7 Facial Fractures
Summary
Treatment of facial fractures in pediatric patients differs from adults. Different mechanisms of injury, dental anatomy, facial proportions, and bone quality result in unique injury patterns not seen in older patients. Treatment plans must account for the possibility that the injury and/or operative treatment can alter future facial growth. Additionally, with future growth and dental eruption, the pediatric facial skeleton is adaptive and minor malocclusions and subtly displaced factures may improve with time. Subsequently, pediatric facial fractures are treated less frequently with open reduction and internal fixation, and are more frequently treated conservatively. Open treatment is sometimes required. In these instances, careful considerations are needed to protect immature dental structures and to avoid or remove permanent implants that may restrict growth. Basic principles of patient assessment and operative/non-operative management of pediatric craniofacial trauma are discussed with special attention brought to the patterns of growth seen in the facial skeleton and how the anatomy at different stages of development should affect a surgeon’s clinical decision making when managing facial trauma in young patients. Unique fracture patterns, fixations techniques, and potential complications seen only in the pediatric population are discussed.
7.1 Introduction
Management of facial fractures in pediatric patients differs from that of their adult counterparts. Variations in anatomy, different daily activities, and the presence of ongoing growth account for these differences. Facial trauma is more common in children than in adults, but the facial skeleton is fractured less frequently. Pediatric patients account for 15% of facial fractures. When fractures occur, they affect areas of the face with different frequencies, occur in different patterns, and are more commonly greenstick fractures. The treatment goal is not simply to reestablish normal anatomy and function but to do so without disrupting future growth. The plasticity of the growing skull allows significant remodeling with growth, so more conservative modalities are often successful. When open reduction and fixation is necessary, surgeons must avoid injury to developing dental structures and growth sites, consider the effect that wide subperiosteal undermining may have on future growth, and recognize that fixation hardware has the potential to restrict growth or migrate into the bone or cranium. The specter of the fourth dimension haunts every surgeon caring for these injuries and must weigh on every management decision.
7.2 Patterns of Craniofacial Growth
Understanding facial development is key to understanding fracture patterns in pediatric patients and anticipating how they might affect future growth. At birth, the cranium is significantly larger than the face, with a proportion of 8:1. With pneumatization of the paranasal sinuses and eruption of the dentition, this proportion changes over time. At 5 years of age, this ratio shifts to 4:1 and reaches a ratio of roughly 2:1 at skeletal maturity. The majority of cranial growth is completed by the age of 3 years. The brain’s volume triples in the first year of life. The neurocranium is 25% of its adult volume at birth, 75% at 2 years of age, and 95% at 10 years of age. Growth is much more delayed in the facial skeleton, with the face achieving only 65% of its adult proportions by 10 years of age.
The cranium and the upper face grow as a result of brain and ocular expansion. During the early rapid expansion of the cranium, growth is dependent on bone deposition at the cranial sutures. As this process slows, further growth results more from appositional bone deposition. Although some growth remains, expansion of the cranium and orbits are nearly completed by 6 years of age.
The maxilla and midface continue to grow until roughly 12 years of age. Flattening of the cranial base with early brain development, growth at cranial base synchondroses, transduction of forces from nasal septal growth via the septovomerian ligament, and vertical alveolar elongation with dental eruption, all account for the forward and downward displacement of the maxilla. In addition, pneumatization of the paranasal sinuses contributes to maturation of the midface. All of the primordial paranasal sinuses have formed at the time of birth, but only the maxillary sinus is radiographically visible and clinically relevant. Ethmoid air cells begin to pneumatize in the first 2 years of life and continue to enlarge thereafter. The sphenoid and frontal sinuses are not radiographically apparent until after the age of 6 years and will continue to enlarge in early adulthood.
The mandible begins as two separate bones joined by a cartilaginous suture at the symphysis, which fuses during the first year of life. Mandibular growth continues with the rest of the face during childhood. The mandible continues to grow until skeletal maturity. Growth occurs both at the condylar head, which functions as a growth site, and through centripetal expansion. Condylar growth contributes to vertical and anteroposterior (AP) enlargement of the mandible. Eruption of dentition also adds to the vertical height of the alveolar segments, as it does in the maxilla.
7.3 Varied Fracture Characteristics in Children versus Adults
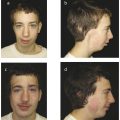
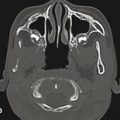
The high craniofacial proportion in early life and the lack of pneumatized sinuses account for the different fracture patterns seen in younger patients. In children younger than 6 years, the most common fractures are orbital and cranial fractures. These structures are proportionally larger and have greater prominence in the immature facial skeleton. Without a frontal sinus to absorb the energy of impact, fractures are prone to propagation into the frontal bone and along the orbital roof toward the apex. Fractures may also extend in an oblique trajectory into the midface, crossing either the nasal cavity or the buttresses surrounding the zygoma (Fig. 7‑1).
The midface is shielded from trauma and is more elastic and resistant to fracture in young children. Younger patients rarely exhibit the classic Le Fort pattern fractures seen in adults and adolescents. Immature bone is more cancellous and is less mineralized. The midface of younger patients is void of sinuses and is filled with dental follicles. Without aerated maxillary sinuses, force on the maxilla is transferred to the alveolus and results in dentoalveolar trauma. The palatal suture remains patent into adolescence and results in a higher incidence of palatal split fractures in children.
The lower mineral content and greater percentage of cancellous bone often result in deformations and greenstick fractures in children. When fractures require open reduction, they may require osteotomy to allow mobilization and proper reduction. In addition, dental eruption and remodeling can often overcome minor deformities and occlusal irregularities. Fractures that would clearly require open treatment in adults can be successfully managed with conservative measures and subsequent orthodontic correction in children. A new balance must be found while treating pediatric facial fractures when deciding what constitutes an operative case.
7.3.1 Diagnosis
Initial Assessment and Physical Examination
Evaluation of pediatric patients with facial trauma should begin with a comprehensive evaluation according to the principles of Advanced Trauma Life Support (ATLS) to avoid missing concurrent significant or life-threatening injuries. Another serious injury is present in 56% of patients. The most common associated injuries are neurologic, which occur in 47% of patients. Half of these injuries are concussions. Patients must be adequately screened and referred for appropriate follow-up. A low threshold for evaluation of the cervical (C)-spine and ophthalmologic consultation must be maintained as well. There is a 2.3 to 10% concurrence rate between facial fractures and C-spine injuries. Blindness has been reported in as high as 3% of orbital fractures, and 12% of orbital fractures have concurrent ocular injuries.
Diagnosing facial fractures in pediatric patients may be difficult. Unless the patient is being evaluated immediately after the injury, swelling will have begun to set in. This may mask subtle physical findings and asymmetries. If a clear indication for operative intervention is not apparent, a delayed reevaluation after the swelling is resolved is necessary to appreciate the resultant deformity. Examination of the available patient photographs, dental records, and occlusal models is helpful.
A comprehensive craniofacial examination from the vertex to the clavicles should be completed for all patients. The patient must be rolled, and the entire face and scalp must be examined and palpated for lacerations, ecchymosis, swelling, hematomas, and step-offs. Lacerations should be irrigated and probed for fractures prior to closure. A complete examination includes otoscopic examination of the tympanic membrane, ophthalmologic evaluation, examination of the C-spine, and palpation of the neck for sub-Q subcutaneous emphysema and expanding hematoma. If concern is raised for airway stability, the patient should be intubated. This may be complicated by the presence of a C-collar and/or midface or mandibular instability. Pediatric anesthesia may be of assistance in complicated cases, and tracheostomy and cricothyrotomy are always means of establishing a definitive airway in an emergency setting. Further evaluation includes a full cranial nerve examination.
Signs of orbital trauma such as swelling, ecchymosis, subconjunctival hemorrhage, enophthalmos, exophthalmos, diplopia, limited extraocular mobility, altered visual acuity, irregularity or asymmetry of the pupil, a relative afferent pupillary defect (RAPD), and medial or lateral canthal malposition should be noted. Careful intra- and extranasal evaluations should be carried out. Palpation of fractured nasal bones may reveal irregularities, crepitus, or mobility. Loss of nasal projection may indicate a complex nasal bone and septal injury, or it may indicate the presence of a naso-orbito-ethmoid (NOE) fracture when the intracanthal distance is increased. The nasal septum must be carefully evaluated for the presence of a septal hematoma, as failure to drain this may result in septal necrosis and a subsequent saddle-nose deformity.
Intraoral examination must also be carried out, with attention paid to dental trauma, intraoral lacerations, widened interdental distances, ecchymosis, and hematomas. These may indicate the presence of alveolar, maxillary, or mandibular fractures. Bimanual manipulation of the maxilla may indicate the presence of midface instability and a Le Fort fracture. A complaint of subjective malocclusion is sensitive but not specific for detecting fractures. This history is often unattainable in children and is therefore even less useful. A unilateral or anterior open bite may indicate the presence of condylar fracture. Trismus, or difficulty in opening the mouth, may be caused by a mandibular fracture, impingement by a depressed zygomaticomaxillary complex (ZMC) or zygomatic arch fracture, or swelling in the muscles of mastication from direct trauma. The mandibular condyle can be palpated by moving the jaw while placing one finger in the external auditory canal (EAC) and one finger in front of the ear. Laceration of the EAC on otoscopic examination may indicate a condylar fracture as well as a temporal bone fracture.
Radiographic Evaluation
Fine-cut (1–2 mm) maxillofacial computed tomographic (CT) scans have become the gold standard for assessing fractures of the craniofacial skeleton. Plain radiographs lack sensitivity, and adequate interpretation of these studies is becoming a lost art. The one possible exception to this is the pantomogram, or Panorex. This panoramic study provides a comprehensive, two-dimensional (2D) survey of the anatomy of the maxilla and mandible with low costs and radiation doses. It has comparable sensitivity to current CT scanners in identifying fractures of the mandible but has less inter-interpreter agreement when ruling out the presence of a fracture. Furthermore, it lacks the three-dimensional (3D) information obtainable from a CT study. These 3D reformats have become invaluable for characterizing fracture patterns and optimizing exposures and fixation strategies.
When caring for pediatric patients, surgeons must be thoughtful of the possible effects radiations have on the cognitive development of the growing brain and of the oncologic risk from such exposures. In a large retrospective study of the British National Health Service, obtaining two to three head CTs in patients younger than 15 years could lead to a threefold increase in the incidence of brain tumors. The absolute risk is less staggering. This number translates to only 1 additional tumor for every 10,000 scans performed. This risk is small, and although it must be considered, it should not dissuade a surgeon from obtaining studies needed for good clinical management.
7.4 Cranial/Frontal Sinus Fractures
7.4.1 Diagnosis
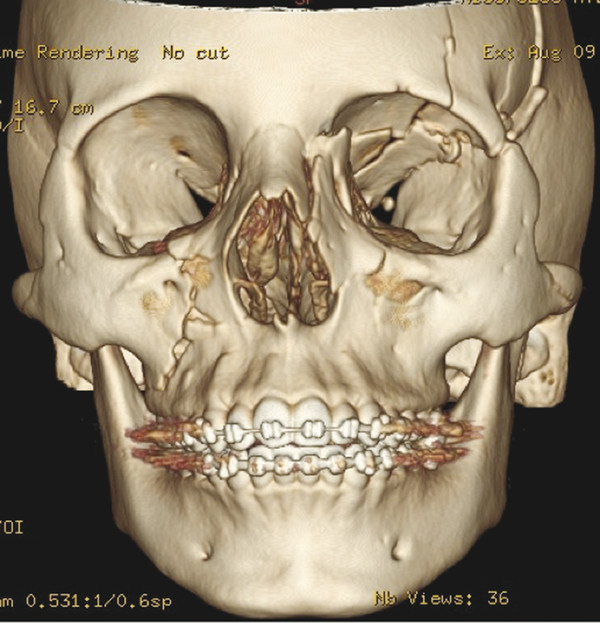
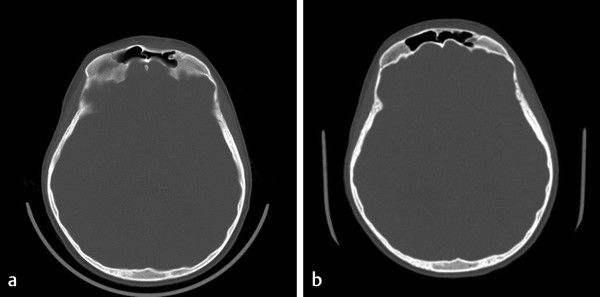
With the increased craniofacial proportions observed in small children, skull fractures are common and diminish in prevalence with age. In the authors’ series, they represented the second most common fracture type in children younger than 6 years. These injuries typically present as linear fractures that may extend into the orbital roof, skull base, nasal capsule, or zygoma in oblique trajectories. In children younger than 5 years, accompanying dural lacerations may result in a growing skull fracture, as pulsations cause fracture widening and herniation of cranial contents. This occurs in less than 1% of skull fractures. The parietal bone is the most common location, but it can occur in the frontal bone or orbital roof. It then results in frontal swelling or pulsatile exophthalmos (Fig. 7‑2).
Patients with frontal sinus, cribriform plate, and other cranial base fractures should be interrogated for cerebrospinal fluid (CSF) leaks. In compliant patients with a cleared C-spine, they should be asked to sit forward and observed for clear-liquid draining from the nose. They may complain of salty fluid in the pharynx. Edema, rhinorrhea, epistaxis, an unstable spine, or altered mental status may complicate this assessment. Cerebrospinal fluid will demonstrate a “ring sign” when bloody drainage is blotted on an absorbent surface. Clear CSF will migrate further than blood when absorbed. In addition, when available, a β-2 transferrin assay is highly sensitive in differentiating CSF from other body fluids. Otorrhea may indicate a temporal bone fracture with CSF leak. Otoscopic examination is necessary to detect this and other signs of temporal bone trauma such as hemotympanum and laceration of the EAC.
7.4.2 Nonoperative Management
The goals of managing cranial and frontal bone injuries are protection of the neurocranium, repair of dural injuries, resolution of CSF leak, prevention of infection, and correction of aesthetic/contour deformities. In the absence of significant displacement or step-off, nasofrontal outflow obstruction, pneumocephalus indicating dural injury, gross contamination, or underlying neurotrauma, nonoperative management is preferred. Owing to the rapid cranial expansion seen in the first 3 years of life, significant remodeling can be expected, and depressed skull fractures are more likely to be successfully managed nonoperatively in small children. Most CSF leaks will resolve spontaneously. Typically, a course of bed rest and head elevation is adequate. If this fails, a period of CSF diversion via a lumbar drain or external ventricular drain is indicated before surgery.
In patients with a frontal sinus, patency of the nasofrontal duct is paramount. Delayed abscess and mucocele may develop years after an injury if this structure is injured. In the setting of a patent duct and minimally displaced fractures, nonoperative management is warranted, but serial examination and repeat imaging 3 to 5 years later are necessary. Serial follow-up is necessary in younger patients with frontal fractures to clinically rule out the development of a growing skull fracture.
7.4.3 Operative Management
Surgical treatment of skull fractures in children is rarely needed. In the senior author’s series of isolated cranial fractures, 86% were managed nonoperatively; 7.5% required neurosurgical interventions such as external ventricular drain (EVD) placement, hematoma evacuation, and decompressive craniectomy; 6.5% underwent operative repair of the fracture, open reduction internal fixation (ORIF), frontal sinus repair, and fracture debridement. Frontal bone injuries were more likely to necessitate operative intervention due to the presence of the frontal sinus, orbital complications due to orbital roof involvements, and greater cosmetic sensitivity in this location.
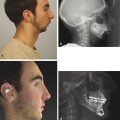
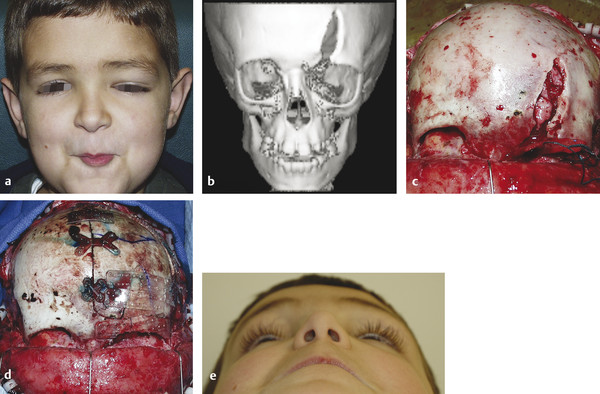
Frontal bone injuries can be approached directly through extensive local lacerations or via a coronal approach. Any child with remaining cranial growth potential must have fractures fixated with absorbable hardware, as metallic implants are known to translocate through the skull with future appositional growth (Fig. 7‑3). These plates and screws may become imbedded in dura, brain parenchyma, or dural sinuses and present significant potential morbidity. Growing skull fractures are repaired with craniotomy to allow access and repair of the underlying dural injury. Reconstruction then requires reconstitution of the normal anatomy, with cortical bone grafts in a configuration that places a contiguous piece of cortical bone overlying this repair.
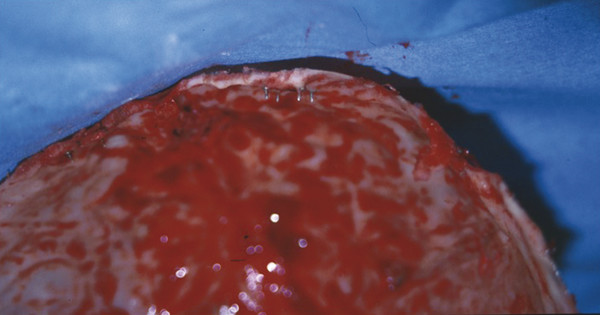
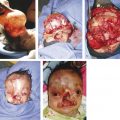
When a frontal sinus is present, the algorithm developed by Rodriguez and Manson represents the most evidence-based approach to managing these injuries. The essential decision-making factor is the presence of trauma and obstruction of the nasofrontal outflow complex. When the nasofrontal outflow complex is uninjured, displaced anterior wall fractures may be managed with simple ORIF, as long as duct patency can be documented intraoperatively. If the drainage pathway is either obstructed or likely to become obstructed due to scaring from an injury, the sinus must be defunctionalized. This requires complete removal of the sinus mucosa by burring the entire cortical surface, as epithelial cells are known to be present along draining venous sinuses in the bone and can serve as a tissue source for reepithelialization. The sinus duct should be filled with a well-fitting cortical bone plug to promote ossification. Management of the remaining sinus depends on the fracture patterns. In the setting of significantly displaced posterior table fractures, it is best to cranialize the sinus. The posterior table is removed, dural injuries are repaired, the obliterated sinus duct is covered with vascularized tissue such as a pericranial or galeal frontalis flap, and the cranial contents are allowed to expand into the frontal sinus space. When the posterior sinus wall is intact, the sinus may be managed by obliteration, in which the defunctionalized sinus is filled with various graft materials, including fat, fascia, and bone. Osteoneogenesis—a technique in which the duct is obliterated, the sinus is demucosalized, and the empty sinus is allowed to fill with bone and fibrous tissue over time—has also been described. Higher complication rates have been shown with the use of fat to fill the sinus dead space and with osteoneogenesis, and the authors’ preferred technique is to utilize bone, harvested locally from the cranium or the iliac crest. In a patient with a developing frontal sinus, the authors have demonstrated that even significantly displaced anterior table fractures have the potential of remodeling with time and continued pneumatization of the frontal sinus (Fig. 7‑4).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree