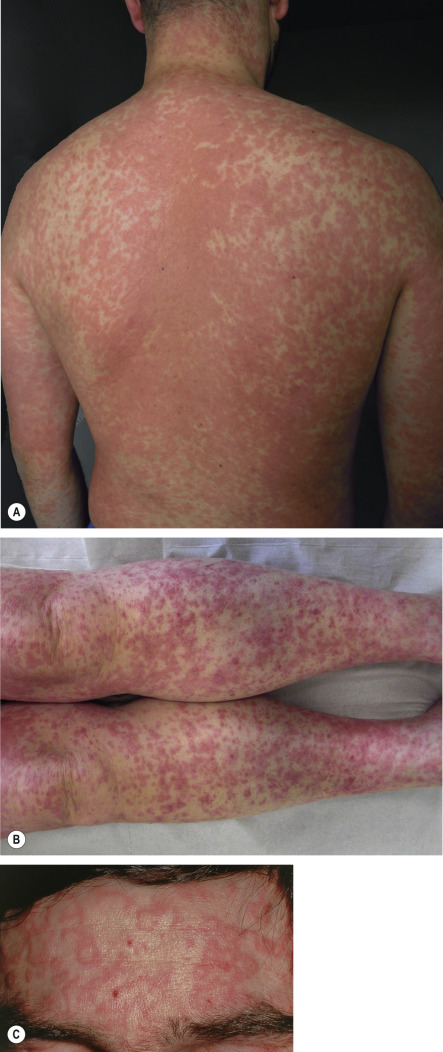
A morbilliform eruption classically begins 7 to 14 days after the initial drug administration, but appears earlier in the case of rechallenge. Symmetrically distributed erythematous macules, papules and/or urticarial lesions initially appear on the trunk and upper extremities; over time they can become confluent ( Fig. 21.3A ). The eruption is typically more polymorphous than a viral exanthem. Sometimes, due to dependency, the lesions on the distal lower extremities become petechial or purpuric ( Fig. 21.3B ). Mucous membranes are usually spared but pruritus and a low-grade fever are often present. There may also be annular plaques ( Fig. 21.3C ) or atypical “target” lesions, leading to a misdiagnosis of erythema multiforme. Once the offending drug is discontinued, the eruption gradually resolves over one to two weeks, without complications and/or sequelae. However, for 1 to 3 days immediately following discontinuation of the responsible medication, an increase in extent and intensity may be observed.
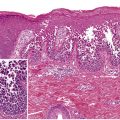
Signs and symptoms that point to the possibility of a more severe drug-induced eruption (see Table 21.1 ) include edema of the face, pustules, vesicles, dusky or painful lesions, skin fragility, mucous membrane involvement, and marked peripheral blood eosinophilia. Histopathologically, nonspecific findings are typically seen in morbilliform drug eruptions, i.e. a mild superficial perivascular and interstitial lymphocytic infiltrate that may contain eosinophils (up to 70% of cases) in addition to interface changes .
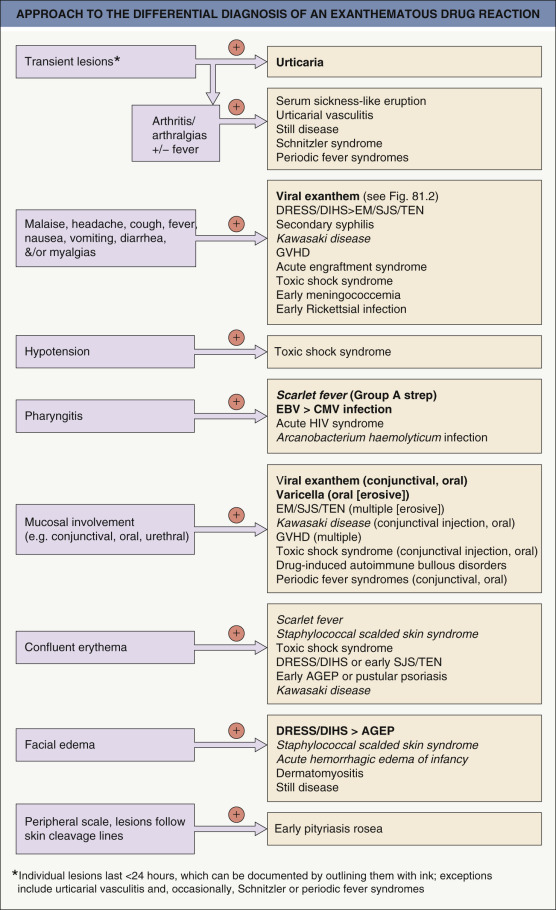
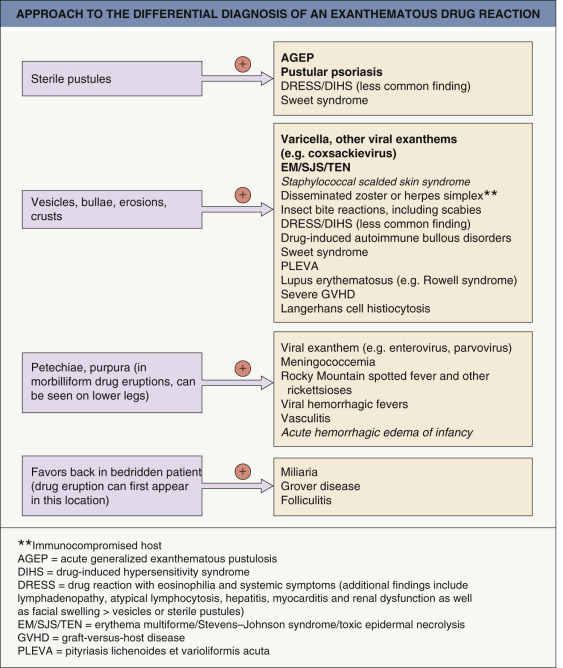
The major entity in the differential diagnosis of a morbilliform drug eruption is a viral exanthem (e.g. Epstein–Barr virus [EBV], enterovirus, adenovirus, early HIV infection, human herpesvirus type 6 [HHV-6]; see Fig. 81.2 ). Peripheral blood eosinophilia and a polymorphous appearance point to a drug eruption, and in the absence of definitive evidence, drug eruptions are favored in adults whereas viral exanthems are favored in children. Occasionally, there is a viral infection that enhances the risk of developing a drug eruption (see above) . Toxic shock syndromes, scarlet fever, acute GVHD, Kawasaki disease, and SCARs should be excluded on the basis of associated clinical features ( Fig. 21.4 ).
Treatment is largely supportive. Topical antipruritics and corticosteroids may help to alleviate pruritus. Discontinuing the offending agent is the first therapeutic intervention. “Treating through”, i.e. continuing the drug despite the cutaneous eruption, can be considered when the suspected drug is of paramount importance for the patient and there is no satisfactory substitute drug. Usually, the eruption will disappear, but a few patients may experience progressive worsening, leading to erythroderma. Desensitization may be considered in HIV-infected patients who require sulfonamides.
Drug Reaction With Eosinophilia and Systemic Symptoms (DRESS)
▪ Drug-induced hypersensitivity syndrome (DISH) ▪ Drug-induced delayed multi-organ hypersensitivity syndrome (DIDMOHS)
| RegiSCAR SCORING SYSTEM FOR DRESS | |||
|---|---|---|---|
| Criteria | No | Yes | Unknown/unclassifiable |
| Fever (≥38.5°C) | −1 | 0 | −1 |
| Lymphadenopathy (≥2 sites; >1 cm) | 0 | 1 | 0 |
| Circulating atypical lymphocytes | 0 | 1 | 0 |
| Peripheral hypereosinophilia | 0 | 0 | |
| 0.7–1.499 × 10 9 /L – or – 10–19.9% * | 1 | ||
| ≥1.5 × 10 9 /L – or – ≥20% * | 2 | ||
| Skin involvement | |||
| 0 | 1 | 0 |
| −1 | 1 | 0 |
| −1 | 0 | 0 |
| Internal organs involved † | 0 | 0 | |
| One | 1 | ||
| Two or more | 2 | ||
| Resolution in ≥15 days | −1 | 0 | −1 |
| Laboratory results negative for at least three of the following (and none positive): (1) ANA; (2) blood cultures; (3) HAV/HBV/HCV serology; and (4) Chlamydia and Mycoplasma serology | 0 | 1 | 0 |
| Final score: < 2, no case; 2–3, possible case; 4–5, probable case; >5, definite case | |||
* If leukocytes <4.0 × 10 9 /L
** At least two of the following: edema, infiltration, purpura, scaling.
† Liver, kidney, lung, muscle/heart, pancreas, or other organ and after exclusion of other explanations.
| J-SCAR DIAGNOSTIC CRITERIA FOR DIHS/DRESS |
|
* This can be replaced by other organ involvement, such as renal involvement.
Although the precise pathogenesis of DRESS is still not fully understood, several underlying mechanisms have been proposed, including a specific alteration in the metabolism of particular drugs. For example, genetic polymorphisms that affect detoxification of anticonvulsants and sulfonamides have been identified in patients recovering from DRESS . For aromatic anticonvulsants, the inability to detoxify toxic arene oxide metabolites is probably a key factor and would provide an explanation for the cross-reactivity between phenytoin, carbamazepine, and phenobarbital which has been well documented, both in vivo and in vitro .
Immune mechanisms have also been implicated based upon several observations including the requirement for sensitization, positive skin tests for the culprit drug in some patients, and a shorter time-to-onset upon rechallenge . Notably, distinct HLA alleles have been associated with a significantly increased risk of developing drug-specific DRESS (see Table 21.4 ) . In addition, IL-5 plays a role in the generation of eosinophilia and drug-specific T cells, activated in the skin and internal organs, serve to mediate the disorder.
Also implicated in the pathogenesis of DRESS is reactivation of human herpes viruses, primarily HHV-6 and HHV-7, but also CMV and EBV . In patients with DRESS, transmission of HHV-6 to skin-infiltrating CD4 + T cells (and subsequent replication) was found to require recruitment of HHV6 + peripheral monomyeloid cells to damaged skin . A study of 40 patients with DRESS noted reactivation of EBV, HHV-6, or HHV-7 in 76% of patients; activated circulating CD8 + T cells with cutaneous homing markers secreted large amounts of TNF-α and IFN-γ, especially in those with the most severe visceral involvement . In patients’ EBV-transformed B cells, culprit drugs triggered the production of EBV .
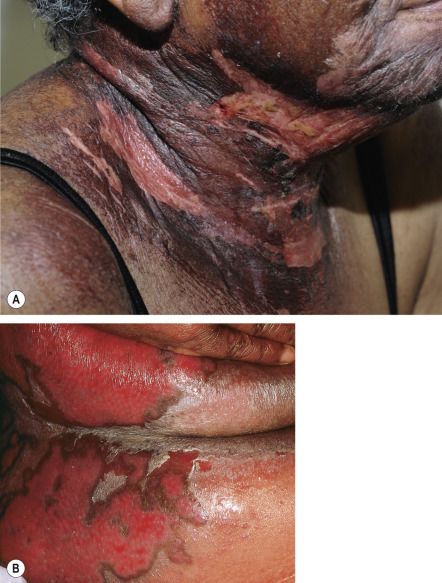
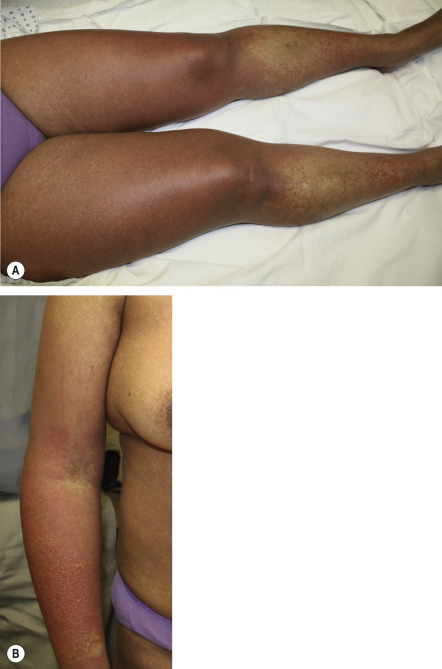
Clinically, DRESS develops 2 to 6 weeks after drug initiation, i.e. later than most other immunologically mediated skin reactions. With re-exposure, there can be a shorter time to onset. Fever and a cutaneous eruption are the most common symptoms, seen in 85% and 75% of patients, respectively . Cutaneous involvement usually begins as a morbilliform eruption ( Fig. 21.5A ), which later becomes edematous, often with a follicular accentuation. Less common manifestations include vesicles ( Fig. 21.5B ), follicular or non-follicular pustules (~20% of patients), erythroderma, and purpuric lesions. The face, upper trunk, and extremities are usually the initial sites of involvement. Edema of the face is a frequent finding and is a hallmark of DRESS whereas mucosal involvement, if present, is mild.
Internal manifestations include lymphadenopathy and hepatic involvement (~80% of patients); rarely, the latter may become life-threatening (see Tables 21.9 and 21.10 ). Patients may develop interstitial nephritis, myocarditis, interstitial pneumonitis, myositis, thyroiditis, and even infiltration of the brain by eosinophils. The cutaneous and visceral involvement may persist for several weeks or months after drug withdrawal, and additional sites of involvement (e.g. cardiac, thyroid) may develop weeks or months later, including following a taper of corticosteroids. Overall mortality due to DRESS ranges from 2 to 10% .
Histopathologically, various inflammatory patterns can be seen, including eczematous, interface dermatitis, AGEP-like, and erythema multiforme-like. Prominent peripheral blood eosinophilia is common and is a very characteristic feature. It is often accompanied by mononucleosis-like atypical lymphocytosis. Elevation of hepatic enzymes can be a worrisome finding and requires serial evaluation. Thyroid and cardiac dysfunction (as detected by an ECG and echocardiogram) may develop as delayed complications and patients should therefore undergo longitudinal evaluation ( Table 21.11 ).
| ASSESSMENT AND LONGITUDINAL EVALUATION OF PATIENTS WITH DRESS (DRUG REACTION WITH EOSINOPHILIA AND SYSTEMIC SYMPTOMS) |
| Basic laboratory screening during the acute phase with recommended repetitive tests in italics ^ |
|
| Additional testing |
|
| Further testing based upon laboratory abnormalities or signs and symptoms ** |
|
^ Testing is more frequent during the acute phase (e.g. twice weekly) with frequency also a reflection of disease severity. Longitudinal evaluation is recommended for at least one year.
* Allows for immediate assessment for proteinuria.
The differential diagnosis includes other cutaneous drug eruptions, acute viral infections, hypereosinophilic syndrome, lymphoma, and pseudolymphoma . Involvement of multiple internal organs differentiates DRESS from the more common morbilliform eruptions. In addition to the most common etiologies – the aromatic anticonvulsants (phenobarbital, carbamazepine and phenytoin), lamotrigine (especially when coadministered with valproic acid), and sulfonamides – minocycline, allopurinol, and dapsone may also induce this syndrome, as well as drugs used to treat HIV infection, e.g. abacavir ( Table 21.12 ).
| DRUGS ASSOCIATED WITH DRUG REACTION WITH EOSINOPHILIA AND SYSTEMIC SYMPTOMS SYNDROME (DRESS) | |
|---|---|
| Drug category | Specific drugs |
| Anticonvulsants | Carbamazepine, lamotrigine * , phenobarbital, phenytoin , oxcarbazepine, zonisamide > valproic acid |
| Antimicrobials | Ampicillin, cefotaxime, dapsone , ethambutol, isoniazid, linezolid, metronidazole, minocycline , pyrazinamide, quinine, rifampin, sulfasalazine (salazosulfapyridine), streptomycin, trimethoprim–sulfamethoxazole , teicoplanin, vancomycin |
| Antiretrovirals | Abacavir , nevirapine , zalcitabine |
| Antidepressants | Bupropion, fluoxetine |
| Antihypertensives | Amlodipine, captopril |
| NSAIDs | Celecoxib, ibuprofen |
| Miscellaneous | Allopurinol ** , a zathioprine , imatinib, mexiletine, ranitidine, ziprasidone |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree