8 Cranioplasty
Summary
The two most important variables to be considered for cranioplasty are whether the problem is related to contour (onlay) or protection of the brain (inlay) and the age of the patient. Because onlay cranioplasty generally is performed to improve an aesthetic deformity, we prefer to intervene when children are older and use an alloplastic material because there is less resorption compared to bone grafts. We favor porous polyethylene because it allows fibrovascular ingrowth and is easy to contour. Large full-thickness cranial defects necessitate closure to protect the brain. We allow spontaneous ossification to occur during infancy and intervene after this time. Particulate bone grafting/exchange cranioplasty is our first-line technique in children and adults because the bone grows with the child and completely osseointigrates. In adults with significant morbidities we will consider inlay cranioplasty using alloplastic materials to limit the extent of the operation
8.1 Introduction
Cranioplasty involves either the augmentation (onlay) or replacement (inlay) of cranial bone. Substances used for cranioplasty include (1) autologous bones (typically cranium, iliac crest, and rib) and (2) alloplastic substances (hydroxyapatite, demineralized bone, methylmethacrylate, porous polyethylene, and titanium). Advantages of autologous bone grafts are that they completely osseointegrate and thus provide strength and durability and have minimal risk of infection. Disadvantages of autologous grafts are limited availability, increased operating room time, and donor-site morbidity.
Alloplastic materials have three advantages: (1) supply is abundant, (2) there is no donor site, and (3) operative time may be shortened compared with autogenous techniques. However, these substances are not osteogenic (capable of new bone formation) and tend to be expensive. Demineralized bone is osteoinductive (able to induce transformation of undifferentiated mesenchymal cells to osteoblasts). The remaining substrates are either osteoconductive and provide a scaffold for bony ingrowth (e.g., hydroxyapatite and porous polyethylene) or have no biological activity (e.g., methylmethacrylate and titanium). Most bioactive alloplastic substances undergo incomplete revascularization and osseointegration and are susceptible to latent infection, foreign body reaction, displacement, and breakdown. The major variables that determine which material is used for cranioplasty are (1) whether the reconstruction is onlay or inlay and (2) the age of the patient.
8.2 Diagnosis
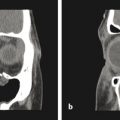
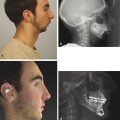
Most partial and full-thickness cranial defects can be diagnosed by physical examination. Large areas are noticed visually, especially if they involve a non–hair-bearing region. Moderate to large bony deficiencies can be palpated. Generally, patients who are planning on cranial reconstruction have imaging of their defect to facilitate planning of the procedure. Computed tomography (CT) is the most commonly used imaging modality because it best delineates the bone. CT subjects the patient to radiation and thus should be used sparingly. Although we typically obtain CT prior to an operation, we often do not obtain postoperative CT to assess the reconstruction if the procedure is successful clinically.
8.3 Nonoperative Treatment
Intervention for patients with osseous defects is not mandatory. Those with partial-thickness deficits may suffer psychosocial morbidity if the deficiency is causing a visible deformity. A concavity in a hair-bearing region can be camouflaged with a wig, hat, scarf, etc. Patients with frontal bone deformities may be able to grow their hair sufficiently to cover the abnormality.
Full-thickness defects less than 1 cm in an adult often heal spontaneously (critical-size defect). In children younger than 12 months, much larger areas of exposed dura may spontaneously ossify. Consequently, it is best to wait several months in adults with small full-thickness cranial defects or longer in infants with larger areas to allow as much spontaneous ossification as possible. Some individuals will no longer require intervention, and if cranioplasty is required, the area to reconstruct will be smaller.
Patients with small full-thickness defects often do not require cranioplasty to protect the brain from trauma. However, individuals with larger deficits are at a greater risk for brain injury from trauma. Helmeting can be used to protect the brain in patients awaiting cranioplasty (usually children who are most susceptible to incidental trauma).
8.4 Operative Treatment
8.4.1 Onlay Cranioplasty
Children
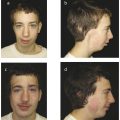
The primary indication for onlay cranioplasty is to correct a visual deformity to improve the patient’s self-esteem. Because the cranium has achieved more than 80% of its growth by the age of 4 years, elective procedures to improve appearance should be done after this time. In addition, a child’s self-esteem does not begin to form until approximately 4 years of age. Onlay cranioplasty is often performed when patients are older and verbalize that they are bothered by the deformity.
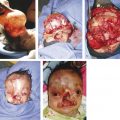
Although autologous bone grafts are preferred for full-thickness defects in children, their use for onlay cranioplasty in the pediatric population is uncommon. Because underlying osseous continuity exists, there is less concern that an alloplastic substance will restrict cranial growth or become dislodged with growth. Nevertheless, bone thickening will occur around the implant, and loss of contour over time is not uncommon. Some surgeons, however, prefer to use autologous bone grafts for onlay cranioplasty in children. Split cranial bone is the best autologous material. When harvested before 5 years of age, split cranial bone yields limited bone thickness and can be challenging due to a poorly formed diploic space. In situ harvest is not generally recommended before 9 years of age. Split graft is primarily cortical and heals by osteoconduction. The recipient site should not be burred to punctate bleeding, because enhanced vascularization will increase resorption of the graft through greater osteoclast activity. Cancellous bone graft from the iliac crest and particulate bone graft are not indicated for onlay cranioplasty because they resorb. These types of grafts require immediate vascularization to survive, which does not occur when they are placed on top of cortical bone.
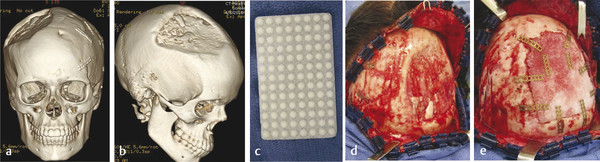
Drawbacks of split cranial bone for onlay cranioplasty are donor-site morbidity (e.g., injury to the dura, bleeding, and thinner bone at donor site) and resorption of the graft. Cortical grafts may undergo significant resorption when placed over bone, and thus, patients are at risk for an unfavorable outcome and repeat cranioplasty. Most surgeons prefer alloplastic materials for onlay cranioplasty (even in older children), because there is no donor-site morbidity and minimal resorption (Fig. 8‑1). However, nonautologous substances have a higher infection rate than bone grafts.
Adults
Because the cranium is almost fully grown by 5 years of age, concerns regarding dislodgement of alloplastic substances or restriction of growth are not as relevant after this time. Consequently, many surgeons prefer alloplastic substances instead of autologous bone grafts, because they shorten operative time, do not expose the patient to donor-site morbidity, and have minimal resorption. Although there are numerous alloplastic substances that are used for onlay cranioplasty, the most common are hydroxyapatite, methylmethacrylate, and porous polyethylene (Medpore).
Porous polyethylene is our preferred alloplastic material. It allows osseointegration/neovascularization at the periphery of the implant and is easily moldable to fit the partial-thickness defect. Although custom implants can be ordered based on CT images, contouring standard pieces to the defect is easy and less expensive. Bone substitutes (e.g., hydroxyapatite) allow some osseointegration at their periphery but are typically harder to contour than porous polyethylene and are more susceptible to fracture and infection. Methylmethacrylate is not commonly used for onlay cranioplasty, because it does not osseointegrate and has a higher fracture and infection rates compared with other materials.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree