Abstract
This chapter describes disorders in which there is an increase in the amount of collagen within the skin, followed by those with an increase in elastic fibers. The first group includes hypertrophic scars, which remain confined to the site of the original injury, and keloids, which extend in a claw-like fashion beyond the original wound margin into adjacent normal skin. Additional entities in this group include fibromatoses such as Dupuytren disease, characterized by a thickening of the palmar and digital fascia that can lead to flexion contractures of the digits, and cutis verticis gyrata, in which there is hypertrophy and folding of the skin of the scalp. The range of clinical findings in hyaline fibromatosis syndrome, a genodermatosis due to CMG2 mutations, is also reviewed. Lastly, disorders characterized by an increase in elastic tissue are discussed, including late-onset focal dermal elastosis, linear focal elastosis, elastoderma, elastofibroma dorsi, and elastoma.
Keywords
hypertrophic scar, keloid, Dupuytren disease, Dupuytren contracture, cutis verticis gyrata, hyaline fibromatosis syndrome, juvenile hyaline fibromatosis, infantile systemic hyalinosis, late-onset focal dermal elastosis, linear focal elastosis, elastoderma, elastofibroma dorsi, elastoma
This chapter will discuss disorders in which there are increased amounts of collagen within the skin, followed by those with increased elastin.
Hypertrophic Scars and Keloids
- ▪
Conventional scars are preceded by injury, immediate in onset, flat, and asymptomatic
- ▪
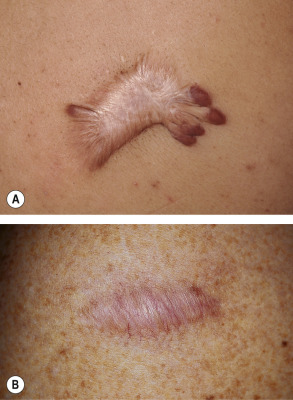
Hypertrophic scars are raised and confined to the wound margin. Sometimes they improve spontaneously and tend to have a good response to treatment
- ▪
Keloids extend beyond the wound margin and are delayed in onset. They seldom resolve spontaneously and response to treatment is often poor
Introduction
All wounds heal with some degree of scar formation, but the mechanisms that govern whether the result will be a fine thin scar, a prominent hypertrophic scar, or a tumor-like keloid remain unclear. The latter represent two forms of abnormal wound healing. Both are characterized by local fibroblast proliferation and excessive collagen production in response to cutaneous injury. However, their clinical and histopathologic features differ, as well as proposed pathogeneses.
Epidemiology
There is a higher prevalence of keloids in African, African-American, Spanish and Asian populations, with an incidence ranging from 4–16% . Although the incidence of hypertrophic scars is probably higher than that of keloids, precise data are lacking . Neither entity has a gender prevalence; they occur most commonly in those 10 to 30 years of age . Individuals in this age group are more frequently subjected to trauma and their rate of collagen synthesis is higher. Younger skin also possesses greater tension, as compared to older skin which has less elasticity and is more redundant .
Etiology
Proposed etiologic factors include: genetic predisposition; depth, type and location of the skin injury or wound; degree of tension; local infection or inflammation; and hormonal influences . There is often a familial tendency to develop hypertrophic scars and keloids and possibly an autosomal dominant mode of inheritance with incomplete clinical penetrance and variable expression . A range of skin injuries can lead to abnormal scarring, from lacerations, burns, surgical excisions and skin piercings to injections (vaccines, tattoo inks) and cutaneous inflammation (e.g. acne vulgaris, insect bites) . Moreover, tension is constantly transmitted to the skin from the underlying cartilaginous and bony skeleton, and skin tension can be aggravated by loss of tissue, as occurs with surgical excisions . In addition, hormonal influences have been proposed as an explanation for the appearance of keloids at or after puberty and their resolution following menopause, as well as reports of the onset or enlargement of keloids during pregnancy . Lastly, insights should come from genetic disorders such as Goeminne syndrome and Bethlem myopathy in which spontaneous keloids are a characteristic feature .
Pathogenesis
In fetuses, cutaneous wounding during the first and second trimesters of gestation does not lead to scarring. This is thought to reflect a healing process that is occurring via tissue regenerative pathways in a sterile environment and in the absence of inflammation . The ability to heal wounds created by injuries, in particular trauma, offers an evolutionary advantage, with associated scarring being less of an issue. However, cutaneous scarring resulting from surgical procedures is viewed as an unwanted consequence. A regenerative response would be preferred, thereby conceivably providing a superior cosmetic result and avoiding the formation of hypertrophic scars and keloids .
Conventional wound healing occurs in three phases (see Fig. 141.3 ): an inflammatory phase which aims to contain the injury and prevent infection; a proliferative phase in which there is formation of granulation tissue; and a remodeling (maturation) phase in which the redness of the wound lightens and its nodularity softens and flattens due to simultaneous collagen synthesis and degradation . The latter phase can last up to one year. Greater understanding of this remodeling phase could provide targets for preventative or therapeutic interventions.
Since Chapter 141 discusses the molecular basis of wound healing, this section will focus on key mediators of tissue scarring . Within keloids, collagen synthesis by fibroblasts is markedly increased as is production of transforming growth factor (TGF)-β. TGF-β has been shown to play a pivotal role during the proliferative phase of wound healing. Expression of TGF-β1 or TGF-β2 leads to an increase in scarring , whereas expression of TGF-β3 is associated with a reduction in scarring . This has led to the introduction of therapies based upon the biologic effects of these different forms of TGF, e.g. injections of recombinant TGF-β3, injections of mannose-6-phosphate which inhibits TGF-β1 and TGF-β2 signaling (see Table 98.4 ).
In regenerative healing, there is both a lack of scarring and an absence of inflammation. Support for this association comes from studies in which proinflammatory cytokines interleukin-6 (IL-6) and IL-8 enhanced scarring, while the anti-inflammatory cytokine IL-10 decreased the amount of scar tissue . Several additional factors propagate a robust fibroblast response within the healing wound that can lead to increased scar formation, including platelet-derived growth factor (PDGF), two transcription factors – homeobox B13 and early growth response protein 1, and components of the Wnt signaling pathway . Of note, fibroblasts isolated from keloids have increased expression of receptors for PDGF and TGF-β.
Clinical Features
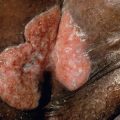
Although distinguishing a hypertrophic scar from a keloid clinically can sometimes be difficult, especially if the lesion is small or of recent onset, each has several characteristic features ( Table 98.1 ). Both keloids and hypertrophic scars have a smooth surface and are firm to palpation. They may be pruritic or painful and occasionally inhibit normal motion of adjacent tissues. The color can vary from pink–purple (early lesions) to skin-colored to hypo- or hyperpigmented. While keloids may be more elevated above the skin surface than hypertrophic scars, the key difference is that keloids extend beyond the boundary of the original wound into adjacent normal skin, often with claw-like extensions resembling the pincers of a crab (in Greek, chelè means claw) ( Figs 98.1 & 98.2 ). In contrast, hypertrophic scars remain confined to the site of the original injury ( Fig. 98.3 ).
| CLINICAL FEATURES OF CONVENTIONAL SCARS, HYPERTROPHIC SCARS, AND KELOIDS | |||
|---|---|---|---|
| Conventional scar | Hypertrophic scar | Keloid | |
| Preceded by injury | Yes | Yes | Not always |
| Onset | Immediate | Immediate | Delayed |
| Erythema | Temporary | Prominent | Varies |
| Profile | Flat | Raised | Raised |
| Symptomatic | No | Yes | Yes |
| Confined to wound margin | Yes | Yes | No |
| Spontaneous resolution | N/A | Possible, gradual | Rare |
| Treatment response | N/A | Good | Poor |
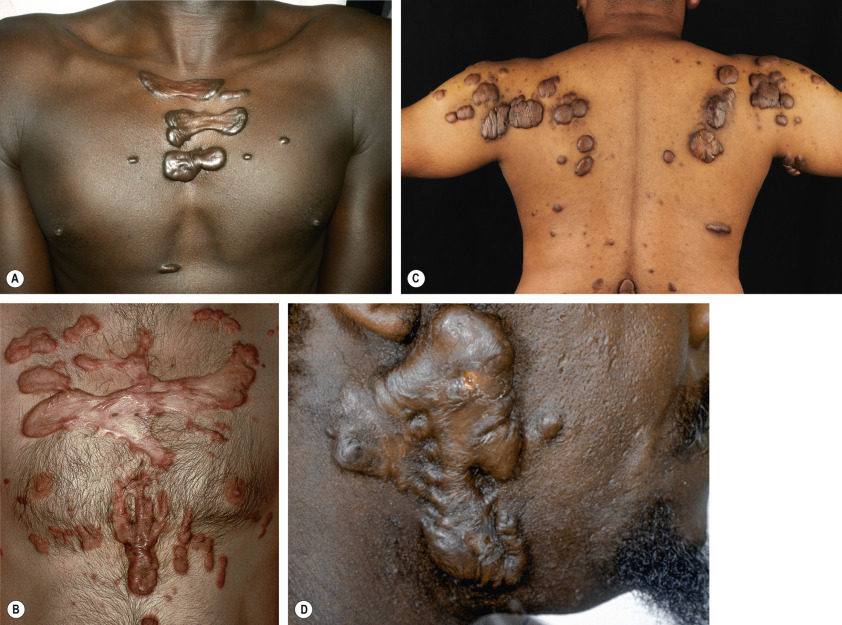
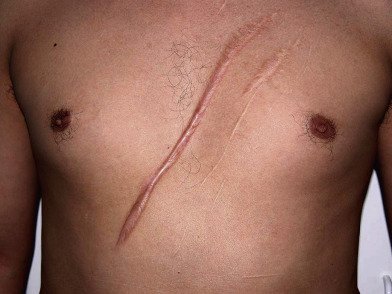
Most keloids appear within one year of an injury, although the intervening interval may be up to 24 years . Hypertrophic scars usually develop within a few weeks to months after wounding, and they frequently flatten spontaneously within 1 to 2 years. Keloids, on the other hand, do not regress over time. While both favor sites of increased wound tension such as the upper trunk, shoulder, and upper outer arm, keloids can also develop in sites such as the earlobe, where there is minimal tension. In addition, keloids can form spontaneously, most often in the midchest region.
Scar assessment
Scar evaluation ranges from simple scales to use of technologically advanced devices that analyze one or more variables in a reproducible manner . The Vancouver Scar Scale ( Table 98.2 ) and the Patient & Observer Scar Assessment Scale (POSAS) are the two that have been most commonly employed . These scales have significant inter-patient variation because they score subjective features from color to degree of pain and pruritus. More recently, a Scar Cosmesis Assessment and Rating (SCAR) scale was proposed which consists of six observer-scored parameters – scar spread, erythema, dyspigmentation, suture marks, hypertrophy/atrophy, and overall impression (desirable/undesirable) – in addition to two yes/no questions answered by the patient regarding itch and pain . Non-invasive techniques can be used to more objectively measure scar color, blood flow, transcutaneous oxygen tension, skin hardness, elasticity, and hydration . In addition, skin imaging techniques such as dedicated scar photography, dermoscopy, high-frequency ultrasound, laser Doppler perfusion imaging, confocal microscopy, and magnetic resonance imaging can be employed to evaluate scars .
| VANCOUVER SCAR SCALE | ||||||
|---|---|---|---|---|---|---|
| Score for each clinical feature | 0 | 1 | 2 | 3 | 4 | 5 |
| Pliability | Normal | Supple | Yielding | Firm, solid unit | Banding/“ropes” | Contracture |
| Height | Flat | <2 mm | 2–5 mm | >5 mm | – | – |
| Vascularity | Normal | Pink | Red | Purple | – | – |
| Pigmentation | Normal | Hypopigmentation | Hyperpigmentation | – | – | – |
Pathology
Distinguishing between a keloid and a hypertrophic scar is important when contemplating treatment, especially more aggressive therapies such as radiation. In some patients, histologic examination is required to make this distinction ( Table 98.3 ). In hypertrophic scars, there is an increase in both the number of fibroblasts and the density of collagen fibers within the dermis, both of which are oriented parallel to the skin surface ( Fig. 98.4 ). Keloids are characterized by the presence of whorls and nodules of strikingly thick, glassy, homogeneous collagen bundles that are composed of densely packed fibrils and oriented haphazardly throughout the dermis (keloidal collagen) ( Fig. 98.5 ). Early on, there are abundant deposits of fibrillary collagen within the reticular dermis of keloids, while mature lesions often have the characteristic thick sclerotic collagen. In longstanding keloids, there may be a return to the earlier fibrillary pattern.
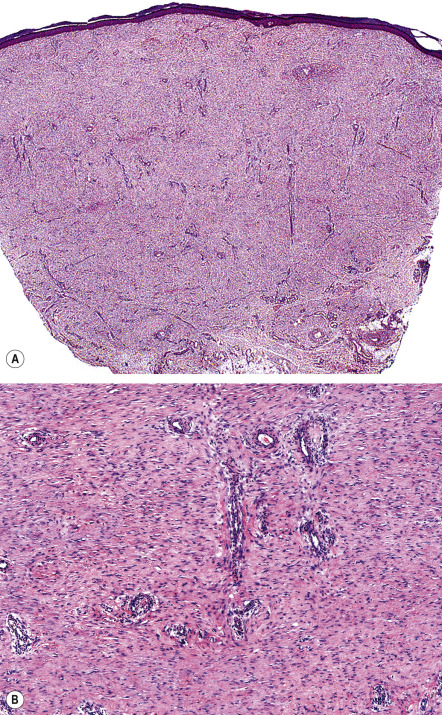
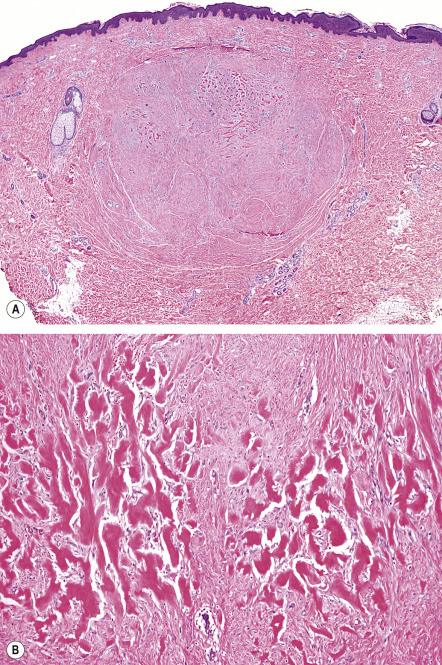
It is noteworthy that keloidal collagen may be absent in up to 45% of keloids . In scars with no detectable keloidal collagen, histologic features that favor a keloid include: no flattening of the epidermis; a lack of fibrosis within the papillary dermis; a tongue-like advancing edge as the scar tissue extends through the reticular dermis; a horizontal cellular fibrous band within the upper reticular dermis with a sharp demarcation from the normal-appearing papillary and reticular dermis; and prominent fascia-like fibrous bands in the deeper portion of the scar .
Attempts to differentiate hypertrophic scars from keloids via immunohistochemistry has led to conflicting results . In one study, nodules of α-smooth muscle actin (α-SMA)-positive cells (myofibroblasts) were only observed in hypertrophic scars , whereas another investigation found α-SMA expression in both hypertrophic scars (70%) and keloids (45%) . Expression of cyclooxygenase (COX)-1 was observed in 100% of keloids, but it was also expressed in ~50% of hypertrophic scars ; thus COX-1 expression favored, but did not specifically identify, keloids.
Differential Diagnosis
For keloids, the clinical differential diagnosis includes the sclerotic form of xanthoma disseminatum, lobomycosis (keloidal blastomycosis; lacaziosis), and the keloidal forms of scleroderma and morphea. Rarely, carcinoma en cuirasse can present as keloidal nodules. Patients with the vascular type of Ehlers–Danlos syndrome may occasionally develop keloidal plaques on their lower extremities, and spontaneous formation of keloids can be a clinical clue to other rare inherited syndromes such as Bethlem myopathy, Rubenstein–Taybi syndrome, and Goeminne syndrome (see below).
Histopathologically, keloids and hypertrophic scars need to be differentiated from spindle cell tumors, primarily dermatofibroma (DF), dermatofibrosarcoma protuberans (DFSP), desmoplastic melanoma, and the scar-like variant of squamous cell carcinoma (SCC). Immunostaining for CD34 and factor XIIIa, which is usually positive in DFSP and DF, respectively, is negative in keloids and hypertrophic scars . While there is minimal or no immunostaining for S100 protein in scars, desmoplastic melanomas usually demonstrate strong expression of this marker . Finally, the unusual scar-like variant of SCC may only have focal keratin expression, and a complete panel of keratin stains (to increase sensitivity) may be required for its detection .
Treatment
The management of keloids and hypertrophic scars continues to challenge clinicians, and there is no universally accepted treatment algorithm. Potential evidence-based treatments are outlined in Table 98.4 . Prevention remains the best strategy in predisposed patients, including avoidance of nonessential surgery in high-risk anatomic sites and attention to postsurgical wound care . There is limited evidence to support over-the-counter scar reduction products such as silicone sheeting and topical creams containing vitamin E . While potent topical corticosteroids are sometimes used to address pruritus, intralesional triamcinolone is the major first-line therapy for flattening hypertrophic scars and keloids . Its utility was confirmed in a recent meta-analysis . An alternative, especially for needle-averse individuals, is clobetasol propionate 0.05% cream under silicone dressing occlusion . Intralesional or topical corticosteroids may be combined with intralesional 5-fluorouracil .
Surgical revision of both hypertrophic scars and keloids must be undertaken with caution given the high recurrence rates. Cryosurgery has been used successfully and is often combined with intralesional triamcinolone. Pulsed dye laser therapy has become popular as a modality for postsurgical scar reduction and is associated with few side effects . Due to theoretical concerns regarding carcinogenesis, radiotherapy is reserved primarily for treatment-resistant keloids. However, current postoperative protocols limit total exposure and vary from 12–16 Gy in 3–4 fractions to 20 Gy in 5 fractions, beginning within 24–48 hours following surgery (see Ch. 139 ).
Potential therapies based upon insights into pathogenesis (see above) include recombinant TGF-β3 (avotermin), human recombinant IL-10, and mannose-6-phosphate which is a potent inhibitor of TGF-β1 and TGF-β2 signaling .
Dupuytren Disease
▪ Dupuytren’s contracture ▪ Palmar fibromatosis ▪ Dupuytren’s diathesis
- ▪
Thickening of palmar and digital fascia
- ▪
Progressive flexion contracture of affected digits
- ▪
Myofibroblast proliferation followed by excess collagen synthesis
- ▪
Treatment includes injections of collagenase and surgical fasciotomy/fasciectomy
History
Henry Cline described thickening of the palmar fascia in 1777 and proposed palmar fasciotomy as a cure. In 1831, Guillaume Dupuytren characterized and popularized this entity .
Epidemiology
Dupuytren disease is common in Caucasians of northern European ancestry, with a prevalence in Western countries that ranges from 1–30%; it is rare in patients with dark skin phototypes. The incidence of Dupuytren disease increases with age and it is most common in middle-aged and older men . Familial associations suggest an autosomal dominant inheritance, but sporadic cases are common. The lesion is associated with other fibromatoses (e.g. plantar fibromatosis; see Ch. 116 ) as well as diabetes mellitus, alcoholism, and perhaps cigarette smoking . Recently, use of vemurafenib was associated with a new onset or worsening of Dupuytren disease and plantar fascial fibromatosis.
Pathogenesis
Fibrogenic cytokines (e.g. TGF-β1, TGF-β2) are thought to play a role due to their ability to induce collagen production, proliferation of fibroblasts, and the differentiation of fibroblasts into myofibroblasts. Of nine susceptibility loci identified in a genome-wide association study, 6 of 9 harbored genes that encode proteins in the Wnt signaling pathway . In more recent genome-wide assays of fibroblasts from Dupuytren disease, the differential expression patterns included genes that encode proteins involved in the extracellular matrix (e.g, collagen), fibrosis (e.g. follistatin), tissue remodeling (e.g. collagenases, matrix metalloproteinases), cellular movement (e.g. kinesins), and signaling pathways (e.g. STAT1, Wnt2), as well as growth factors (e.g. fibroblast growth factor 9) .
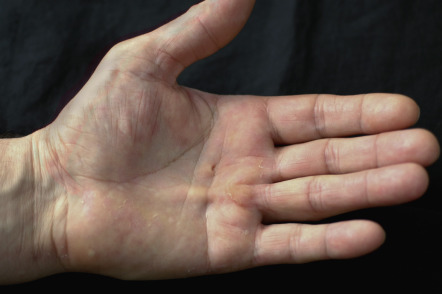
Clinical Features
Patients often present late in the course of their disease with flexion contractures of the affected digits. Lesions usually have an ulnar distribution, and the ring finger is most commonly affected ( Fig. 98.6 ). The disorder begins with a nodule in the fascia of the palm, which grows to produce a cord which then contracts, producing a flexion contracture of the metacarpophalangeal and proximal interphalangeal joints. Complications of Dupuytren disease include nerve injury, loss of joint mobility, and the development of reflex sympathetic dystrophy .