Cross Face Nerve Graft
Michael J. A. Klebuc
DEFINITION
Cross face nerve graft—interposition nerve graft utilized to transmit facial nerve motor axons from a neurologically intact to a paralyzed/paretic, contralateral hemiface.
Utilized to restore motor innervation to denervated muscles of facial expression and to neurotized free, functional muscle flaps, also can be employed for sensory restoration.
Frequently utilized sources of nerve graft (sural nerve, saphenous nerve, medial antebrachial cutaneous nerve).
Approximately 50% of a facial nerve’s zygomatic and buccal branches can be selectively transected and their function redirected to the paralyzed hemiface without producing significant donor weakness (functional reserve).
The facial nerve is the only donor nerve that can consistently restore spontaneous, emotionally mediated facial motion.
ANATOMY
Facial nerve (somatic motor efferent component)—innervates the muscles of facial expression, auricular muscles, occipitalis, posterior belly digastric, stylohyoid, and stapedius.
The facial nerve main trunk emerges from the skull base via the stylomastoid foramen where it can be identified coursing 1 cm deep to the tragal pointer and medial to the posterior belly of the digastric muscle.1
Within the parotid gland, the facial nerve divides into a temporofacial and cervicofacial trunk.
Distal branches exit the parotid on its anteromedial surface.
The following branches innervate the corresponding muscles of facial expression2:
Frontal branch—frontalis muscle
Zygomatic branches—orbicularis oculi, zygomaticus major, zygomaticus minor, levator labii superioris, levator labii superioris alaeque nasi, corrugator supercilii, procerus
Buccal branches—buccinator, risorius, orbicularis oris
Marginal mandibular branches—depressor anguli oris, depressor labii inferioris, mentalis
Cervical branches—platysma
Muscles of facial expression are innervated on their deep surface with the exception of the levator anguli oris, buccinator, and mentalis.
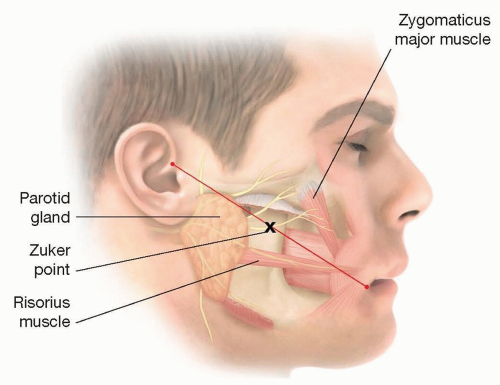
Zuker point describes the approximate location of the facial nerve branch supplying the zygomaticus major (FIG 1):
Midway point of a line drawn from the root of the helix to the lateral commissure.
Preferentially divided and coapted to a cross facial nerve graft (CFNG) as the first step in a two-staged free, functional muscle flap reconstruction.3
Pitanguy line describes the course of the frontal branch of the facial nerve. The line extending 0.5 cm below the tragus to 1.5 above the lateral brow with the nerve running on the undersurface of the temporoparietal fascia.4
The marginal mandibular branches—consistently lie on top of the facial artery and vein 1 to 3 cm below the mandibular border.5
NATURAL HISTORY
Axonal growth through the cross face graft proceeds at approximately 1 mm/d.
Ten to twelve months is typically required for the axons to traverse the graft.
After 18 months of denervation, mimetic muscles undergo significant atrophy and poor functional outcomes are associated with nerve grafting.
A narrow time window is present for the use of cross face nerve grafts as an isolated procedure to restore innervation to the paralyzed muscles of facial expression.
The therapeutic window of CFNG is enhanced by the addition of a nerve “babysitter.”6
Transfer of an adjacent cranial nerve to the injured facial nerve to achieve rapid reinnervation and motor end plate preservation.
Partial hypoglossal-to-facial nerve transfer with an interposition jump graft and secondary selective facial nerve branch transection and distal end-to-end repair to CFNG.
Masseter-to-facial nerve transfer with immediate CFNG and distal end-to-end and/or end-to-side coaptation.7
PATIENT HISTORY AND PHYSICAL FINDINGS
Etiology of the facial paralysis:
Developmental
Traumatic
Post-tumor extirpation
Bell palsy
Ramsay Hunt syndrome
Duration of paralysis:
Denervation periods greater than 18 months are associated with poor functional recovery (irreversible muscle atrophy) without utilization of a muscle flap.
Patient age:
Peripheral nerve regeneration declines with advanced age.
There is no definitive age cut off for cross face nerve grafting; however, after the age of 50 to 55 years, the ipsilateral motor nerve branch to masseter or other adjacent cranial nerves often provide a more reliable source of innervation to free muscle flaps with more powerful muscle flap contraction.
Comorbidities impairing nerve regeneration:
Smoking—higher risk of skin flap necrosis
Diabetes
Vascular disease
Previous radiation
Neurologic disorders
History of hypercoagulable state:
Patient or family history of pulmonary emboli or deep venous thrombosis signal the need for a hematologic workup if a subsequent free, functional muscle flap is planned.
Physical Examination
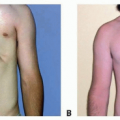
A strong smile, normal blink, forceful eye closure, good lip pucker, and depression without synkinesis should be present on the donor hemiface.
Perform a complete cranial nerve exam to look for additional deficits.
Examine the eye on the paralyzed side for signs of exposure keratitis.
Look for previous incisions that could compromise perfusion to the cheek flaps elevated during facial nerve exploration.
Palpate and Doppler the superficial temporal and facial artery and veins on the paralyzed hemiface if a subsequent free, functional muscle flap is planned.
Palpate lower extremity pulses if sural or saphenous nerve grafts are to be harvested to rule out peripheral vascular disease.
The progressive growth of axons through the CFNG can be monitored by eliciting a Tinel sign.
An active Tinel sign at the distal end of the cross face nerve graft suggests signals readiness for the next phase of the reconstruction.
IMAGING
MRI of brain and facial nerve
High-resolution multiplanar T1 and T2 images, gadolinium-enhanced, fat-suppressed, and internal auditory canal (IAC) protocol
Used to evaluate injury to the facial nerve (ie, Bell palsy, tumors, stroke)
Computed tomography
0.625-mm or thinner axial images, reconstructions in coronal plane
Valuable for imaging temporal bone fractures
CT angiogram
Useful for evaluating possible sites for microvascular anastomosis in patients with multiple head-neck surgeries or developmental differences including hemifacial microsomia.
EMG of the facial nerve
Look for signs of early facial nerve recovery.
Look for signs of reversible muscle atrophy.
SURGICAL MANAGEMENT
Preoperative Planning
Facial nerve EMG is obtained to evaluate for irreversible mimetic muscle atrophy.
Facial nerve imaging as guided by the etiology of the paralysis (MRI, CT, CT angiogram).
Determine the number of cross face nerve grafts to be utilized and evaluate the potential nerve graft donor sites.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree