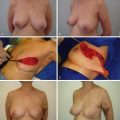
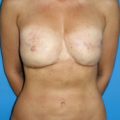
Key Words
tuberous breast deformity, tubular breasts, constricted breasts, breast asymmetry, congenital breast deformity, areolar herniation, dual plane breast augmentation, breast implants
Introduction
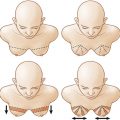
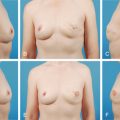
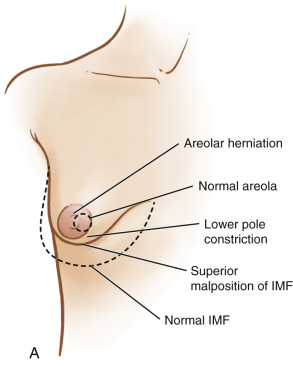
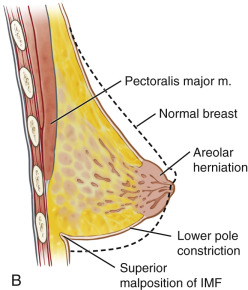
Tuberous breast deformity is a congenital anomaly with widely variable morphologic presentation. The etiology remains uncertain, although it is postulated that the deformity has an embryologic origin with a possible genetic predisposition; however no clear link has yet been determined. The physical manifestations of tuberous breasts have been attributed to periareolar fibrous ring constriction and hypoplastic, elastic areolar fascial support, the combination of which predisposes to narrow ventral-vector development of the breast bud into a more pliable and less resilient areola, resulting in a peripherally constricted appearance of portions of the breast with herniation of the areolar complex ( Fig. 24.1 ). The most frequently observed characteristics of tuberous breast deformity include breast base constriction, parenchymal hypoplasia, lower pole skin deficiency, superior malposition of the inframammary fold (IMF), areolar herniation, and ptosis, as demonstrated in Fig. 24.2 . The physical manifestations can vary widely in severity, with one, several, or all of these features present ( Fig. 24.3 ). Asymmetry is also a frequent hallmark of tuberous breast deformity, with discrepancies seen in breast size, shape, areolar dimension and herniation, and degree of ptosis.
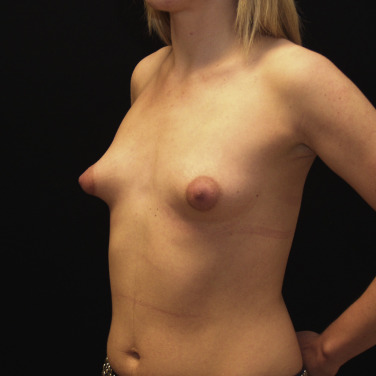
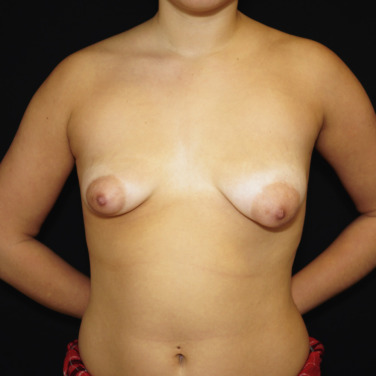

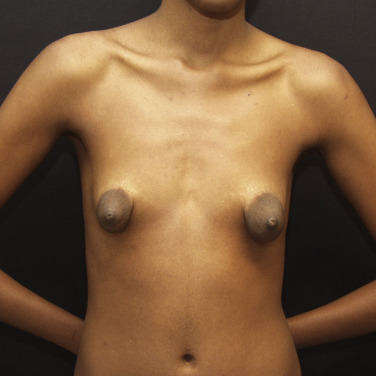

Tuberous breast deformity can present exceptional challenges for surgeons as much from the subtle cases as the severe. The technical demands of enhancing or creating a lower breast pole, controlling areolar herniation, correcting ptosis, and improving symmetry of breast size, shape and areolar dimensions are frequently substantial. Tuberous breasts are often underappreciated and underdiagnosed, with patients often expecting, and surgeons regrettably performing, a standard breast augmentation.
Identification and classification of the presence and severity of each feature of the deformity, or combination thereof, is critical in the establishment of a treatment plan. Von Heimburg, Grolleau, Meara, and Costagliola have proposed classification systems to define the spectrum of tuberous breast deformity. We prefer employing a three-tiered classification that incorporates the specific pathologic hallmarks of the deformity ( Fig. 24.4 ), and that facilitates the formulation of a treatment strategy that can be tailored on an individual basis to all patients based on morphology and deformity severity ( Table 24.1 ).
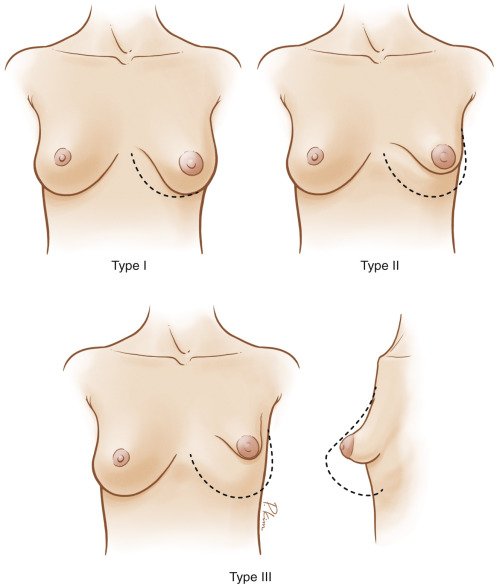
| Type | Base | Inframammary Fold | Skin Envelope | Breast Volume | Ptosis | Areola |
|---|---|---|---|---|---|---|
| I | Minor constriction | Normal laterally, minor elevation medially | Sufficient | Minimal deficiency, no deficiency, or hypertrophy | Mild, moderate, or severe | Enlargement |
| II | Moderate constriction | Medial and lateral elevation | Inferior insufficiency | Moderate deficiency | None or mild | Normal, mild or moderate herniation |
| III | Severe constriction | Elevation of entire fold, or fold absence | Global insufficiency | Severe deficiency | Mild/moderate | Severe herniation |
The keys to success in tuberous breast correction include the interruption of the fibrous ring constriction, release and expansion (in one or more stages) of lower pole deficiency, establishment of an appropriately positioned IMF, overcoming pre-existing IMF memory, augmenting the deficient lower pole for both volume and shape maintenance, reduction of areolar herniation, balancing areolar dimensions, correcting ptosis, and correcting asymmetry.
Indications and Contraindications
Women with any degree of tuberous breast deformity may be candidates for surgical correction, as the effects of the developmental anomaly are most often psychological, related to the self-perception of having “abnormally shaped” breasts. Ideal candidates are women with tuberous breast deformity who have completed breast development, and optimally have been at a plateau of stable breast shape and size for one year. Around 18–19 years is a common age for initial consultation and treatment. Consideration may occasionally be given to younger girls (15–17 years) with marked asymmetry, deformity or extreme emotional distress, with the caveat that continued breast development may increase the likelihood of a secondary surgery. In such cases, particularly in those with unilateral severe hypoplasia, the use of tissue expanders or adjustable implants may be beneficial to remain apace with potential progressive breast growth. Surgery before 15 years of age is not recommended.
Not unique to women, tuberous breast deformity is also seen in men as a variant of gynecomastia with periareolar fibrous ring constriction and areolar herniation. The technical considerations in male tuberous deformity differ in that the treatment is entirely reductive, and no effort is required to release, augment or enhance the breast lower pole.
Preoperative Evaluation
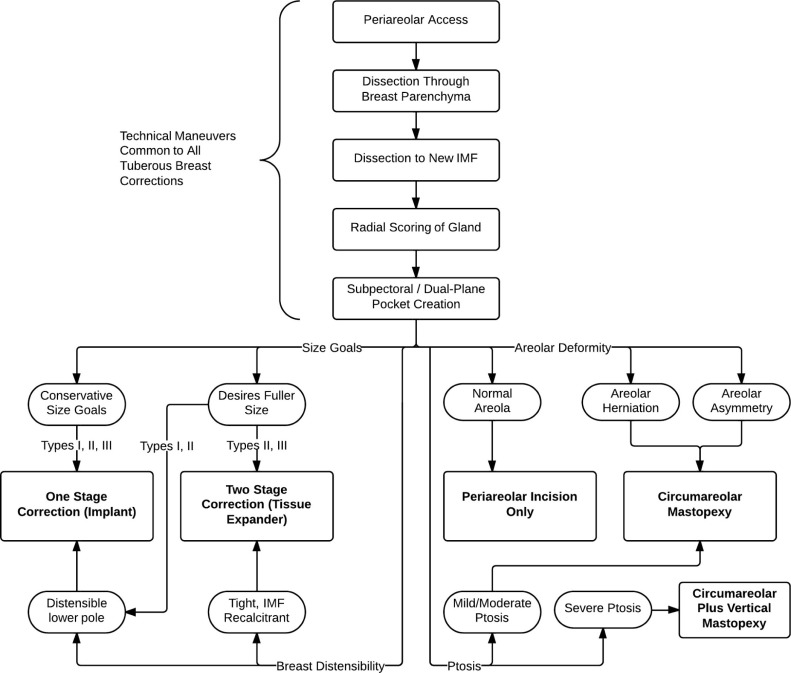
Detailed history and physical examination include measurements of breast dimensions and careful evaluation of breast base constriction, IMF position, skin envelope sufficiency and distensibility, breast mass, ptosis, areolar herniation, and any asymmetry. Of foremost importance in the treatment of tuberous breast deformity is the careful identification and stratification of all anatomic features. This assessment facilitates the categorization of each patient using the three-tiered classification system, and to further establish a treatment algorithm tailored to specific needs of the individual. Fig. 24.5 demonstrates the algorithm that is used to establish a treatment strategy for each patient based on the anatomic features noted above as well as the patient’s desires.
Surgical Techniques
Relevant Surgical Anatomy
The pathologic anatomy of tuberous breast deformity is demonstrated in Fig. 24.2 . The anatomic hallmarks can manifest as mild, moderate or severe expressions of the following: constriction of the breast base, hypoplasia or hyperplasia of breast parenchyma, skin deficiency of the breast lower pole, a high-riding or superiorly malpositioned IMF, herniation of the areolar complex, and ptosis. The surgical options for treatment are informed by the presence and severity of the individual features. Once carefully identified, the authors’ treatment algorithm ( Fig. 24.5 ) aids in addressing each facet of the deformity and the establishment of a surgical treatment plan.
Classical Technique Descriptions
Historically, the approaches to tuberous breast deformity correction have included a variety of incisional access sites (areola, IMF, axillary) and multiple techniques for parenchymal reshaping, and methods to control areolar herniation. Rees and Aston favored prepectoral implant insertion and circumareolar mastopexy in the setting of herniation of the areolar complex, and inframammary incisions with glandular scoring and prepectoral implant placement in the setting of constricted breast base. In 1990, Puckett and Concannon described an “unfurling” technique using a periareolar access incision, performing a subcutaneous plane dissection of the lower pole, followed by subglandular dissection to the midpoint of the breast mound. The breast parenchyma is then “split” horizontally from its deep surface, creating a superiorly based glandular flap that is “unfurled” and sutured to the inferior pole after placement of a subpectoral implant. Ribeiro and colleagues described a circumareolar de-epithelialization, after which the breast parenchyma is dissected down to prepectoral fascia to create superior and inferior breast flaps. The inferior breast parenchyma is then dissected free from the overlying subcutaneous tissue. The medial and lateral “prolongations” of the inferior glandular flap are excised, helping to disrupt the fibrous ring. The inferior glandular flap is then folded over itself to improve lower pole fullness and breast projection. The circumareolar incision is then closed using a round-block suture technique. We have found the majority of these approaches dissatisfying in the achievement of consistent results.
Among the fundamental underpinnings of our strategy are periareolar access, transglandular dissection, and radial glandular scoring, all of which play a role in releasing the fibrous ring constriction that is present to varying degrees in all tuberous breast morphology. We have found little utility in the use of parenchymal flaps, and prefer bisection of the gland through the areolar access, maintaining a lower pole “dermoglandular flap” that can be addressed with scoring maneuvers to improve shape. We favor a dual plane dissection and positioning of all breast implants for tuberous correction, and employ interlocking circumferential purse-string sutures for control of areolar herniation.
Periareolar Access Incisions
The periareolar approach affords the most flexibility and predictability, and we employ this approach exclusively for the correction of tuberous breast deformities. Areolar access is superior to an IMF incision because the final location of the new IMF is often extremely difficult to precisely determine, and an areolar incision obviates an inframammary scar that can be superiorly or inferiorly malpositioned. Additionally, the areolar approach also affords access to a dissection plane directly through the subareolar breast parenchyma, effectively releasing any internal glandular ring constriction that may exist with the deformity. Finally, any areolar positional asymmetries, shape abnormalities, and herniation are readily adjustable via the areolar approach.
Transglandular Dissection and New IMF Definition
Through the areolar incisional approach, dissection in a perpendicular fashion through the gland establishes a lower pole dermoglandular flap that allows for both contouring of the lower pole and for maintenance of soft tissue coverage. This initial dissection directly through the gland effectively bisects the gland in an equatorial plane, “breaking the ring” in two places. (When cutting a ring with any integrity in one place, it is likely to maintain its shape, and a minimum of two cuts are required to “break” its circular form to allow it to be more easily reshaped). Further constriction release and additional “break-points” in the “ring” are later created with incisions made radially, perpendicular to the vector of constriction, helping to more reliably overcome the memory of the pre-existing deformity.
After dissecting through the breast parenchyma to the prepectoral fascial plane, dissection is further developed inferiorly to the proposed new limit of the breast lower pole. Care must be taken not to undermine past this proposed line; it is simple to adjust inferiorly if necessary and considerably more difficult and time consuming to re-establish if disrupted. This initial prepectoral dissection also serves as the first component of the dual plane dissection.
Radial Scoring
The release of glandular and breast base constriction with radial scoring maneuvers is employed in all cases. With the deep surface of the gland exposed, electrocautery scoring of the gland is performed. The constricting horizontally oriented fascial bands within the gland lower pole and along the pre-existing IMF are incised in a perpendicular (vertical/radial) fashion from the deep subglandular plane, progressing superficially to the point of release. The periareolar transglandular approach is invaluable for facilitating the exposure to this lower pole subglandular plane, and for radially dividing the fascial constriction. The extent and depth of scoring is predicated on the degree of constriction, and the endpoint is the expansion of the breast lower pole and the visual release of any pre-existing, native IMF memory. In cases of more severe soft tissue and native fold constriction, the radial scoring maneuvers can be carried to the subdermal plane if necessary.
The glandular parenchymal division and scoring maneuvers over a large surface area are often productive of considerable serous fluid effluent, hence closed suction drains are used in all cases. Volumes are not uncommon.
Implant versus Autologous Reconstruction
Despite accounts of breast glandular division and transposition, we feel that the amount of breast parenchyma is often diminutive, and have found only rare circumstances with enough autologous breast tissue to satisfactorily augment the hypoplastic lower pole. Breast implants are standard-of-care in cases of micromastia or relative hypoplasia for the purposes of augmenting breast size, improving lower pole shape, and long-term maintenance of the constriction release achieved by radial scoring maneuvers and re-establishment of a new IMF position.
While autologous fat grafting has been described as an augmentation modality in tuberous breast correction, consistency of outcomes and patient satisfaction have been found to be limited compared to implant based correction.
Dual Plane versus Subglandular
From the earliest reports of tuberous breast deformity correction, and prevailing today, the most common position for placement of an implant has been in the subglandular space. Subglandular placement enables the prosthesis to shape and expand the breast without being restricted by the pectoralis major muscle. Unfortunately, the negative sequelae of subglandular implant placement, including increased contracture rates, visible implant margins and rippling, late malposition, and unnatural breast appearance can ensue, and may be even more prevalent after tuberous breast correction as a result of the necessary scoring and thinning of the parenchyma. This is why dual plane augmentation is our preferred approach.
In consideration of the relationship of the pectoralis muscle to the gland and the implant during subpectoral breast augmentation, it is clear that to varying degrees all submuscular breast augmentations are dual plane by definition, with the implant in the breast lower pole effectively subglandular, and in the upper pole submuscular. In tuberous deformity correction in particular, the lower pole is the region of the breast that requires the most “expansion” and implant-related shape definition, with little need for more aggressive fill and expansion of the upper pole, hence dual plane submuscular augmentation is ideal. Dual plane maneuvers also result in the craniad shift of the inferior incised margin of the muscle, away from the region of the released pre-existing IMF. The result is an uninhibited fill of the lower pole, while promoting the many advantages of submuscular augmentation including softer, more natural contour of the upper pole, increased soft tissue coverage, more durable breast position, and decreased capsular contracture rates. Additionally, with our technique of preserving the inferomedial origins of the pectoralis major muscle, muscle coverage of the implant along the décolletage maintains a natural appearance.
Implant Selection
For one-stage tuberous breast correction, there are only subtle differences in the use of silicone gel implants vs saline implants, or round cohesive gel implants compared with anatomic form-stable highly cohesive gel implants; we feel the implant choice may be the least consequential of all of the steps of the multifaceted process. Nevertheless, we currently favor highly cohesive gel form-stable implants for use in most one-stage tuberous breast corrections. These firmer anatomic implants, with increased silicone crosslinking and greater cohesivity, may confer an advantage of exerting more direct force on the breast base and lower pole, thus maximizing the stretch of the released tissues. These implants also appear to be associated with a low incidence of rippling and capsular contracture. It should be noted that our previously reported experience using smooth round cohesive gel implants exclusively yielded acceptable and consistent aesthetic results, high degrees of patient satisfaction, and modest complications, and these implants remain a viable option for correction. When two-stage correction is undertaken, the final implant selection at the second stage exchange procedure is even less critical as the expansile “work” has already been achieved by the tissue expander.
One versus Two Stage
Correction in one stage is achieved in the majority of cases. When deficient lower pole skin and soft tissue is distensible, and particularly when size goals are conservative, single stage correction is most often attainable. In cases of moderate and severe breast hypoplasia (types II and III) where there is severe constriction of the skin envelope and a tight, recalcitrant IMF, or if the patient desires a fuller result than the deficient lower pole skin and soft tissue will allow in one stage, two-stage correction with tissue expander placement is advisable. In instances where it becomes clear intraoperatively, despite all radial scoring maneuvers, that the pre-existing IMF memory cannot be overcome sufficiently during the first stage, tissue expander insertion is recommended. Additionally, two-stage correction can provide for a planned return for a “second look,” enabling adjustments in areolar position and shape, and fine-tuning of breast symmetry.
One of the more arduous tasks in the selection of a two-stage approach with expanders is patient education. We uniformly have tissue expanders available for cases wherein the lower pole and inframammary crease distensibility is in question, and educate patients accordingly. While every effort is made to achieve safety and consistency in aesthetic results in one stage, it is better for both surgeon and patient to strive toward a superior result in two stages than a suboptimal outcome, subject to the likelihood of revision surgery, in one.
Mastopexy and Asymmetry Considerations
Breast ptosis requires clear identification in every case when formulating a treatment plan. We have found that more classical grading of breast ptosis (grade I, II, III, pseudoptosis) does not adequately aid in the description and treatment planning of tuberous ptosis. With inframammary crease malposition, deficiency of lower pole skin and soft tissue, and frequent enlargement, malposition and herniation of the areola, we commonly see relative glandular hypertrophy and breast ptosis that defies standard classification. With the areolar position well below the pre-existing IMF, suggesting a grade II or III ptosis, the nipple-to-IMF distance is often quite short in tuberous breast deformity. Whereas short-scar, circumvertical mastopexy incisions or extended incisions may be required for non-tuberous moderate and high grade ptosis, more limited incisions are often sufficient in tuberous ptosis. After the often necessary maneuvers to lower the IMF and recruit and expand the lower pole, the “lift” of the breast and areola can often be adequately managed with a circumareolar incision.
Breast size asymmetry is adjusted with the selection of different implant volume, projection, or both, and/or by the removal of excess gland from the larger breast. We use gland excision when possible, as it affords the opportunity to remove a saucer or wedge of parenchyma from the deepest aspect of the subareolar gland (which may minimize the herniation), and keeps the implant sizes more similar, theoretically lending to longer-term durability of results, however the amount that can be safely removed is generally less than 30–40 g. Suction-assisted lipectomy can be employed in select cases, however this option is frequently limited given the relative youth of this patient population and attendant significant breast glandular density.
Areolar Herniation Control
When areolar herniation and deformity are identified, a circumferential approach is warranted. Round-block suture technique has been described for both circumareolar mastopexy and for reducing areolar diameter and projection. We have employed the interlocking purse-string suture technique described by Hammond, which has promoted consistency in areolar diameter as well as excellent correction by a more uniform flattening of the areolar projection.
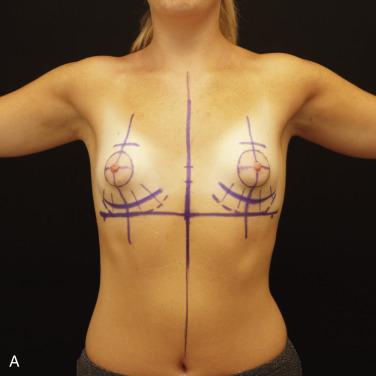
Preoperative Markings
All patients are marked preoperatively in the upright position. A vertical midline is marked from the sternal notch through the xiphoid process and to the mid-epigastric midline. The existing IMF lines and the periphery of the areolar-complex margins are then marked. With the patient’s arms elevated above her head, the level of the nipple is transposed to the midline. A caliper is then used to measure the breast base; one half of this number (the “radius”) is then calculated, and this measurement is transposed to the midline to establish the level of the new IMF. A horizontal line is then drawn through this point, perpendicular to the vertical plumb line, and across the anterior chest beneath each breast. Limits of planned dissection, locations of glandular scoring, and proposed periareolar/circumareolar patterns are marked, and anticipated adjustments of areolar size or position are also marked ( Fig. 24.6 ).