Abstract
This chapter reviews the human immunodeficiency virus (HIV) pandemic and the broad spectrum of infectious, inflammatory, and neoplastic skin conditions that may occur in the setting of HIV-induced immune dysfunction. The cutaneous findings often correlate with immune status, and knowing the viral load and CD4 + T-cell count can help to generate a more focused differential diagnosis. The possibility of HIV infection should be considered when patients present with severe, unusual, and treatment-resistant skin conditions; examples include giant molluscum contagiosum, extensive seborrheic dermatitis, and refractory herpes simplex virus infection. Following initiation of antiretroviral therapy (ART), skin disorders such as herpes zoster and non-tuberculous mycobacterial infection may become clinically apparent or worsen when the immune status improves, a phenomenon known as the immune reconstitution inflammatory syndrome (IRIS). Although ART has led to a reduction in the frequency of severe opportunistic infections and skin conditions associated with advanced HIV disease, e.g. Kaposi sarcoma, other dermatologic problems have become more common, e.g. lipodystrophy, neoplasia related to human papillomavirus infection. In addition, the medications used in ART can lead to cutaneous drug reactions.
Keywords
HIV infection, AIDS, antiretroviral therapy (ART), opportunistic infections, immune reconstitution inflammatory syndrome, adverse drug eruption, oral hairy leukoplakia, pruritic papular eruption, eosinophilic folliculitis of HIV infection
- ▪
A broad spectrum of infectious, inflammatory, and neoplastic skin conditions may occur in the setting of HIV-induced immune dysfunction
- ▪
The cutaneous findings often correlate with immune status, and knowing the viral load and CD4 + T-cell count can help to generate a more focused differential diagnosis
- ▪
The possibility of HIV infection should be considered when patients present with severe, unusual, and treatment-resistant skin conditions; examples include giant molluscum contagiosum, extensive seborrheic dermatitis, and refractory herpes simplex virus infection
- ▪
Following initiation of antiretroviral therapy (ART), skin disorders such as herpes zoster and Mycobacterium avium complex infection may become clinically apparent or worsen when the immune status improves, a phenomenon known as the immune reconstitution inflammatory syndrome (IRIS)
- ▪
Although ART has led to a reduction in the frequency of severe opportunistic infections and skin conditions associated with advanced HIV disease, e.g. Kaposi sarcoma, other dermatologic problems have become more common, e.g. lipodystrophy, neoplasia related to human papillomavirus infection
- ▪
The medications used in ART may lead to cutaneous drug reactions
Introduction
Mucocutaneous disorders not only provide important clues to the diagnosis of HIV infection, they also serve as markers of progression and possible associated systemic disease in addition to significantly impacting quality of life. Conditions that typically develop at different stages of HIV infection are presented in Tables 78.1 and 78.2 . Effective antiretroviral therapy (ART) has transformed HIV infection from a progressive, fatal disease to a chronic disorder requiring maintenance therapy. As a result, cutaneous disorders associated with severe immunosuppression, such as Kaposi sarcoma and serious opportunistic infections, are observed less frequently. However, with longer survival, other diseases have become more common (e.g. anal intraepithelial neoplasia), and ART itself has led to dermatologic challenges, including drug reactions, lipodystrophy, and immune reconstitution inflammatory syndrome (IRIS). In low-income countries where ART is not readily available, opportunistic infections are still a common cause of morbidity and mortality. Approximately 80–95% of HIV-infected individuals develop at least one skin disorder, and dermatologists worldwide continue to play important roles in their care.
| WHO CLINICAL STAGING OF HIV/AIDS IN THE SETTING OF CONFIRMED HIV INFECTION | ||
|---|---|---|
| All ages | Adults and adolescents | Children |
| Clinical stage 1 – asymptomatic; immunologic correlate * : >500 CD4 + cells/mm 3 | ||
| ||
| Clinical stage 2 – mild; immunologic correlate * : 350–499 CD4 + cells/mm 3 | ||
|
|
|
| Clinical stage 3 – advanced; immunologic correlate * : 200–349 CD4 + cells/mm 3 | ||
|
|
|
| Clinical stage 4 – severe/AIDS-defining conditions; immunologic correlate * : <200 CD4 + cells/mm 3 | ||
|
|
|
* In individuals ≥6 years of age; corresponding counts in children <6 years of age are available at www.who.int/hiv/pub/guidelines/hivstaging/en/ .
| CORRELATION OF CD4 + CELL COUNT WITH SPECIFIC HIV-ASSOCIATED DISORDERS | ||||
|---|---|---|---|---|
| System * | >500 CD4 + cells/mm 3 | <500 CD4 + cells/mm 3 | <250 CD4 + cells/mm 3 | <50 CD4 + cells/mm 3 |
| Dermatologic |
|
|
|
|
| Respiratory | Bacterial pneumonia and sinusitis |
| Pneumocystis jiroveci pneumonia (PCP) | Pseudomonas spp. pneumonia |
| Nervous |
| Mononeuritis multiplex |
| Primary CNS lymphoma |
| Hematologic | Persistent generalized lymphadenopathy |
| Non-Hodgkin lymphoma | |
| Other | Myopathy |
|
|
|
* Diseases occur with increasing frequency and severity at lower CD4 + cell counts.
History
In 1981, reports of Pneumocystis jiroveci ( formerly carinii ) pneumonia, Kaposi sarcoma, and chronic ulcerative herpes simplex virus (HSV) infections in homosexual men in association with reduced cell-mediated immunity were first published . Soon thereafter, the underlying condition was termed acquired immune deficiency syndrome (AIDS), with descriptions of other associated opportunistic infections as well as additional affected populations, including hemophiliacs/blood transfusion recipients, intravenous drug users, sexual partners of persons with the disease, infants, and heterosexual Haitians . The causative T-lymphotropic retrovirus was identified in 1983 and named the human immunodeficiency virus (HIV) ; homology to an African zoonosis originating from non-human primates was demonstrated.
Identification of blood-borne, sexual, and mother-to-child transmission of HIV led to universal precautions for the handling of blood and body fluids as well as specific protocols for delivery and breastfeeding. The first effective therapy against HIV, the nucleoside reverse transcriptase inhibitor zidovudine (AZT), was FDA-approved in 1987. Highly active antiretroviral therapy (HAART; now referred to as ART), which utilizes several agents to suppress viral replication and thereby delay clinical progression, was introduced in 1996. The latter has led to a substantial reduction in HIV-related morbidity and mortality.
Epidemiology
The HIV/AIDS pandemic continues to be a major worldwide public health challenge in the 21 st century. It disproportionately affects resource-poor countries and specific populations, including sex workers, persons who inject drugs, and men who have sex with men (MSM). Globally in 2016, nearly 37 million people were estimated to be living with HIV, a further increase from previous years that reflects a longer life expectancy due to ART ( Fig. 78.1 ). The proportion of HIV-infected individuals receiving ART has risen dramatically, from <3% in 2000 to ~50% in 2016 , and the number of yearly AIDS-related deaths worldwide decreased from a peak of 2 million in 2005 to 1 million in 2016 . There were ~1.8 million new HIV infections globally in 2016, representing a 44% decline from ~3.2 million in 2000 and a 15% decline from 2.2 million in 2010; the annual number of new infections in children decreased ~50% between 2010 and 2016 .
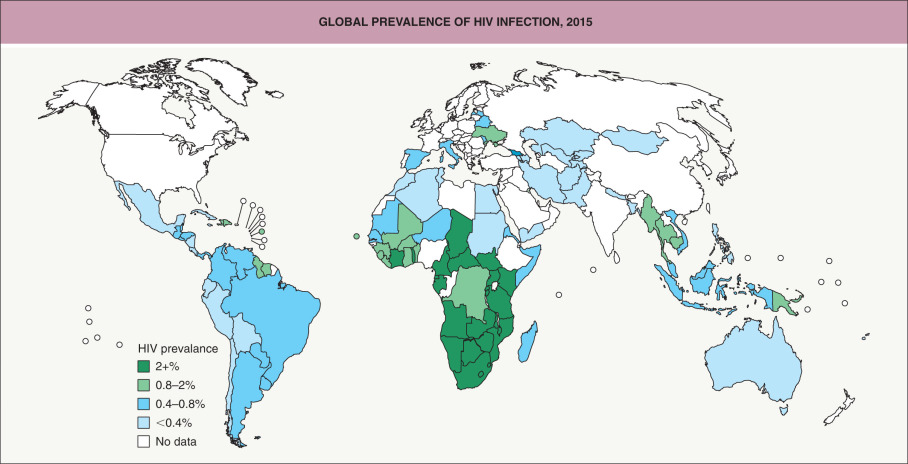
The HIV/AIDS pandemic most severely affects sub-Saharan Africa, which accounts for ~70% of all people living with HIV and ~65% of new HIV infections. However, since 2010 the annual number of new HIV infections among adults decreased 10–15% in sub-Saharan Africa but increased >50% in Eastern Europe and Central Asia . During the past decade, there has been an overall trend towards safer sexual behaviors in high-prevalence countries. Despite this, the continued high incidence of HIV among MSM in most parts of the world remains a major concern .
The HIV care continuum has emerged as an important tool to track the steps in the treatment cascade that are required to achieve the viral suppression necessary for successful HIV therapy and prevention . These steps include diagnosis, linkage to care, retention in care, adherence to ART, and viral suppression.
The distribution of the modes of transmission of HIV infection can vary depending upon the country or region. In the US, the transmission of new HIV diagnoses in 2015 was as follows: male-to-male sexual contact, 67%; heterosexual contact, 24%; injection drug use, 6%; male-to-male sexual contact plus injection drug use, 3%; and perinatal, 0.02%. In resource-limited areas, heterosexual contact is responsible for ~70–80% of HIV infections, while perinatal transmission and injection drug use each account for ~5–10% of cases. Risk factors for HIV transmission include high viral load, unprotected receptive anal intercourse, lack of circumcision, and the presence of other sexually transmitted infections.
Pathogenesis
AIDS is caused by HIV-1 and much less often HIV-2. Both are enveloped, single-stranded RNA lentiviruses in the Retroviridae family. They are zoonotic and originated from simian immunodeficiency viruses (SIVs) that infect African primates: HIV-1 from chimpanzees and HIV-2 from sooty mangabeys. HIV-1 has four distinct lineages, each resulting from an independent cross-species transmission event; group M is responsible for ~99% of pandemic HIV infections, while groups N, O, and P are restricted to West Africa . HIV-2 is also largely restricted to West Africa, where its overall prevalence is declining. HIV-2 is less transmissible and virulent than HIV-1, and persons infected with HIV-2 have lower viral loads and do not usually progress to AIDS. HIV exhibits substantial genetic variability due to the error-prone nature of its reverse transcriptase as well as very rapid virus replication in infected individuals.
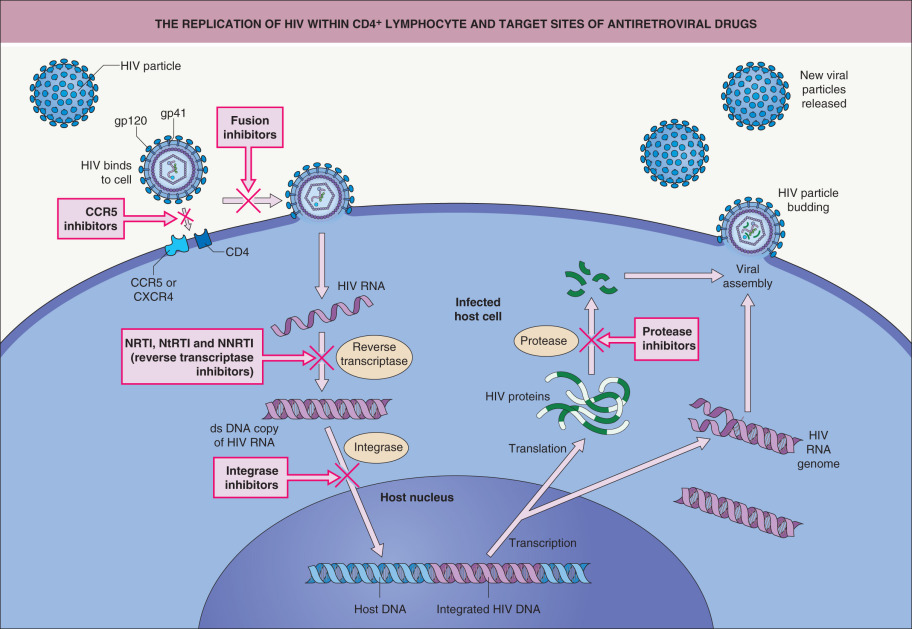
During HIV-1 infection, the viral envelope proteins gp120 and gp41 initially interact with the CD4 molecule followed by a second interaction with a chemokine receptor, usually CCR5 or CXCR4 ( Fig. 78.2 ). These receptors are found on activated CD4 + T cells, monocyte-macrophages, and dendritic cells. CXCR5 is the predominant fusion cofactor in most transmitted HIV infections ; however, CD4-independent cell infection of renal cells and astrocytes gives rise to chronic renal disease and neurocognitive disorders. Following binding, changes in conformation induce fusion of the viral envelope with the plasma membrane, and the virus particle is thereby internalized. The RNA genome is transcribed by a reverse transcriptase enzyme that produces a DNA copy of the HIV RNA. This viral DNA copy is spliced into the host DNA through the action of an integrase enzyme. Transcription of viral DNA into RNA yields genomic material for new viral particles or it can be translated into viral proteins. Cleavage of the latter into structural components of the virus is accomplished by proteases. Intact viruses are then produced and host cells are destroyed.
Infection by the transmitted/founder virus is followed by a rapid increase in HIV replication ( Fig. 78.3 ). After ~6 months, the viral load falls to a set point that is determined by the host’s innate natural killer (NK) cell response plus an adaptive immune response by CD8 + cells and neutralizing antibodies . The initial antibody response develops within 1–3 months after transmission and is strain-specific. This antibody response drives viral escape, and virus mutants become resistant to neutralization in 80% of patients. However, in 20% of patients, variants of the transmitted/founder virus evolve and induce antibodies with considerable neutralization breadth. CD8 + cytotoxic T cells directed against HIV begin to function within 10 days of exposure, correlating with the reduction in viremia during the acute retroviral syndrome; these cells also help to control the HIV infection throughout the disease course.
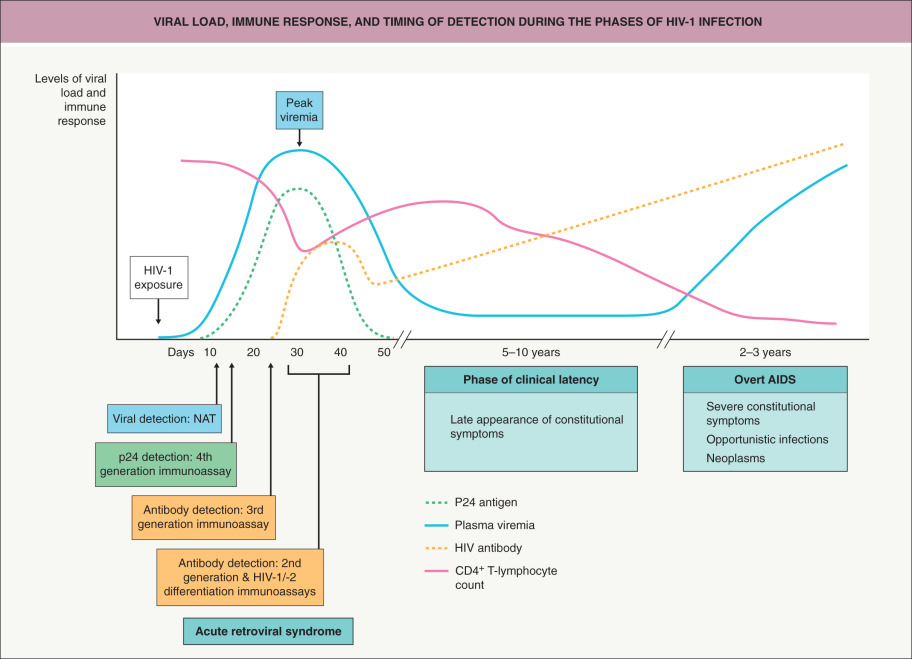
Initial infection results in a reduction in circulating CD4 + T cells, which is followed by a recovery to nearly normal levels and a subsequent slow fall of about 50 to 100 cells/mm 3 per year (see Fig. 78.3 ). In contrast, the CD4 + T cells residing within the gastrointestinal tract are rapidly depleted early on. The progressive depletion of CD4 + T cells is the result of killing by direct infection, bystander effects of syncytia formation, chronic immune activation, lymphoid tissue damage, and senescence.
The marked chronic immune activation that characterizes HIV infection is thought to be related to low-grade HIV replication, immune responses to HIV, reactivated infections, loss of mucosal integrity with consequent microbial translocation, and increased production of proinflammatory molecules . In patients receiving ART, increased levels of markers of inflammation have been associated with higher mortality rates and risk of cancer as well as cardiovascular, neurologic, and liver disease.
In a person infected with HIV, AIDS is defined by a CD4 + cell count of <200/mm 3 or <14% CD4 + T cells and/or the presence of an AIDS-defining condition (see Table 78.1 ). The natural history of HIV infection varies considerably, with the rate of decline in CD4 + T cells correlating with viral burden. For untreated infections, the median time for progression to AIDS is ~10 years. However, ~10–15% of untreated patients, historically referred to as “long-term non-progressors”, have their HIV infections for ≥10 years with no symptoms and CD4 + T-cell counts of >500 cells/mm 3 ; in addition, <0.5% of patients are “elite controllers” with no evidence of viremia. The ability of ART to suppress viral replication and enable reconstitution of CD4 + T cells has an immense impact on the disease course. Immune reactivation due to ART can result in a variety of inflammatory sequelae referred to as IRIS, ranging from unmasking or paradoxical “worsening” of infections or neoplasms to exacerbations of inflammatory disorders (see below).
HIV-Related Infectious Skin Conditions
In addition to the primary HIV exanthem, HIV-positive individuals often develop cutaneous viral, bacterial, and fungal infections as well as infestations. Patients who acquired HIV via sexual contact should also be tested for other sexually transmitted diseases (see Ch. 82 ); the converse is also true. The presence of erosions and ulceration in the anogenital region due to conditions such as syphilis and herpes simplex virus infections can also increase the risk of HIV transmission.
Viral Infections
Primary HIV exanthem (acute retroviral syndrome)
Primary HIV infection refers to the initial disease phase, when HIV-1 RNA, DNA, and/or p24 antigen are present in the plasma prior to the development of HIV-1 antibodies. Up to 80% of newly infected individuals develop an “acute retroviral syndrome” resembling influenza or mononucleosis. Symptoms develop 2–6 weeks after HIV exposure and often include fever, headache, myalgia/arthralgia, pharyngitis, lymphadenopathy, and night sweats. A morbilliform exanthem also occurs in 40–80% of patients; it lasts 4–5 days and is typically generalized, with the most prominent involvement on the face and trunk. Oral and genital ulcers are occasionally present. Symptoms are self-limited and resolve in a few days to weeks, with a median duration of 14 days. The initial phase of rapid HIV-1 replication causes a temporary fall in CD4 + cell counts, occasionally to an extent that gives rise to opportunistic infections.
Greater severity and longer duration of the acute retroviral syndrome is associated with a higher viral load set point and more rapid disease progression . Early initiation of ART during primary HIV infection may delay disease progression .
Herpes simplex virus (HSV)
Relatively immunocompetent HIV-infected individuals usually have typical, self-limited HSV disease. As immunity wanes, the frequency of HSV recurrences increases and lesions take longer to heal, potentially evolving into chronic, extensive, deep, and painful ulcers that favor the perianal region, genitalia, and tongue ( Fig. 78.4 ). Atypical presentations of HSV infection include folliculitis, verrucous plaques, and hypertrophic anogenital masses simulating neoplasia. Infection may become disseminated or occur at unusual sites such as the non-keratinized (unattached) mucosa of the oropharynx (see Fig. 72.1 ) and the esophagus.
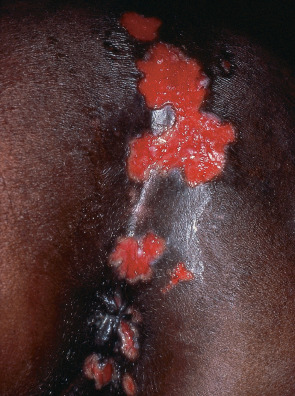
The ulcer edge should be scraped for HSV PCR (most sensitive), direct fluorescent antibody assay (DFA), and/or viral culture and, if negative, a skin biopsy performed; a Tzanck smear has low sensitivity. Higher doses and longer courses of antiviral medications are typically required, with continuation until all mucocutaneous lesions are healed. Resistance to acyclovir, usually due to reduced viral thymidine kinase activity, occurs more commonly in immunosuppressed (~5%) compared to immunocompetent (~1%) hosts. Alternative agents that do not rely on thymidine kinase include foscarnet, cidofovir, and imiquimod . Of note, HSV-2 can also promote HIV-1 replication .
Varicella–zoster virus (VZV)
In HIV-infected individuals, varicella tends to have new lesion formation over a longer period of time, a higher lesion count, and (particularly in adults) complications such as pneumonitis, hepatitis, and encephalitis. Lesions that persist as slowly healing ulcers have been reported.
HIV-infected individuals have a 7–15-fold higher incidence of herpes zoster compared to the general population . In addition to classic dermatomal eruptions, HIV-associated herpes zoster can present as chronic non-healing ulcers, hyperkeratotic verrucous plaques, and multidermatomal or disseminated skin lesions; potential complications include bacterial superinfection, systemic involvement, and multiple recurrences . Half of HIV-positive children with varicella develop herpes zoster or a disseminated eruption reminiscent of varicella within 2 years of the initial varicella eruption .
HIV-infected patients without VZV immunity should receive varicella–zoster immune globulin within 10 days of exposure to VZV. Antiviral treatment of VZV infection in HIV-positive patients should be continued until clinical resolution occurs (see Ch. 80 ); acyclovir resistance is possible. Of note, patients treated with ART may experience paradoxical worsening of herpes zoster when their CD4 + T-cell counts rise (see IRIS below).
Molluscum contagiosum virus
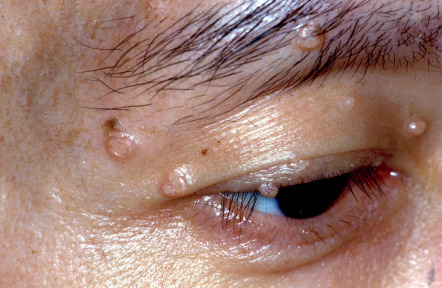
Molluscum contagiosum (MC) commonly occurs and tends to be more persistent in individuals with significantly reduced CD4 + T-cell counts. In addition to the typical presentation of small umbilicated papules ( Fig. 78.5 ), HIV-infected patients can develop larger (>1 cm), coalescent, verrucous, and widespread lesions . The clinical differential diagnosis may include basal cell carcinoma and cutaneous lesions of cryptococcosis, histoplasmosis, or other dimorphic fungal infections. Dermoscopic evaluation, microscopic examination following curettage of lesional contents, or biopsy can help to confirm the diagnosis. In addition to curettage and other destructive modalities, topical agents such as cidofovir have been used successfully for MC in HIV-infected patients. MC may resolve spontaneously after initiation of ART but can also present as a manifestation of IRIS.
Human papillomavirus (HPV)
The spectrum of mucocutaneous lesions due to HPV infection ranges from common warts and condylomata acuminata to squamous intraepithelial neoplasia and carcinoma. The prevalence of HPV infection is higher in HIV-infected individuals than in the general population and correlates with decreasing CD4 + T-cell counts. HIV-infected individuals have reduced HPV clearance, more treatment-resistant mucocutaneous lesions, and accelerated development of HPV-associated carcinoma. Skin lesions are often multiple, coalescent, and extensive ( Fig. 78.6 ). “Acquired epidermodysplasia verruciformis” (AEDV) is characterized by hypopigmented to pink tinea versicolor-like lesions and numerous flat-topped papules that are due to HPV-5, -8, and other β HPV types . HIV-infected individuals may develop squamous cell carcinomas (SCCs) in association with AEDV and other cutaneous warts, e.g. digital lesions due to high-risk HPV types, as well as anogenital warts.
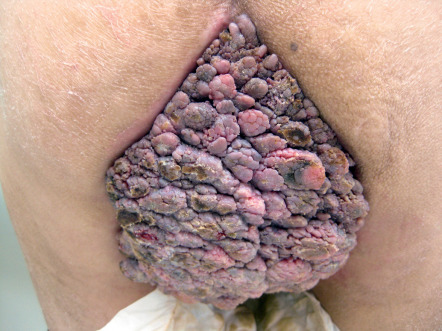
Compared to HIV-negative women, HIV-infected women have a 3-fold increase in the incidence of cervical intraepithelial neoplasia (CIN) as well as higher rates of high-grade CIN . Anal HPV is present in >90% of HIV-infected MSM, and most have at least one high-risk HPV type. HIV-infected MSM also have an up to 50% prevalence of high-grade anal intraepithelial neoplasia (AIN) and a 30- to 50-fold higher risk of anal cancer than does the general population ; a 5-fold increased risk of penile cancer has also been documented . Monitoring should include serial physical examinations of the anogenital area, colposcopy/proctoscopy, cervical/anal HPV determination and cytology, and histologic examination when indicated.
ART has not shown a demonstrable effect on the occurrence or clearance of cutaneous or anogenital warts. Enlargement or inflammation of pre-existing warts and an eruption of new warts can occur in the setting of IRIS . Some studies have shown a protective effect of ART on the development and progression of CIN and AIN . However, the longer life expectancy afforded by ART may increase the number of HIV-infected individuals susceptible to anal and cervical cancers .
The choice of therapy for anogenital warts depends upon their size, number, and location. Options include destructive modalities such as cryotherapy, electrosurgery, laser therapy and surgical excision, in addition to patient-applied topical therapies including imiquimod and podophyllotoxin (see Ch. 79 ). Immunosuppressed patients require more treatment sessions to achieve clearance and have frequent recurrences. The threshold for biopsy should be low, and several sites may need to be sampled to exclude SCC. HPV vaccines are immunogenic and well-tolerated in HIV-infected individuals.
Epstein–Barr virus (EBV)
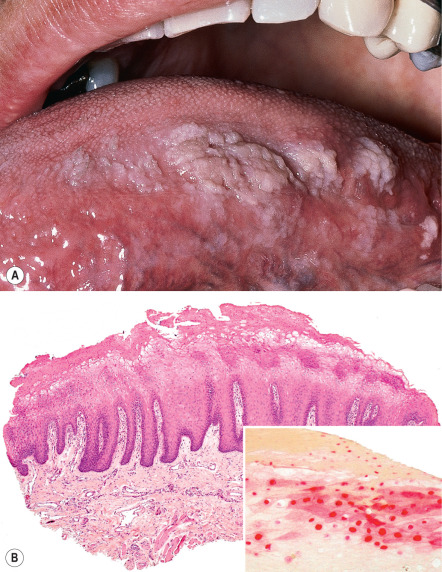
Oral hairy leukoplakia (OHL) results from EBV infection of the oral mucosa and typically presents as hyperkeratotic, corrugated white plaques with hair-like projections on the lateral aspect of the tongue ( Fig. 78.7 ). It can occur at any stage of HIV infection but is more prevalent in men and those with CD4 + T-cell counts below 200/mm 3 . It represents a predictor of rapid disease progression if ART is not initiated . OHL can also occur in patients with other forms of immunosuppression, such as organ transplant recipients. Although treatment is usually not necessary, topical or oral antiherpetic antiviral medications and topical podophyllin, tretinoin, or gentian violet may be of benefit, but recurrence is common. OHL may also regress with ART.
Cytomegalovirus (CMV)
Reactivation of CMV is associated with CD4 + T-cell counts that are <100/mm 3 , and it remains a significant cause of morbidity and mortality in patients with AIDS. Retinitis, esophagitis, and colitis represent common clinical manifestations, and mucosal ulcers may occur in the setting of disseminated disease. Cutaneous presentations are uncommon and include ulcers favoring the anogenital area, verrucous or hyperpigmented plaques, prurigo nodularis-like lesions, purpuric papules, vesicles, and morbilliform eruptions . Histologically, characteristic cytomegalic intranuclear inclusion bodies are seen within endothelial cells in the dermis of lesional skin (see Fig. 80.24 ). Ulcerative lesions are often co-infected with HSV or VZV, and because CMV can also be detected in nonlesional skin, its pathogenic role has been questioned . Specific treatments include oral valganciclovir and intravenous ganciclovir, foscarnet, and cidofovir.
Bacterial Infections
HIV infection predisposes affected individuals to recurrent and potentially severe cutaneous bacterial infections, which can be localized or widespread and sometimes have unusual clinical appearances. In addition to diminished antibody- and cell-mediated immune responses, predisposing factors include skin barrier impairment from excoriations or other cutaneous infections, indwelling catheters, and malnutrition. In addition to the entities discussed below, cutaneous bacterial infections that can occur in association with HIV infection include necrotizing fasciitis, nocardiosis, malacoplakia, pseudomonal “hot tub” folliculitis, “malignant” otitis externa, and ecthyma gangrenosum.
Staphylococcus aureus
Staphylococcus aureus is the most common bacterial pathogen in HIV-infected individuals. Cutaneous presentations include impetigo, folliculitis, furunculosis, wound infections, cellulitis, and rarely botryomycosis. Compared with the general population, HIV-infected individuals have a higher prevalence of methicillin-resistant S. aureus (MRSA) colonization of the skin and a 6-fold higher incidence of MRSA skin/soft-tissue infections; they also have more frequent recurrences . The lower extremities, buttocks, and scrotum are commonly affected, and risk factors include a low CD4 + T-cell count, recent treatment with antibiotics other than trimethoprim–sulfamethoxazole (TMP-SMZ), hospitalization, and illicit drug use. Use of intranasal mupirocin ointment and chlorhexidine washes can temporarily eradicate colonization but may not reduce infection rates .
Bacillary angiomatosis
Bacillary angiomatosis is a rare condition that is characterized by vascular proliferation and is due to the Gram-negative bacilli Bartonella henselae and B. quintana . It occurs in HIV-infected individuals who are severely immunocompromised, usually with CD4 + T-cell counts <100/mm 3 . Bacillary angiomatosis favors the skin and subcutaneous tissues, although virtually any internal organ can be affected. Patients typically present with a single to numerous, firm, red or violaceous papules and nodules; these lesions may be painful, ulcerate, or bleed profusely after trauma. Large subcutaneous nodules of bacillary angiomatosis may also ulcerate. Disseminated infection is associated with systemic symptoms such as fever, night sweats, and weight loss.
The differential diagnosis may include Kaposi sarcoma, pyogenic granulomas, and non-tuberculous mycobacterial infections. Histologically, a lobular proliferation of capillaries and venules with large, protuberant endothelial cells is seen, surrounded by a neutrophilic infiltrate. Lesional bacilli can be visualized with a Warthin–Starry stain or identified via PCR-based assays; serologic testing for anti- Bartonella antibodies may assist in diagnosis. Treatment consists of doxycycline or macrolides. The median time to complete response is ~1 month, and 3–4 months of therapy is recommended to prevent relapse .
Mycobacteria
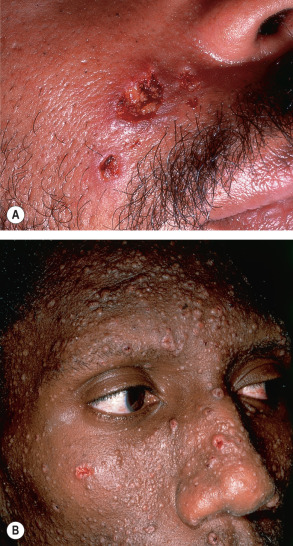
A diverse clinical spectrum of tuberculous and non-tuberculous mycobacterial skin infections can develop in HIV-positive patients. Cutaneous findings include erythematous to violaceous papules and nodules as well as ulcers, acneiform eruptions, abscesses, and verrucous plaques ( Fig. 78.8 ); a sporotrichoid (lymphocutaneous) distribution pattern is sometimes seen. The risk of infection increases with progressive immunosuppression, and the classic histologic finding of caseating granulomas may be absent in patients with markedly diminished cell-mediated immunity.
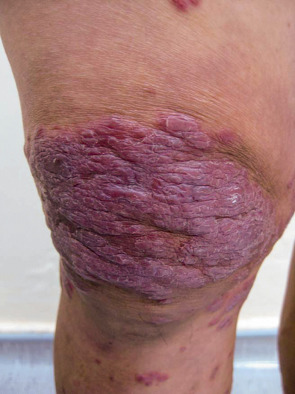
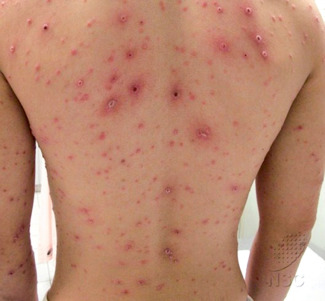
Syphilis
Although classic papulosquamous lesions of secondary syphilis are often seen, HIV-infected patients may also present with multiple primary chancres, concomitant lesions of primary and secondary syphilis, palmoplantar keratoderma, annular plaques, and lues maligna . The latter, which is also known as malignant or ulceronodular syphilis, is a severe form of secondary syphilis (see Ch. 82 ). A prodrome of fever, arthralgia/myalgia, headache, and photophobia precedes the appearance of papules, pustules, and necrotic nodules with ulceration and crusting ( Fig. 78.9 ); lesions are symmetrically distributed, with frequent involvement of the palms and sometimes the oral mucosa . HIV-infected patients have a higher likelihood, earlier onset, and greater severity of neurosyphilis. The recommended stage-specific treatment regimens are the same as those for HIV-negative persons, but with longer monitoring .
Fungal and Related Infections
In HIV-infected individuals, clinical presentations of cutaneous fungal infections range from localized tinea corporis to widespread papulonodules due to disseminated dimorphic fungal infections.
Candidiasis
Candidiasis is the most common fungal disease in those with HIV infection, and its incidence and severity are both increased in the setting of lower CD4 + T-cell counts. Oropharyngeal candidiasis is often the first clinical manifestation of HIV infection . Additional presentations include angular cheilitis, chronic paronychia, and onychodystrophy, as well as intertriginous, refractory vaginal, esophageal, and disseminated candidiasis. Continuous or intermittent use of systemic antifungal medications can prevent recurrences of candidiasis (see Ch. 127 ), but this must be weighed against the risk of developing resistance .
Dermatophytoses
Dermatophytosis in HIV-infected individuals may be more widespread, atypical in appearance, and refractory to treatment. For example, tinea pedis may extend onto the dorsum of the feet or give rise to chronic hyperkeratotic plaques, and nodular perifolliculitis (including Majocchi granuloma) can present with follicular papulopustules or deep granulomatous nodules . Proximal subungual onychomycosis is seen more frequently in immunocompromised patients and should prompt HIV testing . Some azole antifungals (e.g. itraconazole) interact with antiretroviral medications (see Ch. 131 ). Use of absorbent powders and topical antifungal agents can help to prevent recurrences.
Systemic fungal infections
HIV-infected individuals are predisposed to the development of disseminated cryptococcosis and dimorphic fungal infections, including histoplasmosis, blastomycosis, coccidioidomycosis, paracoccidioidomycosis, penicilliosis, and sporotrichosis (see Ch. 77 ).
Cryptococcus neoformans is an encapsulated yeast that is found worldwide in bird droppings and contaminated soil. Patients with CD4 + T-cell counts <100/mm 3 are particularly susceptible, and pulmonary or central nervous system disease may be the initial presentation of AIDS. Cutaneous lesions occur in 10–20% of patients with disseminated cryptococcosis and often appear as molluscum contagiosum-like umbilicated papules, nodules and pustules that preferentially affect the head and neck ( Fig. 78.10 ). Violaceous nodules and ulcerations may also be seen on the palate and tongue . The yeast’s polysaccharide capsule can be highlighted using the mucicarmine stain or India ink; detection of cryptococcal antigen in the serum or CSF can also be utilized for diagnosis.