Cosmeceuticals
Michelle Henry, MD
Chapter Highlights
Cosmeceuticals are topically applied, over-the-counter skincare products containing key active ingredients.
Sunscreens are topical preparations that reflect or absorb ultraviolet radiation and can be physical, chemical, or combination products.
To be labeled as “broad-spectrum”, sunscreens must block both ultraviolet A and ultraviolet B radiation.
Retinoids are vitamin A derivatives used in topical preparations for their antiaging effects.
Various skin-lightning agents, including hydroquinone, retinoids, and ascorbic acid, are available in over-the-counter cosmeceuticals.
Individuals with sensitive skin must be familiar with and avoid common antigens in skincare products; these individuals may benefit from patch testing.
Increasingly, patients are looking for cost-effective noninvasive methods to prevent and reverse the signs of skin aging. Topically applied, over-the-counter skincare products containing key active ingredients, commonly referred to as cosmeceuticals, are the first line of defense against aging and are ubiquitously used for a variety of aesthetic indications. Compared to other noninvasive or minimally invasive strategies such as energy-based devices, fillers, and neurotoxins, the use of cosmeceuticals has the lion’s share of the market due to their ease of use and the fact that they are accessible to consumers regardless of their socioeconomic status. According to the latest data, the global cosmeceuticals market was USD 45.47 billion in 2017 and is estimated to reach 72.99 billion by 2023 at a compound annual growth of 8.21% during the forecasted period. In fact, the global cosmeceuticals market is outpacing all other product segments in the personal care products and cosmetics industry.1
Albert Kligman coined the term “cosmeceutical” in 1984 to refer to substances that exerted both cosmetic and therapeutic benefits.2 According to Kligman, a product should address three main questions to qualify as a cosmeceutical. First, does the active ingredient
penetrate the stratum corneum and reach in sufficient concentrations to its intended target in the skin? Second, does it have a known specific biochemical mechanism of action in the target cell or tissue in human skin? Finally, are there published, peer-reviewed, double-blind, placebo-controlled, statistically significant, clinical trials to substantiate the efficacy claims? Since cosmeceuticals are an amalgamation of cosmetics and pharmaceuticals, with the exception of sunscreens, they remain an unrecognized category by the US Food and Drug Administration and stringent regulatory pathways do not exist to guide research and marketing. Only recently have there been efforts to address quality control, substantiate marketing claims, and establish industry standards, since cosmeceuticals intend to deliver results on a higher level than cosmetics that simply color and scent the skin.3
penetrate the stratum corneum and reach in sufficient concentrations to its intended target in the skin? Second, does it have a known specific biochemical mechanism of action in the target cell or tissue in human skin? Finally, are there published, peer-reviewed, double-blind, placebo-controlled, statistically significant, clinical trials to substantiate the efficacy claims? Since cosmeceuticals are an amalgamation of cosmetics and pharmaceuticals, with the exception of sunscreens, they remain an unrecognized category by the US Food and Drug Administration and stringent regulatory pathways do not exist to guide research and marketing. Only recently have there been efforts to address quality control, substantiate marketing claims, and establish industry standards, since cosmeceuticals intend to deliver results on a higher level than cosmetics that simply color and scent the skin.3
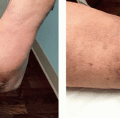
Applications of cosmeceuticals can range from improving skin radiance and texture, reducing acne, decreasing pigmentation, and by far the most sought-after indication, antiaging. Most cosmeceuticals are made from marine algae, fruits, herbs, botanicals, or cell culture extracts. This chapter will focus on cosmeceuticals used for skin protection, retinoids for antiaging, formulations for skin brightening, and products that are safe to use in sensitive skin.
Sunscreens are topical preparations containing filters that reflect or absorb radiation in the ultraviolet (UV) wavelength range. While sunlight is essential for vitamin D synthesis and can boost well-being, chronic or acute exposure to UV radiation (290-400 nm) can have detrimental effects on human skin such as sunburn, photoaging, and skin cancer. The UV radiation that reaches the earth’s surface contains 5% ultraviolet B (UVB) (290-320 nm) and 95% ultraviolet A (UVA) (320-400 nm). UVB radiation includes the most biologically active wavelengths that are responsible for sunburn, inflammation, and skin cancer, while UVA rays are those that significantly contribute to photoaging and play a major role in the development of sunspots, dyschromias, and fine lines.4,5,6
Topical photoprotection works primarily through either scattering and reflection of UV energy or absorption of UV energy. Many current sunscreens contain ingredients that work through both mechanisms in terms of UV protection. In the United States, where sunscreens are considered to be over-the-counter drugs that need to be FDA-approved, there are 17 different UV filters, while many other UV filters are available in Europe, Canada, and Australia.7,8,9 They are classified as organic (formerly known as chemical sunscreens) and inorganic (formerly known as physical sunscreens). Since no single agent effectively provides adequate protection from both UVA and UVB radiation, nearly all commercially available sunscreen products contain agents from both groups. Two or more sunscreen active ingredients may be combined with each other in a single product when used in the concentrations approved by the FDA for each agent. Broad-spectrum sunscreens are generally combinations of sunscreen products that are able to absorb both UVB and UVA radiation.
Organic filters (Table 9.1) include a variety of aromatic compounds that protect the skin by absorbing UV energy and transforming it into a negligible amount of heat energy.19 Specifically, the sunscreen chemical, after being excited to a higher energy state from its ground state via the absorption of UV radiation, returns to the ground state emitting energy in the form of longer wavelengths, typically as very weak red light or mild infrared radiation. The most common sunscreen organic agents, salicylates and cinnamates, both absorb UVB. Salicylates were the first UV chemical absorbers used in commercially available sunscreen preparations with a UV absorbance of about 300 nm. The salicylate
group of sunscreen agents includes octyl salicylate and homomenthyl salicylate. Among cinnamates, octinoxate is the most widely used UVB filter worldwide. Cinnamates, chemically related to balsam of Peru, coca leaves, cinnamic aldehyde, and cinnamic oil, have a peak absorption wavelength of about 305 nm. The chemical structure of the cinnamates, as a group, makes the molecule insoluble in water, requiring more frequent reapplication of the preparation.
group of sunscreen agents includes octyl salicylate and homomenthyl salicylate. Among cinnamates, octinoxate is the most widely used UVB filter worldwide. Cinnamates, chemically related to balsam of Peru, coca leaves, cinnamic aldehyde, and cinnamic oil, have a peak absorption wavelength of about 305 nm. The chemical structure of the cinnamates, as a group, makes the molecule insoluble in water, requiring more frequent reapplication of the preparation.
TABLE 9.1 Sunscreens | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||
Benzophenone derivates and anthranilates are effective at absorbing UVA radiation. Although the primary protective range for benzophenone is in the UVA range, a secondary protective band is also noted in the UVB range. The most commonly used benzophenone agents are oxybenzone and dioxybenzone. Although these ingredients are much less allergenic than first-generation sunscreen, they still carry a risk of contact allergy. Other agents such as avobenzone can be combined with UVB filters such as homosalate and octisalate to yield a broad-spectrum coverage.
Inorganic filters (Table 9.1) are mineral compounds, the most common being zinc oxide and titanium dioxide. These are believed to reflect and scatter UV light over a wide range of wavelengths, effectively serving as a physical barrier to incident UV and visible light.20 However, studies have shown that these compounds, and in particular micronized
preparations, absorb rather than reflect UV radiation. Their popularity has grown in recent years due primarily to their low toxicity profile. Aside from being effective in protecting against both UVA and UVB, these agents are fairly photostable and have not been shown to induce phototoxic or photoallergic reactions. Early formulations of physical sunscreen agents were not widely accepted because the particulate matters had to be incorporated in high concentrations, resulting in an opaque film on the skin that was not cosmetically acceptable. Newer formulations provide “micronized” formulations that allow for adequate protection while having a translucent appearance that leads to improved cosmetic results. Considering zinc oxide and titanium dioxide, the former was shown to provide superior protection for UVA in the 340 to 380 nm range and tends to be less pasty on the skin.
preparations, absorb rather than reflect UV radiation. Their popularity has grown in recent years due primarily to their low toxicity profile. Aside from being effective in protecting against both UVA and UVB, these agents are fairly photostable and have not been shown to induce phototoxic or photoallergic reactions. Early formulations of physical sunscreen agents were not widely accepted because the particulate matters had to be incorporated in high concentrations, resulting in an opaque film on the skin that was not cosmetically acceptable. Newer formulations provide “micronized” formulations that allow for adequate protection while having a translucent appearance that leads to improved cosmetic results. Considering zinc oxide and titanium dioxide, the former was shown to provide superior protection for UVA in the 340 to 380 nm range and tends to be less pasty on the skin.
When determining the efficacy of a sunscreen, the most important assay is the sun protection factor (SPF). The SPF refers to the ratio of the minimal dose of solar radiation that produces perceptible erythema (minimal erythema dose) on sunscreen-protected skin compared with unprotected skin. Thus, it measures a sunscreen’s ability to prevent development of erythema and sunburn upon exposure to primarily UVB radiation. The SPF value is assessed under experimental conditions using a light source that simulates the solar radiation on the skin of light-skinned volunteers who have applied an amount of sunscreen corresponding to 2 mg/cm2. Since SPF does not adequately measure protection from UVA, the FDA issued new regulations, effective in 2012, for labeling sunscreen products. Under these new regulations, only sunscreen products that can pass the FDA’s test for protection against both UVA and UVB rays will be labeled as “broad spectrum.”
Broad-spectrum sunscreen products that contain an SPF of 15 or higher may have the following statement on the label: “If used as directed with other sun-protection measures, decreases the risk of skin cancer and early skin aging caused by the sun.” Those that fail the broad-spectrum test or have an SPF <15 must add the following to their label: “Skin cancer/skin aging alert: Spending time in the sun increases your risk of skin cancer and early skin aging. This product has been shown only to help prevent sunburn, not skin cancer or early skin aging.” Moreover, the FDA no longer allows sunscreen products to be labeled “sweat proof” or “water proof.” Sunscreens can be labeled as “water resistant” or “very water resistant” if they maintain their SPF after 40 or 80 minutes of swimming or sweating, respectively. Sunscreen products with SPF 15 are generally recommended for daily use, whereas cosmetics that contain sunscreen (e.g., facial moisturizers, foundations) may improve the photoprotection compliance.21 Most cosmetic products are formulated to provide an SPF of 15 to 30 and may or may not be labeled as broad spectrum. Broad-spectrum sunscreen products with SPF 30 or higher are recommended for individuals performing outdoor work, sports, or recreational activities.
The benefits of sunscreens in protecting against the development of several conditions, from photoaging to skin cancer, have been documented in clinical studies. For example, both observational and randomized trials have demonstrated that sunscreens prevent the development of actinic keratoses and squamous cell carcinomas.22,23,24,25 Sunscreens, by protecting the skin against external stimuli that lead to aging, can also shield it against skin changes such as pigmentation and wrinkling.26 Broad-spectrum sunscreens with high SPF are generally used for the prevention of photodermatoses, which can be elicited by either UVB or UVA.
Despite its importance in protecting the skin and preventing skin disease, compliance still remains a challenge. Common reasons for noncompliance have included the stickiness of the product, which is greater with higher SPF, while cosmetic elegance, including texture, absorbability, absence of greasiness, and pleasant smell, was the most common positive feature of a sunscreen product cited by consumers.27,28 Since sunscreen
ingredients are oil-soluble, the most widely commercialized sunscreen products are oil-in-water emulsions, in which microscopic drops of oily materials are dispersed in a continuous water phase that also typically contains other polar ingredients like glycerin or glycols.29 Lotions are thinner and less greasy than creams and are generally preferred for application over large body areas. Adopting simple application techniques that can be incorporated in an individual’s daily routines can also assist with compliance. For example, the so-called “teaspoon rule” that involves applying approximately one teaspoon of sunscreen to the face and neck and each upper extremity and two teaspoons to the front, back torso, and each lower extremity can help adequate application of sunscreen.30,31,32 The time of application is also important as sunscreens should be applied 15 to 30 minutes before sun exposure to allow the formation of a protective film on the skin and should be reapplied at least every 2 hours.33
ingredients are oil-soluble, the most widely commercialized sunscreen products are oil-in-water emulsions, in which microscopic drops of oily materials are dispersed in a continuous water phase that also typically contains other polar ingredients like glycerin or glycols.29 Lotions are thinner and less greasy than creams and are generally preferred for application over large body areas. Adopting simple application techniques that can be incorporated in an individual’s daily routines can also assist with compliance. For example, the so-called “teaspoon rule” that involves applying approximately one teaspoon of sunscreen to the face and neck and each upper extremity and two teaspoons to the front, back torso, and each lower extremity can help adequate application of sunscreen.30,31,32 The time of application is also important as sunscreens should be applied 15 to 30 minutes before sun exposure to allow the formation of a protective film on the skin and should be reapplied at least every 2 hours.33
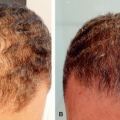
Retinoids have been used for decades in both therapeutic and cosmeceutical settings. The term retinoid refers to a class of substances comprising vitamin A (retinol) and its derivatives, both natural and synthetic. Precursors of retinol are retinyl esters and retinaldehyde that can be oxidized into retinoic acid which is the biologically active form of vitamin A.34,35 Retinol may also be esterified with fatty acids to form retinyl esters. Since retinoids are lipophilic molecules, they can diffuse through plasma membranes or cross the cutaneous barrier when applied topically. Inside the cells, retinol and its active metabolites can bind to nuclear receptors (retinoic acid receptor [RAR] or retinoid X receptor [RXR]). Then, the ligand-receptor complexes bind to a RAR-response element DNA sequence, resulting in the modulation of the expression of genes involved in cellular differentiation and proliferation.36,37,38 This leads to an increase of procollagen production, inhibition of inflammatory mediator release, reduction of collagen breakdown enzymes such as MMPs, and improvement of dermal vasculature. Retinol-dependent biological cascades can lead to increased epidermal cell renewal, expansion of cell layers, strengthening of the skin barrier, and collagen remodeling.
Therapeutic retinoids are typically RAR or RXR ligands that are prescribed to treat conditions such as acne, psoriasis, actinic keratosis, and some types of cancer.39 Other retinol metabolites that can bind to retinoid receptors and affect gene expression such as tretinoin, alitretinoin, isotretinoin, adapalene, and tazarotene are considered therapeutic retinoids.40
On the other hand, precursors of retinoic acids, such as retinyl esters; retinol; retinaldehyde; 4-oxoretinol, 4-oxoretinal, and 4-oxoretinoic acids that do not bind nuclear retinoid receptors, are considered and used in topical cosmeceutical products.38 These agents have been used to prevent and treat aging skin for decades with retinoic acid having the strongest activity, followed by retinaldehyde, retinol, and finally retinyl esters.
Retinoic acid is considered the gold standard in antiaging topical therapy. It is the most extensively investigated topical retinoid, and its safety profile is well established. In the United States, retinoic acid is available by prescription and exists in cream, gel, microsphere gel, and emollient base formulations under branded names and as generic drugs. The optimal concentration needed to have antiaging benefits has not been established, but commercial products to date are available in 0.025%, 0.05%, and 0.1% formulations. Prior to initiating treatment with topical retinoids, patients should be warned that local side effects such as skin irritation, redness, scaling, dryness, burning, stinging, and peeling can occur, that peak during the first 2 weeks and subside thereafter.41 Tretinoin, tazarotene, and adapalene can be used for treatment of photoaging
and all three substances have been validated in clinical studies. In a meta-analysis of 12 randomized trials, it was shown that application of tretinoin cream once daily in concentrations of 0.02% to 0.1% for 16 to 48 weeks was more effective than placebo in overall improvement of photodamage.41 A more recent trial evaluating the long-term efficacy and safety of daily application of tretinoin 0.05% cream in 204 subjects with moderate to severe photoaging demonstrated significant improvement in all signs of photodamage compared to placebo.42 Tazarotene, typically reserved for individuals without skin sensitivity, has also been shown to be safe and efficacious for the treatment of photoaging at a concentration range of 0.05% to 0.1%.43 Adapalene is the mildest type of prescription strength retinoic acid that has been shown in the concentration range of 0.1% to 0.3% to improve the appearance of cutaneous photoaging and fine lines.44 Retinoic acid improves several manifestations of photoaging, including textural changes, dyschromias, and fine wrinkles within the first few weeks of usage. Changes such as improvement of wrinkles are not seen until at least 2 to 4 months of continuous application.
and all three substances have been validated in clinical studies. In a meta-analysis of 12 randomized trials, it was shown that application of tretinoin cream once daily in concentrations of 0.02% to 0.1% for 16 to 48 weeks was more effective than placebo in overall improvement of photodamage.41 A more recent trial evaluating the long-term efficacy and safety of daily application of tretinoin 0.05% cream in 204 subjects with moderate to severe photoaging demonstrated significant improvement in all signs of photodamage compared to placebo.42 Tazarotene, typically reserved for individuals without skin sensitivity, has also been shown to be safe and efficacious for the treatment of photoaging at a concentration range of 0.05% to 0.1%.43 Adapalene is the mildest type of prescription strength retinoic acid that has been shown in the concentration range of 0.1% to 0.3% to improve the appearance of cutaneous photoaging and fine lines.44 Retinoic acid improves several manifestations of photoaging, including textural changes, dyschromias, and fine wrinkles within the first few weeks of usage. Changes such as improvement of wrinkles are not seen until at least 2 to 4 months of continuous application.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree