Dermatology has always been a surgically oriented specialty. Although procedures such as curettage, biopsy, destruction, and excision have been key components of the field, the practice has evolved to include a greater number and extent of surgical procedures. This progression can be attributed to a variety of factors. Dermatologists have a greater understanding of cutaneous pathology, which places them in a unique role to manage complex surgical procedures that arise in the skin. In addition, outpatient dermatologic surgery has been shown to be cost-effective, safe, and efficacious, delivering a greater degree of patient convenience, particularly compared with other fields. The American Board of Dermatology therefore mandates surgical exposure and experience for all residents in dermatology residency programs. Furthermore, with the Accreditation Council for Graduate Medical Education (ACGME) accreditation of Micrographic Surgery and Dermatologic Oncology fellowships, dermatologic surgery has become recognized as a mainstream medical option for patients. This chapter and Chapters 38 and 39 provide a survey of procedures, indications, and appropriate management within the spectrum of the dermatologic surgery field.
Hanke CW, et al: Current status of surgery in dermatology. J Am Acad Dermatol 2013; 69: 972.
Hansen TJ, et al: Patient safety in dermatologic surgery. J Am Acad Dermatol 2015; 73: 1.
Preparation for Surgery
A thorough and complete preoperative evaluation is required before performing any surgical procedure. A detailed medical history must be obtained, including information on drug allergies, current medications (including herbal or natural supplements), smoking/tobacco status, presence of a pacemaker or implantable cardioverter/defibrillator, recently implanted prosthetic devices, history of prior wound infection or perioperative bleeding, and history of endocarditis or cardiac valvular or congenital malformation.
Anticoagulants
Much has been written regarding the role of antithrombotic agents (including both antiplatelet and anticoagulant medications) and surgical bleeding. Dermatologists are frequently presented with the dilemma of whether to discontinue blood thinners in the setting of surgery. Data and multiple reviews have shown that continuous treatment with blood thinners perioperatively in patients undergoing Mohs and cutaneous surgery is not associated with an increase in surgical complications leading to significant morbidity. In contrast, discontinuation of these medications may increase the risk of catastrophic cerebral and cardiovascular complications. Multiple authors believe that the potential adverse effects of discontinuing essential medical blood thinners far outweigh the potential side effects of surgical bleeding (e.g., managing a postoperative hematoma). In fact, despite some surgeons’ claims to the contrary, studies have demonstrated that blinded surgeons are unable to identify intraoperatively which patients are taking anticoagulation medication based on the subjective amount of surgical oozing. As such, it is recommended that patients be maintained on all medically necessary blood thinners during cutaneous surgery. In contrast, patients taking aspirin for primary prevention may discontinue use 2 weeks before any surgical procedure.
Herbal supplements are becoming increasingly popular with patients who are looking for a “natural” option to traditional medication. Patients may not readily volunteer that they are taking these supplements, either because they do not characterize supplements as medication or because they are concerned that physicians will not be accepting of alternative treatments. Therefore physicians should ask patients specifically if they are taking any supplements. Ginkgo, garlic, ginseng, ginger, and vitamin E may increase the risk of perioperative bleeding. These herbal supplements are not medically necessary, so patients should discontinue them for several weeks before undergoing dermatologic surgery.
Brown DG, et al: A review of traditional and novel oral anticoagulant and antiplatelet therapy for dermatologists and dermatologic surgeons. J Am Acad Dermatol 2015; 72: 524.
Callahan S, et al: The management of antithrombotic medication in skin surgery. Dermatol Surg 2012; 38: 1417.
Chang TW, et al: Complications with new oral anticoagulants dabigatran and rivaroxaban in cutaneous surgery. Dermatol Surg 2015; 41: 784.
Dinehart SM, Henry L: Dietary supplements: altered coagulation and effects on bruising. Dermatol Surg 2005; 31: 819.
Gill JF, et al: Tobacco smoking and dermatologic surgery. J Am Acad Dermatol 2013; 68: 167.
O’Neill JL, et al: Postoperative hemorrhage risk after outpatient dermatologic surgery procedures. Dermatol Surg 2014; 40: 74.
Otley CC: Perioperative evaluation and management in dermatologic surgery (review). J Am Acad Dermatol 2006; 54: 119.
Palamaras I, Semkova K: Perioperative management of and recommendations for antithrombotic medications in dermatological surgery. Br J Dermatol 2015; 172: 597.
Plovanich M, Mostaghimi A: Novel oral anticoagulants: what dermatologists need to know. J Am Acad Dermatol 2015; 72: 535.
West SW, et al: Cutaneous surgeons cannot predict blood-thinner status by intraoperative visual inspection. Plast Reconstr Surg 2002; 110: 98.
Zwiebel SJ, et al: The incidence of vitamin, mineral, herbal, and other supplement use in facial cosmetic patients. Plast Reconstr Surg 2013; 132: 78.
Surgical Site Infection
As a general rule, wound infections associated with skin surgery procedures occur at an extremely low rate. There is an extensive literature documenting the safety and overall low infection rate of skin procedures (ranging from surgical excisions, to laser, to liposuction). Postoperative wound infections, when they do occur, most commonly appear in the first 4–10 days after the procedure. In some cases, making a proper diagnosis of infection can be challenging, with other conditions being mimickers (e.g., allergic contact dermatitis or fibrin deposition over the granulation wound).
The Centers for Disease Control and Prevention defines a surgical site infection as occurring within 30 days of the procedure and involving only skin and subcutaneous tissue of the incision. In addition, at least one of the following criteria should also be present: (1) purulent drainage from the incision site, (2) organism isolation from culture of the fluid drainage or tissue, (3) pain/tenderness or localized swelling with warmth and redness, or (4) clinical diagnosis of infection by the physician.
Treatment of Wound Infection
Postoperative surgical site infection is uncommon in dermatologic surgery procedures, with an incidence of 1%–3%. Infections typically present 4–7 days after surgery with increased erythema, tenderness, warmth, and purulent drainage. Sutures can be removed to allow for drainage of exudate. In cases where infection leads to dehiscence, the wound can be packed or allowed to heal by second intention. Scar revision can be performed at a later date should that be necessary. A culture should be performed before initiating empiric antibiotics to determine sensitivities.
Staphylococcus aureus is the most common pathogen, and cephalexin or dicloxacillin is an appropriate first-line treatment. Patients with a penicillin allergy can be treated with clindamycin. Although this antibiotic has been associated with colitis, the short courses of clindamycin typically used with surgical site infection generally do not present a problem. In communities or institutions with a high incidence of methicillin-resistant S. aureus (MRSA), antibiotic choice can be modified based on community sensitivities (e.g., doxycycline, trimethoprim-sulfamethoxazole [TMP-SMX]). Ciprofloxacin can be used for infections with a higher likelihood of gram-negative or Pseudomonas organisms (e.g., ear). Antibiotic choice should be modified based on culture results.
The use of topical antibiotics after routine skin surgery has not been shown to reduce the incidence of surgical site infection. Several studies have demonstrated that petrolatum was as effective as topical antibiotics in terms of incidence of infection. In addition, many patients on topical antibiotics had a higher incidence of allergic contact dermatitis. Finally, studies have shown the overall cost saving of switching from postoperative bacitracin to petrolatum. As such, routine use of topical antibiotics ought to be avoided in the postoperative period.
Antibiotic Prophylaxis
Dermatologists performing cutaneous surgery are often faced with the decision of whether to prescribe prophylactic antibiotics. The main issues surrounding antibiotic prophylaxis are prevention of surgical site infections and reduction of the risk of endocarditis or contamination of prosthetic devices in high-risk patients. As a general rule, wound infections associated with skin surgery procedures occur at an extremely low rate. Although reducing infection is one objective in the use of antibiotics, dermatologists must consider the risks of such treatment, including adverse drug reactions, serious drug reactions, drug interactions, development of resistant strains of bacteria, and increased cost.
Determining the indications for antibiotic prophylaxis for surgical site infections requires an understanding of the various types of wound that the dermatologist may encounter. Wounds can be categorized into the following four groups:
- 1.
Clean wounds (class I) are created on normal skin using clean or sterile technique. Examples include excision of neoplasms, noninflamed cysts, biopsies, and most cases of Mohs surgery. The majority of dermatologic surgery falls into this category. The infection rate of these wounds is less than 5%. Of note, this incidence is based on general surgery cases, which are often of longer duration and a greater extent than most dermatologic procedures. This explains the lower actual infection rate in dermatologic surgery, which is in the 1%–3% range.
- 2.
Clean-contaminated wounds (class II) are created on contaminated skin or any mucosal or moist intertriginous surface, such as the oral cavity, upper respiratory tract, axilla, or perineum. The infection rate of these wounds is 10%.
- 3.
Contaminated wounds (class III) involve visibly inflamed skin with or without nonpurulent discharge and have an infection rate of 20%–30%. Examples included inflamed cysts or traumatic wounds.
- 4.
Infected wounds (class IV) have contaminated foreign bodies, purulent discharge, or devitalized tissue. Examples included necrotic tumors, ruptured cysts, or active hidradenitis suppurativa. These wounds have an infection rate of 40%.
Clean (class I) wounds, which constitute the vast majority of dermatologic surgery procedures, do not require antibiotic prophylaxis.
Although antibiotic prophylaxis in clean-contaminated (class II) wounds is not a clear issue, most cases do not require routine antibiotics. It is preferable to treat infections should they arise (because these are not a common occurrence, even in class II wounds), rather than expose all patients to antibiotics and the increased rate of drug-related adverse events. Some exceptions to this that have been advocated include surgical cases that violate mucosal membranes (oral, nasal, anogenital) and patients with heavily colonized skin (atopic dermatitis, infected skin), as well as those in whom a wound infection would result in significant morbidity. However, dermatologic surgeons do not universally agree on these exceptions, and the role for antibiotic prophylaxis is still debated.
In contaminated (class III) and infected (class IV) wounds, antibiotics serve a therapeutic, rather than a prophylactic, role and should be used routinely in these cases.
Antibiotic Selection and Timing
To achieve optimal prophylaxis, antibiotics should be in the bloodstream, and thus at the surgical site, at the time of incision. Antibiotics given at the conclusion of the procedure are not as effective in preventing infection, because they are not incorporated into the coagulum of the wound. Once the surgical wound is closed, the risk of infection decreases significantly. Most dermatologic procedures are of short duration, so a single preoperative dose of antibiotics 1 hour before the start of the case is sufficient. In rare cases with an extended dermatologic procedure, a second dose of antibiotics can be administered 6 hours postoperatively.
The choice of antibiotic is based on the most likely causative organism at the surgical site ( Table 37.1 ). S. aureus is the most common wound infection in cutaneous surgery. Other pathogens to consider in some situations include Streptococcus viridans (oral mucosa) and Escherichia coli (perineal and genital location).
| Surgical Site | Antibiotic | Regimen (Single Dose 1 Hour Preoperatively) |
|---|---|---|
| Skin | Cephalexin | 1 g orally |
| Dicloxacillin | 1 g orally | |
| Clindamycin | 300 mg orally | |
| Vancomycin | 500 mg intravenously | |
| Oral and respiratory mucosa | Cephalexin Amoxicillin | 1 g orally 1 g orally |
| Clindamycin | 300 mg orally | |
| Gastrointestinal and genitourinary mucosa | Cephalexin Trimethoprim-sulfamethoxazole | 1 g orally 1 double-strength tablet orally |
| Ciprofloxacin | 500 mg orally |
First-generation cephalosporins are an ideal initial choice for the treatment of wound infection because of their coverage of staphylococcal organisms, common gram-negative organisms such as E. coli, and certain Proteus species. Cephalosporins are rapidly absorbed when taken orally and have good tissue penetration. Their estimated cross-reactivity in penicillin-allergic patients is 5%–10%.
Isoxazolyl penicillins, such as dicloxacillin and nafcillin, can also be used because they provide coverage for most strains of streptococci and β-lactamase–producing bacterial strains, such as S. aureus. Aminopenicillins, such as ampicillin and amoxicillin, have better gram-negative, enterococcal, and group A streptococcal coverage. However, aminopenicillins are not effective against β-lactamase–producing bacteria and thus are used more often in procedures involving oral mucosa.
Clindamycin, macrolides (e.g., erythromycin, azithromycin), TMP-SMX, and ciprofloxacin can all be considered in patients with a penicillin or cephalosporin allergy, with the specific choice based on the site of surgery and thus the presumed causative organism. Vancomycin is generally limited to those cases where MRSA is suspected, because it requires intravenous administration and adjustment in patients with impaired renal function.
Endocarditis Prophylaxis
The American Heart Association (AHA) updated its recommendations on infective endocarditis (IE) prophylaxis in 2007. The overall conclusions were that bacteremia from daily activities is much more likely to cause IE than bacteremia associated with dental procedures, and that far fewer patients are now recommended to have antibiotic prophylaxis. Antibiotic prophylaxis has been limited to patients with the conditions listed in Box 37.1 . All other cardiac conditions, including mitral valve prolapse and other forms of congenital heart disease, no longer require prophylaxis for any procedure.
Prosthetic cardiac valve or prosthetic material used for cardiac valve repair
Previous infectious endocarditis
Congenital heart disease (CHD) *
* Except for conditions listed above, antibiotic prophylaxis is not recommended for any other form of CHD.
- •
Unrepaired cyanotic CHD, including palliative shunts and conduits
- •
Completely repaired congenital heart defect with prosthetic material or device, during the first 6 months after the procedure
- •
Repaired CHD with residual defects at the site or adjacent to the site of a prosthetic patch or prosthetic device (which inhibit endothelialization)
- •
Cardiac transplantation recipients who develop cardiac valvulopathy
Antibiotic prophylaxis is reasonable when procedures involve manipulation of gingival tissue, perforation of oral mucosa, or incision or biopsy of the respiratory mucosa, or when performed on infected skin, but only in patients with underlying cardiac conditions associated with the highest risk of adverse outcome, as outlined in Box 37.1 . Antibiotic prophylaxis solely to prevent IE is not recommended for gastrointestinal or genitourinary procedures. The AHA reaffirmed its 1997 statement regarding medical procedures, including incision or biopsy of surgically scrubbed skin, that do not require antibiotic prophylaxis. Antibiotic prophylactic regimens for those select high-risk patients should be a single dose of antibiotic administered 1 hour before the procedure ( Table 37.2 ).
| Situation | Agent | Adults | Children |
|---|---|---|---|
| Able to take oral medication | Amoxicillin | 2 g | 50 mg/kg |
| Unable to take oral medication | Ampicillin | 2 g IM/IV | 50 mg/kg IM/IV |
| or | |||
| Cefazolin or ceftriaxone | 1 g IM/IV | 50 mg/kg IM/IV | |
| Allergic to penicillins or ampicillin and able to take oral medication | Cephalexin * | 2 g | 50 mg/kg |
| or | |||
| Clindamycin | 600 mg | 20 mg/kg | |
| or | |||
| Azithromycin or clarithromycin | 500 mg | 15 mg/kg | |
| Allergic to penicillins or ampicillin and unable to take oral medication | Cefazolin or ceftriaxone * | 1 g IM/IV | 50 mg/mg IM/IV |
| or | |||
| Clindamycin | 600 mg IM/IV | 20 mg IM/IV |
* Cephalosporins should not be used in patients with a history of anaphylaxis, angioedema, or urticaria with penicillins or ampicillin.
There are no formal guidelines regarding the use of antibiotics in patients with orthopedic prosthetic devices undergoing dermatologic surgery. However, guidelines for dental procedures in patients with joint replacement can be extrapolated to certain procedures. Patients with joint replacement probably do not need prophylactic antibiotics for clean wounds. If mucosa is invaded, prophylaxis may be appropriate and reasonable in the small number of patients who might be at high risk of joint infection. Consultation with orthopedic surgery is appropriate in determining whether antibiotic prophylaxis is necessary.
American Dental Association, American Academy of Orthopedic Surgeons: Antibiotic prophylaxis for dental patients with total joint replacements. J Am Dent Assoc 2003; 134: 895.
Bae-Harboe YS, Liang CA: Perioperative antibiotic use of dermatologic surgeons in 2012. Dermatol Surg 2013; 39: 1592.
Levender MM, et al: Use of topical antibiotics as prophylaxis in clean dermatologic procedures. J Am Acad Dermatol 2012; 66: 445.
Lilly E, Schmults CD: A comparison of high- and low-cost infection-control practices in dermatologic surgery. Arch Dermatol 2012; 148: 859.
Maragh SL, et al: Antibiotic prophylaxis in dermatologic surgery. Dermatol Surg 2005; 31: 83.
Maragh SL, Brown MD: Prospective evaluation of surgical site infection rate among patients with Mohs micrographic surgery without the use of prophylactic antibiotics. J Am Acad Dermatol 2008; 59: 275.
Rogers HD: Prospective study of wound infections in Mohs micrographic surgery using clean surgical technique in the absence of prophylactic antibiotics. J Am Acad Dermatol 2010; 63: 842.
Rossi AM, Mariwalla K: Prophylactic and empiric use of antibiotics in dermatologic surgery. Dermatol Surg 2012; 38: 1898.
Saco M, et al: Topical antibiotic prophylaxis for prevention of surgical wound infections from dermatologic procedures: a systematic review and meta-analysis. J Dermatolog Treat 2015; 26: 151.
Wilson W, et al: Prevention of infective endocarditis. Circulation 2007; 116: 1736.
Wright TI, et al: Antibiotic prophylaxis in dermatologic surgery. J Am Acad Dermatol 2008; 59: 464.
Preoperative Antisepsis
Many surgical preparations are available for preoperative antisepsis. Alcohol is frequently used for minor clean procedures, such as biopsies. However, because it has only weak antimicrobial activity, alcohol is not recommended for more extensive procedures.
Chlorhexidine has a broad spectrum against gram-positive and gram-negative organisms, a rapid onset of activity, and sustained residual activity even after being wiped off, and it is nonstaining. Chlorhexidine has been reported to cause both ototoxicity and keratitis from direct tympanic or ocular contact. However, this is mainly in patients under general anesthesia who cannot respond to immediate irritation associated with ocular contact, a problem that is avoided in most dermatologic procedures performed under local anesthesia.
Betadine (povidone-iodine) and all iodine-containing preparations have an excellent bactericidal activity within several minutes of application. However, these agents are often irritating to the skin, leave a residual color, can be absorbed in premature infants, and must dry before the procedure if they are to act as an effective antimicrobial agent.
Hexachlorophene is not bactericidal against many gram-negative organisms. It has the potential for neurotoxicity in children and teratogenicity in pregnancy. Hydrogen peroxide has no significant antiseptic properties, and thus it is not suitable for sterile skin preparation.
If hair must be removed before surgery, this should be done in a manner that does not leave open skin (cuts or scratches), which can serve as a conduit for infection. Preoperative shaving has been associated with a higher rate of bacterial infection secondary to cutting of the skin surface.
Dumville JC, et al: Preoperative skin antiseptics for preventing surgical wound infections after clean surgery. Cochrane Database Syst Rev 2015; 4: CD003949.
Echols K, et al: Role of antiseptics in the prevention of surgical site infections. Dermatol Surg 2015; 41: 667.
Kamel C, et al: Preoperative skin antiseptic preparations for preventing surgical site infections. Infect Control Hosp Epidemiol 2012; 33: 608.
Anesthesia
Anesthetics work by blocking sodium influx into neurons and preventing depolarization and blockage of action potential ( ). Small, unmyelinated C fibers, which carry pain and temperature sensation, are more easily blocked than larger, myelinated A fibers, which carry pressure sensation and motor function. This difference translates clinically, with patients under local anesthesia not experiencing pain from the sharp incision, but still maintaining the sensation of pressure during the procedure.
All local anesthetics have a similar structure, consisting of three parts: an aromatic hydrophobic ring, intermediate chain, and amine end. The aromatic hydrophobic portion is lipophilic and facilitates diffusion through nerve cell membranes, correlating to the potency of the anesthesia. The hydrophilic amine end contributes to the aqueous solubility of the anesthetic and is involved in binding of the molecule to the sodium channel. The intermediate chain consists of either an amide or an ester. Amides are metabolized by hepatic microsomal enzymes, and esters are metabolized in plasma by pseudocholinesterase and excreted by the kidney.
The choice of anesthetic is based on a variety of factors, including patient allergy, renal or hepatic impairment, and type of procedure being performed. The “workhorse” anesthetic of dermatologic surgery is lidocaine, because of its rapid onset of action and intermediate duration of action. Longer-acting anesthetics, such as bupivacaine, have a delayed onset of action, but can be used in special procedures or in combination with lidocaine to maximize duration of anesthesia.
All local anesthetics, with the exception of cocaine and prilocaine, cause vasodilation from relaxation of smooth muscle. As a result, patients experience increased surgical bleeding and shorter duration of action as the anesthesia is cleared from the surgical site because of vasodilation. Epinephrine, which causes vasoconstriction, is often added to local anesthetics to decrease bleeding, increase duration of anesthesia, and reduce systemic side effects caused by systemic absorption. Concentrations of 1 : 100,000 to 1 : 400,000 are typically used, with lower concentrations having fewer side effects while still maintaining clinical efficacy. The vasoconstrictive effect of epinephrine takes 15 minutes for onset, so the surgeon must allow adequate time before starting the procedure. Epinephrine is a strong α- and β-adrenergic receptor agonist and has an absolute contraindication in hyperthyroidism and pheochromocytoma. Large amounts of epinephrine must be used cautiously in patients with severe hypertension or narrow-angle glaucoma, as well as in pregnancy. Patients taking β-blockers, monoamine oxidase inhibitors, tricyclic antidepressants, and phenothiazines are more sensitive to epinephrine. Although the subject of much controversy, epinephrine is safe to use in well-vascularized areas, such as the ear, nose, and genitals. Reports of necrosis are likely the result of excessive volume being placed, which can cause a physical tamponade of vessels, rather than being a direct result of epinephrine.
Sodium bicarbonate (8.4%) can be added (1 : 10 ratio) to reduce the pain and burning associated with the lower pH of lidocaine with epinephrine. However, sodium bicarbonate can reduce epinephrine activity with time, thus requiring freshly mixed preparations on a regular basis.
Side Effects
The most common side effect of local anesthetic is injection site pain. Buffering with sodium bicarbonate, using a small-gauge needle (e.g., 30 gauge), using ice or vibratory distraction at the injection site, injecting slowly into the subcutaneous tissue (rather than the dermis), warming the anesthesia, minimizing the number of injections, and placing subsequent injections in an already-anesthetized location can minimize the pain associated with local anesthesia. Vasovagal reactions are common during anesthesia administration. Patients should lie flat during the injection to reduce this occurrence. Cold compresses and placing the patient in a Trendelenburg position can help if symptoms occur.
Maximum dosage of anesthesia has traditionally been accepted as 5 mg/kg of 1% plain lidocaine and 7 mg/kg of 1% lidocaine with epinephrine. These numbers have been based on traditional industry-based studies, not found in the medical literature. Experience with tumescent liposuction has taught that dosages up to 55 mg/kg are well tolerated and safe in certain clinical situations. Bupivacaine has a greater risk of cardiac toxicity than lidocaine because of its longer duration of action.
Most true allergic reactions to local anesthetics have been reported with esters. The metabolite p -aminobenzoic acid (PABA) is responsible for ester allergies. There is no cross-reactivity between ester and amide classes of anesthetics, so allergy to one type does not preclude the use of the other. True systemic amide allergy is extremely rare. Thorough questioning of patients who report allergy often reveals a vasovagal reaction or epinephrine sensitivity. If local anesthetic use is precluded, intradermal injection with diphenhydramine can be used. Drowsiness can be a side effect when large doses of this agent are used. Bacteriostatic saline, with the benzyl alcohol preservative acting as the anesthetic agent, is often sufficient to provide the brief anesthesia needed to perform small procedures.
Topical anesthetics can be effectively used for many laser procedures, as well as decreasing pain associated with pinpricks of local anesthesia. These products require an extended time of application and/or occlusion to penetrate the stratum corneum and work effectively. The level of anesthesia obtained with these agents is often inconsistent. Topical anesthetics are more effective on mucosa because of the absence of the corneal barrier. There are numerous lidocaine-containing products in a variety of preparations. Eutectic mixture of 2.5% lidocaine and 2.5% prilocaine has also been used extensively. Prilocaine-induced methemoglobinemia has been reported in children from the increased systemic absorption of prilocaine from certain topical products.
Direct application of ice can reduce injection site pain. Ethyl chloride spray rapidly chills the skin and can be used for minor curettage procedures or needle insertion. Refrigerated forced air or water-chilled sapphire crystals can help reduce pain associated with laser procedures. Ophthalmic solutions of proparacaine 0.5% or tetracaine 0.5% can provide rapid anesthesia and are useful when placing corneal shields.
Alam M, et al: Safety of peak serum lidocaine concentration after Mohs micrographic surgery. J Am Acad Dermatol 2010; 63: 87.
Kouba DJ, et al: Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol 2016; 74: 1201.
Park KK, Sharon VR: A review of local anesthetics. Dermatol Surg 2017; 43: 173.
Sobanko JF, et al: Topical anesthetics for dermatologic procedures. Dermatol Surg 2012; 38: 709.
Anatomy
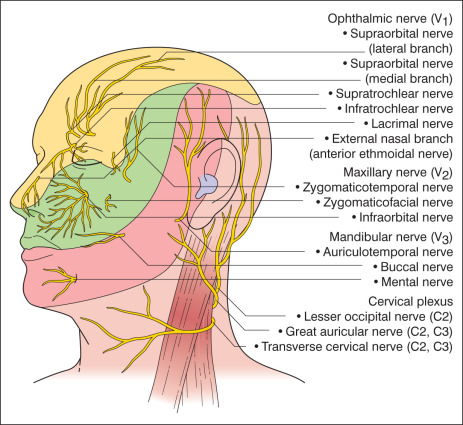
A thorough understanding of anatomy is critical when performing dermatologic surgery. The vascular supply, sensory and motor innervation, and muscles of facial expression all play a role in the successful surgical outcome ( Figs. 37.1–37.3 and Box 37.2 ).
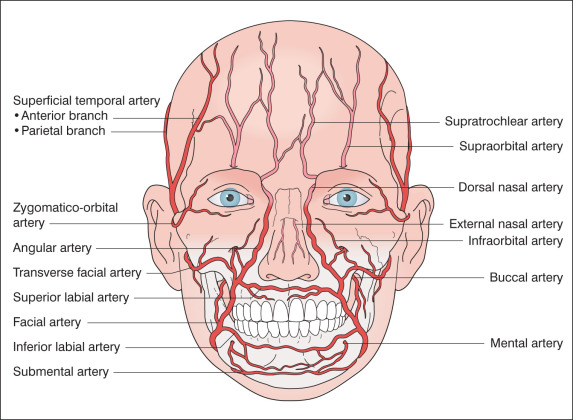
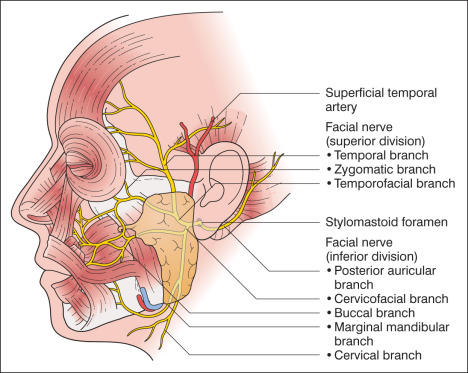
Temporal Branch
Frontalis muscle (m.)
Corrugator supercilii m.
Orbicularis oculi m. (upper portion)
Auricular m. (anterior and superior; also known as temporoparietalis m.)
Posterior Auricular Branch
Occipitalis m.
Auricular m. (posterior)
Zygomatic Branch
Orbicularis oculi m. (lower portion)
Nasalis m. (alar portion)
Procerus m.
Upper lip muscles
- •
Levator anguli oris m.
- •
Zygomaticus major m.
- •
Buccal Branch
Buccinator m. (muscle of mastication)
Depressor septi nasi m.
Nasalis m. (transverse portion)
Upper lip muscles
- •
Zygomaticus major and minor muscles
- •
Levator labii superioris m.
- •
Orbicularis oris m.
- •
Levator anguli oris m.
- •
Lower lip muscles (orbicularis oris m.)
Marginal Mandibular Branch
Lower lip muscles
- •
Orbicularis oris m.
- •
Depressor anguli oris m.
- •
Depressor labii inferioris m.
- •
Mentalis m.
- •
Risorius m.
Platysma m. (upper portion)
Cervical Branch
Platysma m.
Although a full description of the entire anatomy that is relevant to dermatology is beyond the scope of this text, several key danger zones are worthy of mention. The temporal branch of the facial nerve is at greatest risk for injury when it runs superficial to the deep temporalis fascia as it crosses the zygomatic arch. Care must be taken to undermine bluntly in a plane above the superficial muscular aponeurotic system (SMAS). Injury to the temporal nerve results in brow ptosis and inability to raise the eyebrow. The danger zone for the marginal mandibular nerve lies where it crosses over the body of mandible, just anterior to the masseter muscle. Injury to the marginal mandibular nerve causes asymmetric ipsilateral lip elevation and inability to show the lower teeth. The spinal accessory nerve is at risk in a region of the neck delineated by the clavicle inferiorly, sternocleidomastoid muscle anteriorly, and trapezius muscle laterally and posteriorly. Damage to the nerve causes a winged scapula, inability to shrug the shoulder, difficulty abducting the shoulder, shoulder drop, and chronic shoulder pain.
Chow S, Bennett RG: Superficial head and neck anatomy for dermatologic surgery. Dermatol Surg 2015; 41: S169.
Equipment
The choice of instruments and suture depends on the procedure being performed. Most simple, in-office biopsies are performed in a “clean” rather than sterile manner and require minimal instrumentation. More complex excisional and reconstructive surgery is generally performed with sterile technique and employs a surgical tray with a wider range of instruments ( Box 37.3 ).
- •
Scalpel handle (flat No. 3)
- •
Blade (No. 15)
- •
Needle holder (appropriate size)
- •
Sharp curved iris scissors, tissue-cutting scissors
- •
Blunt undermining scissors
- •
Skin hook (dull-tipped, two to four prongs)
- •
Hemostats
- •
Forceps (1 × 2 teeth, with suture platform)
- •
Skin preparatory scrub in sterile basin
- •
Sterile towels
- •
Sterile gauze and cotton-tipped swabs
- •
Hyfrecator cover
- •
Suture
- •
Suture scissors
- •
Blade remover
For procedures requiring sutures, absorbable material is used for deeper, layered closures, whereas surface sutures are generally nonabsorbable or fast-absorbing ( Table 37.3 ). The large number of suture choices relates to both the type of procedure performed and the anatomic location treated. Choices include absorbable and nonabsorbable, synthetic and nonsynthetic, monofilament and braided. The surgeon must consider a variety of other characteristics when choosing which suture to use. Memory is the ability of the suture to return to its original shape after deformation, which results in poor handling and decreased knot security. Plasticity is the ability of the suture to retain its new shape after it has been stretched. Elasticity is the ability of a suture to return to its original length and shape after stretching, an important factor to consider in relation to the resulting edema associated with surgery. The coefficient of friction is the ease with which the suture slides through tissue and is directly related to knot security. Capillarity is the ability of the suture to wick away fluid, with braided sutures having an increased tendency to trap fluid and bacteria. All have appropriate applications.